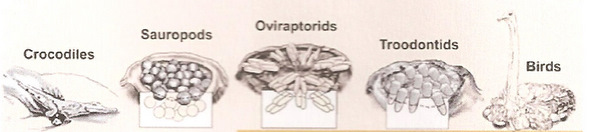
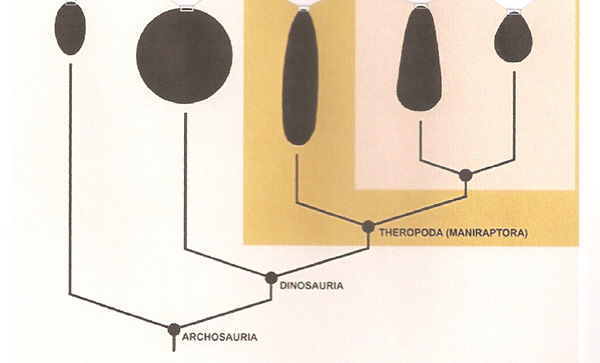
The dinosaur and theropod ancestors of birds were building nests in the early Jurassic
and even the late Triassic. This indicates that the even the earliest birds built nests to protect their eggs and young (From: Chiappe 2009).
Avian Reproduction: Nests
Birds use nests to protect eggs and nestlings from predators
and adverse weather. To minimize predation, birds may use or build nests
that are inaccessible, hidden, or camouflaged. Nests may also help keep
eggs and nestlings warm.
  The dinosaur and theropod ancestors of birds were building nests in the early Jurassic and even the late Triassic. This indicates that the even the earliest birds built nests to protect their eggs and young (From: Chiappe 2009). |
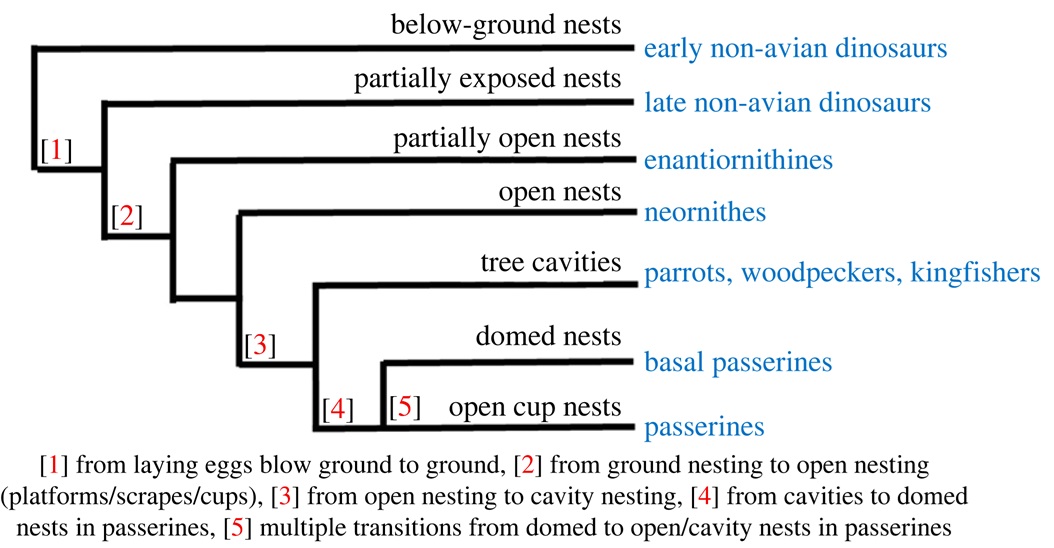
The evolution of nest characteristics in birds and their non-avian ancestors.
The evolution of nest site use and nest architecture in the non-avian ancestors of birds remains poorly understood because nest structures do not preserve well as fossils. Nevertheless, available evidence suggests that the earliest dinosaurs probably buried eggs below ground and covered them with soil so that heat from the substrate fuelled embryo development, while some later dinosaurs laid partially exposed clutches where adults incubated them and protected them from predators and parasites. The nests of euornithine birdsthe precursors to modern birdswere probably partially open and neornithine birdsor modern birdswere probably the first to build fully exposed nests. The shift towards smaller, open cup nests has been accompanied by shifts in reproductive traits, with female birds having one functioning ovary in contrast to the two ovaries of crocodilians and many non-avian dinosaurs. The evolutionary trend among extant birds and their ancestors has been toward the evolution of greater cognitive abilities to construct in a wider diversity of sites and providing more care for significantly fewer, increasingly altricial, offspring. The highly derived passerines reflect this pattern with many species building small, architecturally complex nests in open sites and investing significant care into altricial young (Figure and text from: Mainwaring et al. 2023).
| Orientation and microclimate of Horned
Lark nests -- Across their range, Horned Larks
typically construct nests adjacent to & north of objects such as a
tuft of grass or a rock. Hartman and Oring (2003) studied the importance
of nest orientation to nest microclimate in Horned Larks in California.
Nests showed a significant northern bias in orientation angle and were
49% shaded in the early afternoon, the hottest part of the day. Artificial
nests of eastern, western, and southern orientations exhibited little to
no shade during this time. A northern nest orientation ensures maximal
shading by the grass tuft to the south, may protect nests from cool evening
winds, and provides increased daytime ventilation of the nest through exposure
to prevailing winds. In addition, shade may also help conceal nests from
predators.
|
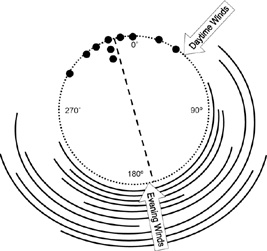
The position of vegetation (curved black bars) around Horned Lark nests relative to the direction of prevailing winds. The dashed circle represents the outline of a nest. Each individual curved black bar represents the vegetation around one nest. Each solid circle represents one entrance orientation. The dashed line designates the mean entrance orientation angle of 345° for all nests (n = 10; Hartman and Oring 2003). |
Types of nests:
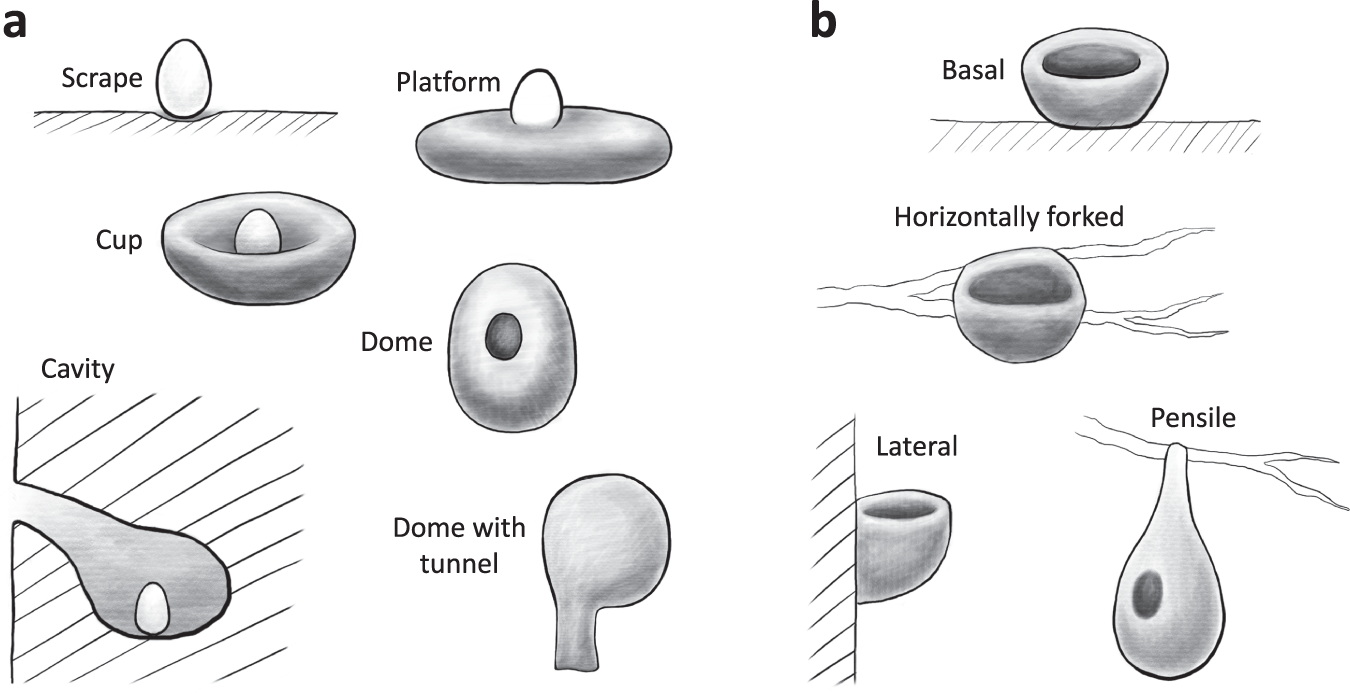
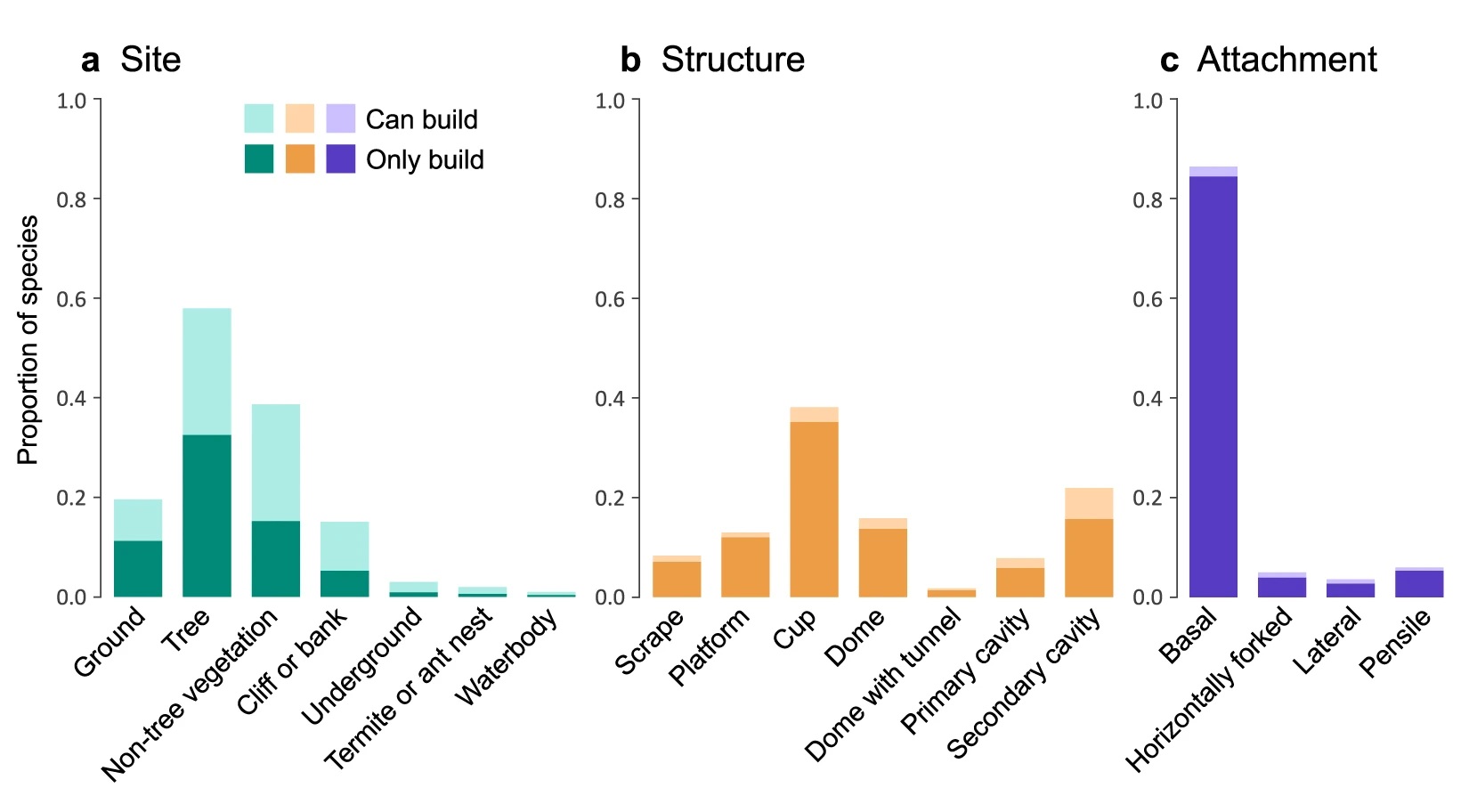
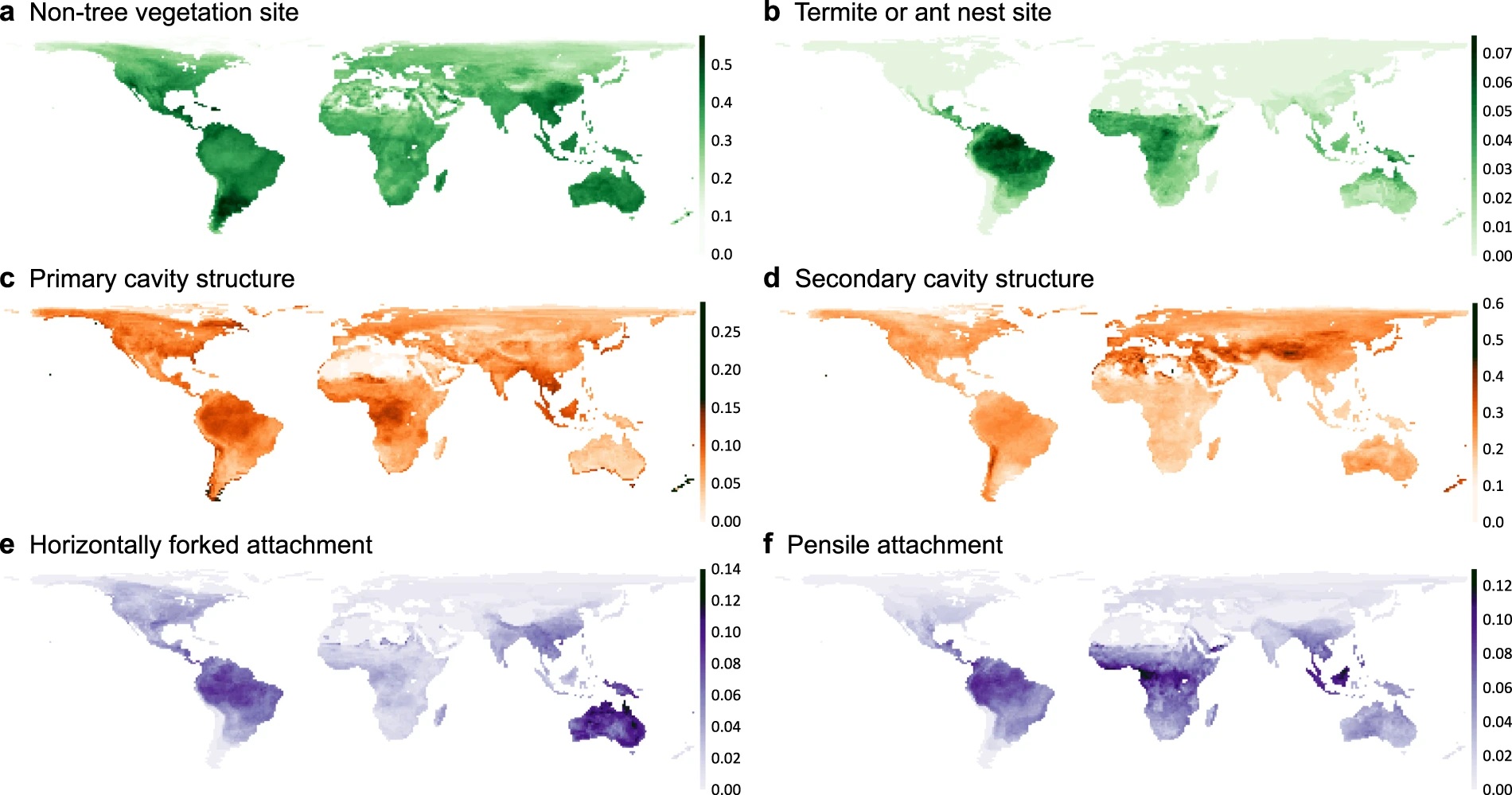
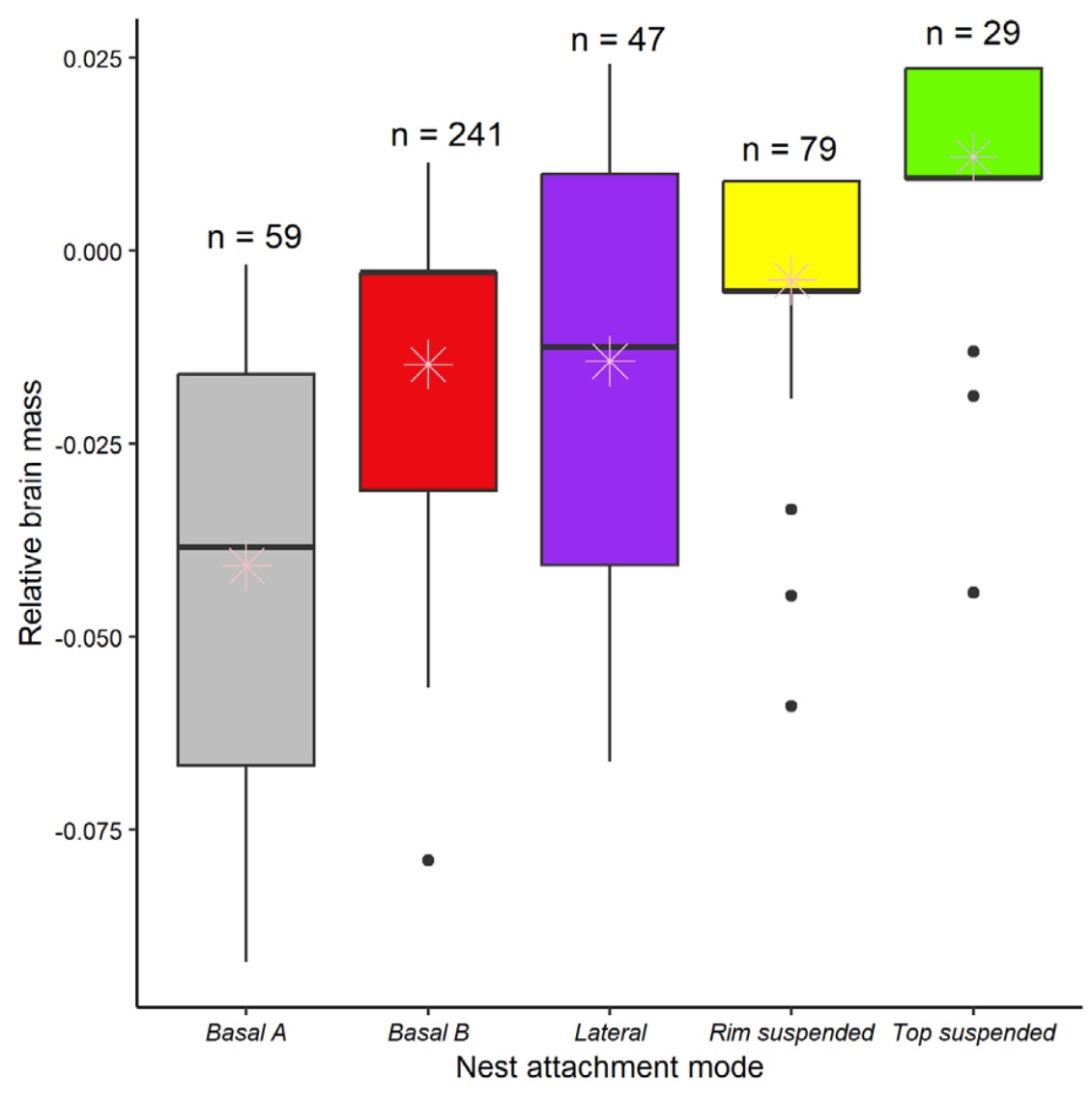
Model-predicted relative brain mass values across different nest attachment methods, with complexity
of attachment methods increasing from left to right. Colored boxes represent the 25% quartiles, whiskers represent
the 75% quartiles, horizontal lines represent the medians, asterisks represent the average values, points
represent outliers and n is the number of species of birds with nests of each attachment type. Basal A =
nests on flat substrates, e.g., on the ground or in tree crevices, with no firm attachment points. Basal B = nests
anchored in vertical forks, e.g., among intertwined tree branches and primarily supported from the bottom. Lateral category =
nests attached to vertical or horizontal branches or stems that are interwoven in lateral nest walls and adherent
nests laterally attached to rocks or cliff walls. Rim suspended = nests attached at the rim to horizontal
forks or parallel sticks. Top suspended = pendant nests attached from the top of the nest (From: Leite et al. 2024).
Leite et al. (2024) used a dataset of 455 species to examine correlations between five methods of nest attachment and relative brain mass of passerine birds. Analysis revealed a relationship between method of nest attachment and relative brain mass, with species building Top suspended nests having close to a maximum relative brain size, and species with Rim suspended nests associated with brain size radiations and probably with the exploration of new ecological niches. These results provide evidence that the construction of nests with different attachment methods requires different levels of cognitive abilities.
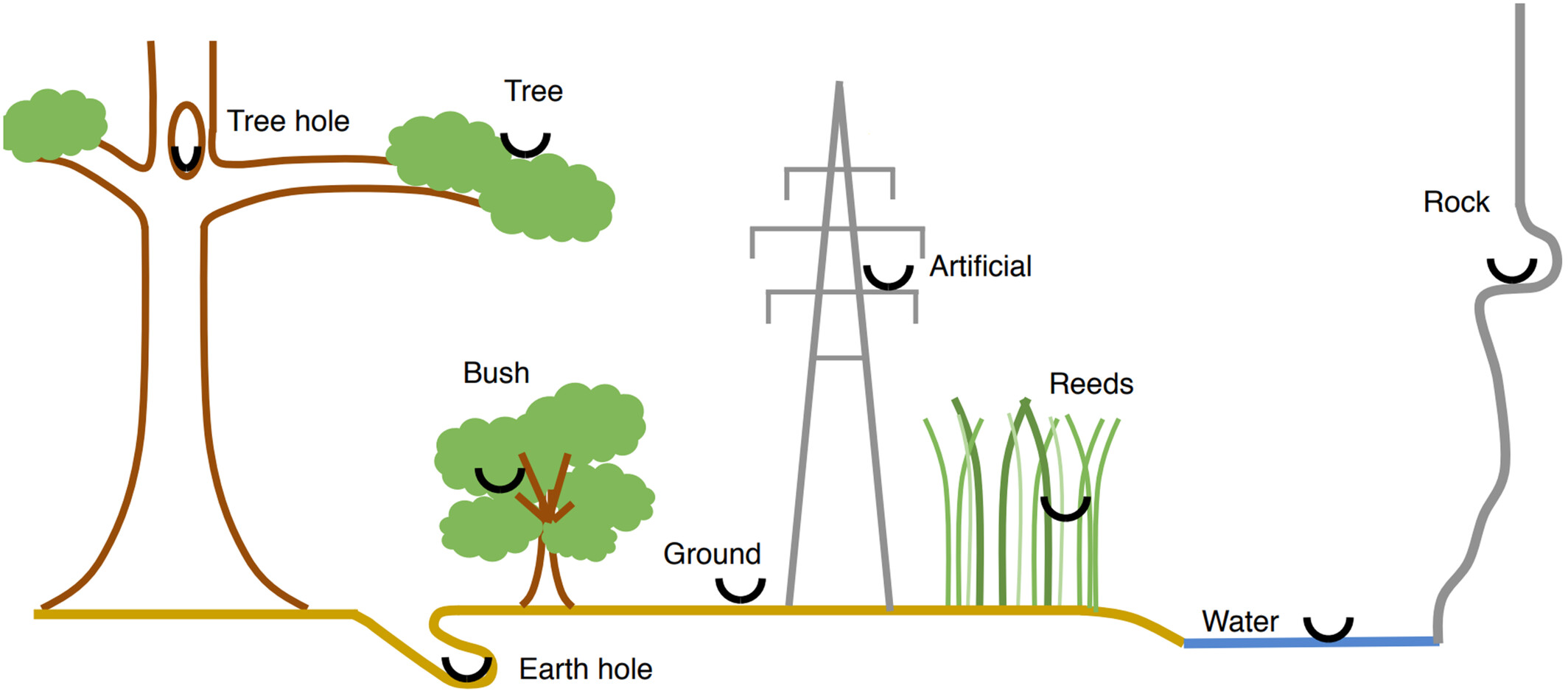
Major nest locations of birds, including the three sub-types of vegetation (tree, bush, and reeds).
Nest locations. Base on a global comparative database of nest building in birds, Sheard et al. (2023) found that species were more likely to nest in artificial locations in warmer and/or drier places with greater temperature seasonality and less precipitation seasonality. Species were more likely to nest in earth holes in cooler and/or drier places, and in tree holes at higher latitudes, in warmer places, in wetter places and/or in places with greater temperature seasonality. Species were more likely to nest on the ground in cooler locations, and in or near water at lower latitudes and/or in places with greater temperature seasonality. Species were more likely to nest on elevated rocks in cooler, drier and less seasonal (both temperature and precipitation) places. Species were more likely to nest in vegetation in warmer and/or rainier places with greater temperature seasonality and/or less precipitation seasonality.
Scrape nests are simple depressions in the ground (sometimes with a few stones added) or in the leaf litter. Such nests are used by some penguins, shorebirds, gulls, terns, nighthawks, vultures (e.g., Black Vulture nest below), and other species.

Source: http://www.ctbirding.org/ct_black_vulture_nest.htm
Spur-winged Plover nest
| Are wader nest scrapes adaptively designed to minimize clutch cooling rate? - Arboreal avian nests likely function partly to insulate clutches. However, reasons for the construction of nest scrapes are poorly understood. Working on Pectoral Sandpipers, Jane Reid tested the hypothesis that using a lined scrape reduces the rate of clutch heat loss & investigated whether scrapes are effectively designed to minimize heat loss rates. The use of both an unlined scrape and of lining material reduced the rate at which a test object lost heat. Constructing a lined scrape is therefore likely to serve to insulate a clutch. The rate of conductive heat loss from within a scrape increased with scrape depth and decreased with lining depth. Convective heat loss increased with wind speed in shallow scrapes but not in deep scrapes. Mean observed scrape depth approximately equaled that which minimized convective cooling while minimizing conductive heat loss. Further, on average, Pectoral Sandpipers used approximately the lining depth that minimized conductive heat loss while minimizing material useage. Scrape dimensions therefore approximated those likely to minimize overall heat loss, given the conflicting thermal pressures of the environment. Available lining materials differed in insulative quality both when wet and dry. Pectoral Sandpipers did not use lining materials in proportions that reflected local availability. Instead, relative use was correlated with a material's insulative quality when wet. Pectoral Sandpipers therefore used lining materials appropriate to minimizing heat loss given their damp breeding environment. - Jane M. Reid, University of Glasgow |

Pectoral Sandpipers nest on the arctic tundra, often near water. The nest is lined with grass, moss and lichens. Typical clutch size is 4 eggs. Photo source: http://picturethis.pnl.gov/picturet.nsf/f/uv?open&SMAA-497TFH |
Burrow nests are very effective at protecting eggs and young from predators & maintaining an appropriate microclimate for eggs & young. Some birds, like Bank Swallows and Belted Kingfishers (pictured below), usually construct their own burrows, while others, such as Burrowing Owls, may use burrows contructed by other species.

Belted Kingfisher digging nest burrow
Spacing of Bank Swallow tunnel entrances
(Ghent 2001) -- Tunnel entrance distributions at six Bank Swallow sand-pit
colonies showed consistently nonrandom, too-regular patterns, supporting
the hypothesis that the distance between tunnel entrances is determined
by territorial disputes at the tunnel  mouths.
Average nearest neighbor distances, and numbers of burrows per unit area
of pit face, were both consistently greater than their random expectations,
a paradox that is explained algebraically. Evidence of tunnel coalescence
and communal nesting was found in a completely evacuated colony of 30 tunnels.
Bank Swallow colonial behaviors may have evolved to maximize populations
on small exposed bank faces along streams and rivers, and that they are
still behaving as if only small fractions of large sand pit banks are available.
On small areas, regular spacings help to avoid too close tunneling and
concomitant bank collapse. It is argued that a tolerance for communal nesting,
once coalescence has occurred, involves less tunneling than the forced
evacuations of entirely new tunnels by ousted pairs. mouths.
Average nearest neighbor distances, and numbers of burrows per unit area
of pit face, were both consistently greater than their random expectations,
a paradox that is explained algebraically. Evidence of tunnel coalescence
and communal nesting was found in a completely evacuated colony of 30 tunnels.
Bank Swallow colonial behaviors may have evolved to maximize populations
on small exposed bank faces along streams and rivers, and that they are
still behaving as if only small fractions of large sand pit banks are available.
On small areas, regular spacings help to avoid too close tunneling and
concomitant bank collapse. It is argued that a tolerance for communal nesting,
once coalescence has occurred, involves less tunneling than the forced
evacuations of entirely new tunnels by ousted pairs.
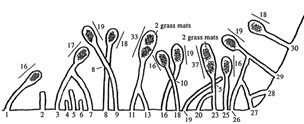
Excavation of 30 tunnels. Three of the five coalescing groups led to double-length nest chambers with two grass-mat nests. Vertical separations (e.g., 8 cm between tunnels 8 and 9) indicate where neighboring tunnels crossed w/out coalescing. |
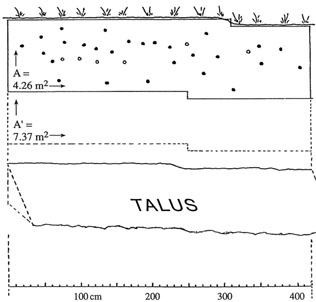
Area A = 4.26 sq. m determined by visual framing of a too regularly spaced burrows of a Bank Swallow colony. A' = 7.37 sq. m is the area on which 27 (solid circle) burrows leading to a nest would have to be distributed to equate their observed average nearest neighbor distance r to its random expectation E[r]. Open circles represent 6 tunnels not leading to a nest (not included in the calculations) |
Mites and birds -- At least 2500 species of mites from 40 families are closely associated with birds, occupying all conceivable habitats in the nests and on the bodies of their hosts. No avian taxon is free from a mite associate because even those that lack feather mites, such as penguins, are attacked by ticks. Bird mites can be divided into those that dwell primarily in, or near, the nest and those that reside mainly on the body of the host. The best studied nest-dwelling mites are blood feeders from the genera
Dermanyssus  and Ornithonyssus (shown here is a micrograph of a female Ornithonyssus bursa, a common nest parasite of passerines. Scale bar = 100 μm. Micrograph from Dave Walter, University of Queensland). Depending on the species involved, adults of these blood feeders live in the nest or on the hosts, but nymphal stages are primarily nestbound and only visit hosts when they need to feed. These mites have short generation times and can rapidly build-up huge populations. For example, half a million northern fowl mites have been extracted from a single nest. Ticks can also be temporary nest parasites. Soft ticks visit the host at night, feed for a few minutes and then retreat to a refuge in, or near, the nest. Hard ticks tend not to be so nestbound and will attack birds as they brush against vegetation during foraging or resting. However, not all nest mites are parasitic. Relatives of human-associated ‘dust mites’ feed on the dermal detritus that sifts down into the nest material. Other nest dwelling mites prey on blood-sucking mites, and thus might act as mutualists.
and Ornithonyssus (shown here is a micrograph of a female Ornithonyssus bursa, a common nest parasite of passerines. Scale bar = 100 μm. Micrograph from Dave Walter, University of Queensland). Depending on the species involved, adults of these blood feeders live in the nest or on the hosts, but nymphal stages are primarily nestbound and only visit hosts when they need to feed. These mites have short generation times and can rapidly build-up huge populations. For example, half a million northern fowl mites have been extracted from a single nest. Ticks can also be temporary nest parasites. Soft ticks visit the host at night, feed for a few minutes and then retreat to a refuge in, or near, the nest. Hard ticks tend not to be so nestbound and will attack birds as they brush against vegetation during foraging or resting. However, not all nest mites are parasitic. Relatives of human-associated ‘dust mites’ feed on the dermal detritus that sifts down into the nest material. Other nest dwelling mites prey on blood-sucking mites, and thus might act as mutualists.
Blood-feeding nest mites can reduce the reproductive success of their hosts by slowing development or even killing chicks. For example, recent experimental work has shown that high densities of nest mites are associated with low hematocrit and small body size in Pied Flycatchers (Ficedula hypoleuca), and low hatching success and postfledging survival in Rock Pigeons (Columba livia) and Barn Swallows (Hirundo rustica). By contrast, however, Darolová et al. (1997) observed a positive association between the percentage of Penduline Tit (Remiz pendulinus) nestlings that survived to fledging and the number of hematophagous mites in the nest. The authors suggest that nestling health determines mite load rather than vice versa. Other researchers have found no relationship between nest–parasite density and nesting success. Merino and Potti (1996) suggested that variable effects of nest parasites are, in part, a result of stochastic climatic factors, such as temperature and rainfall. More work is required to establish why the effects of nest mites on host reproductive success are so variable across studies. -- Proctor and Owens (2000).
Cavity nests (e.g., in trees or cacti) are used by numerous passerines, woodpeckers, owls, parrots, and some waterfowl. Some birds, such as woodpeckers (like the Gila Woodpecker below), construct their own cavity nests and are referred to as primary cavity nesters. Species that use natural cavities or cavities constructed by primary cavity nesters are called secondary cavity nesters.
Black Woodpecker nest cavity
Approximately 18.1% (1,878 species) of all bird species globally nest in tree cavities, and these species are classified into three groups: primary excavators (species that nearly always excavates their own nesting cavities; 19%), facultative excavators (known to regularly use existing cavities, but also able to excavate new cavities or enlarge existing cavities; 7%), and non-excavators (species that only use existing decay- formed or excavated-tree cavities, and either do not or only rarely excavate or renovate cavities; 73%). Cavity-nesting species are found predominantly in 10 avian orders, including Anseriformes, Falconiformes, Psittaciformes, Strigiformes, Caprimulgiformes, Apodiformes, Trogoniformes, Coraciiformes, Piciformes and Passeriformes. The highest richness of tree-cavity nesters is found in the Neotropical (678 species) and Oriental (453 species) regions. Woodpeckers are considered keystone "ecosystem engineers" because their excavated cavities are used by at least 338 species of non-excavators. At least 42% of tree-cavity nesters use excavated cavities, highlighting the importance of woodpeckers and other excavators in maintaining cavity availability.
Thirteen percent (249 species) of tree-cavity nesters are globally threatened (vulnerable, endangered, or critically endangered), with non-excavators more likely to be threatened (15%) than primary (6%) and facultative excavators (6%). Australasia has the highest proportion of threatened species (17%), followed by the Oriental region (13%). Tree-cavity-nesting birds face threats from logging, forest clearing, agriculture, and urbanization, all of which reduce the availability of suitable nesting sites. Maintaining a supply of tree cavities is critical for the survival of tree-cavity-nesting birds. This will require preserving old trees and supporting decay processes. Conservation measures such as adding nest boxes can help protect these species.
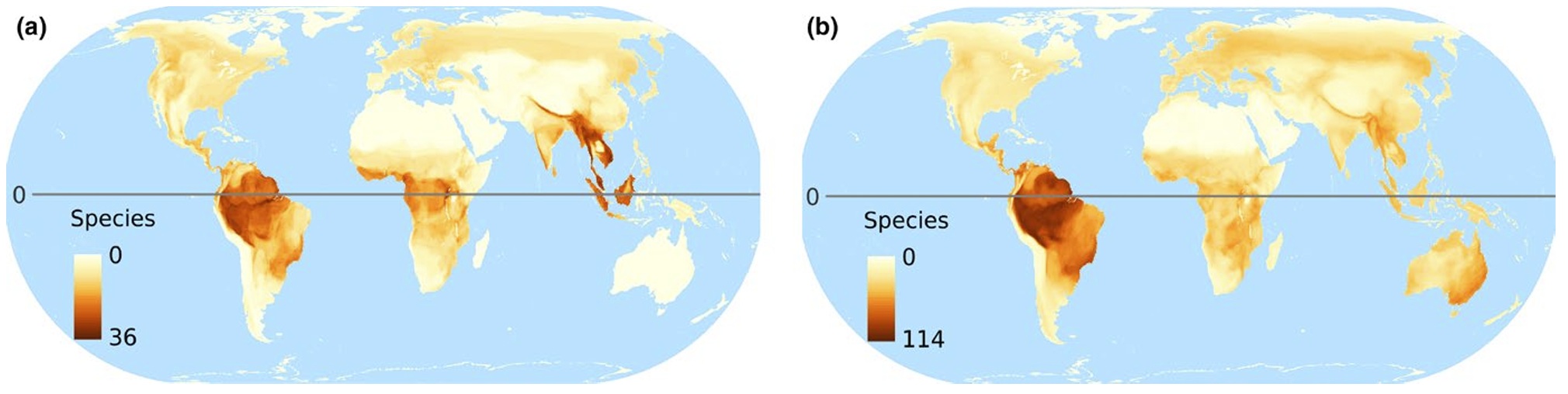
Global maps of richness of all tree-cavity-nesting-bird species classified as either (a) primary or facultative excavators or (b) non-excavators. Patterns of the
global distribution and species richness of tree-cavity-nesting species are largely similar to those of the family Picidae (woodpeckers), and species richness of birds in
general, with the tropics being especially species rich. Given the requirement for trees as nest substrates, the distribution of cavity-nesting species of birds
roughly follows the same pattern as that of the worlds forests (From: van der Hoek et al. 2017).
Platform nests are relatively flat nests that may be located on the ground, in a tree, or on the tops of rooted vegetation or or debris in shallow water (like the Western Grebe nest below).

Black Stork nest
Uncovering the hidden world of a secretive seabird (Marbled Murrelet) by Jim Rivers
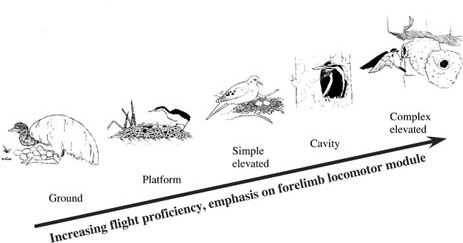
Bird nests vary from a simple accumulation of materials on the ground to elaborate refuges in or on secluded & elevated substrates. Dial (2003) observed that nest construction and placement are correlated with other features such as flight ability. For example, basal avian taxa (ratites & many Galliformes) create a simple depression in the ground to harbor their incubating eggs, like those of nonavian dinosaurs. The progression of nest complexity moves from cryptic ground nests of some galliforms to simple elevated nests (e.g., Columbiformes, Cuculiformes, & Ciconiiformes). Taxa that construct elevated nests in a bush or tree or on a cliff or rock ledge tend to be better fliers than simple ground nesters.Young raised in elevated & cavity nests, including primary (Psittaciformes, Piciformes, & Coraciformes) and secondary (many Passeriformes) cavity nesters, have a robust forelimb flight apparatus, and less hindlimb mass, which is consistent with increasing flight capacity. As nest placement (e.g. invisibility, inaccessibility), construction (e.g. impregnability, camouflage), and attendance (e.g. feeding, protection, incubation) increase in complexity, a concomitant enhancement of flight styles is observed, including maneuverability and acceleration. The most complex nests are associated with some Passeriformes, particularly swallows, oropendolas, and weaver finches. Weaver finches (Ploceidae, Passeridae) and oropendolas (Icteridae) build intricately woven chambered, pendant nests hung from the resilient thin branches of bushes and trees in predator-rich environments. Perhaps the most predator-proof nests are those of swallows (Hirundinidae) and swifts (Apodidae) that often construct mud encasements secured to the most remote overhanging feature within their habitat (e.g. cliffs and human-made structures) (Dial 2003). |
Cupped nests are, of course, cup-shaped. Such nests may be constructed of various materials and in a variety of locations. Pettingill (1985) categorized cup nests as follows:
statant cupped nests - nests located in the crotches and branches of trees and shrubs and supported mainly from below. Many passerines and hummingbirds build such nests.
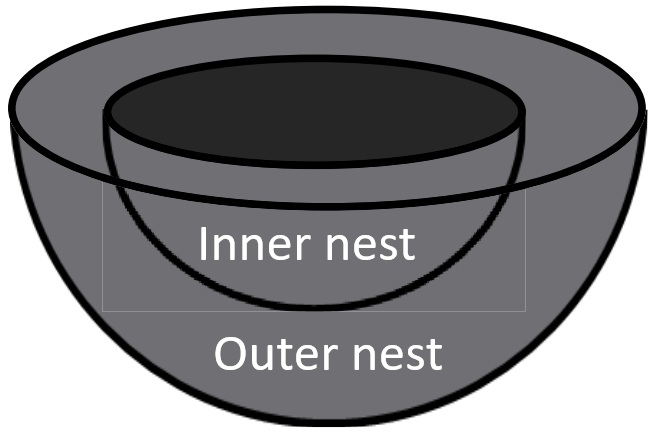
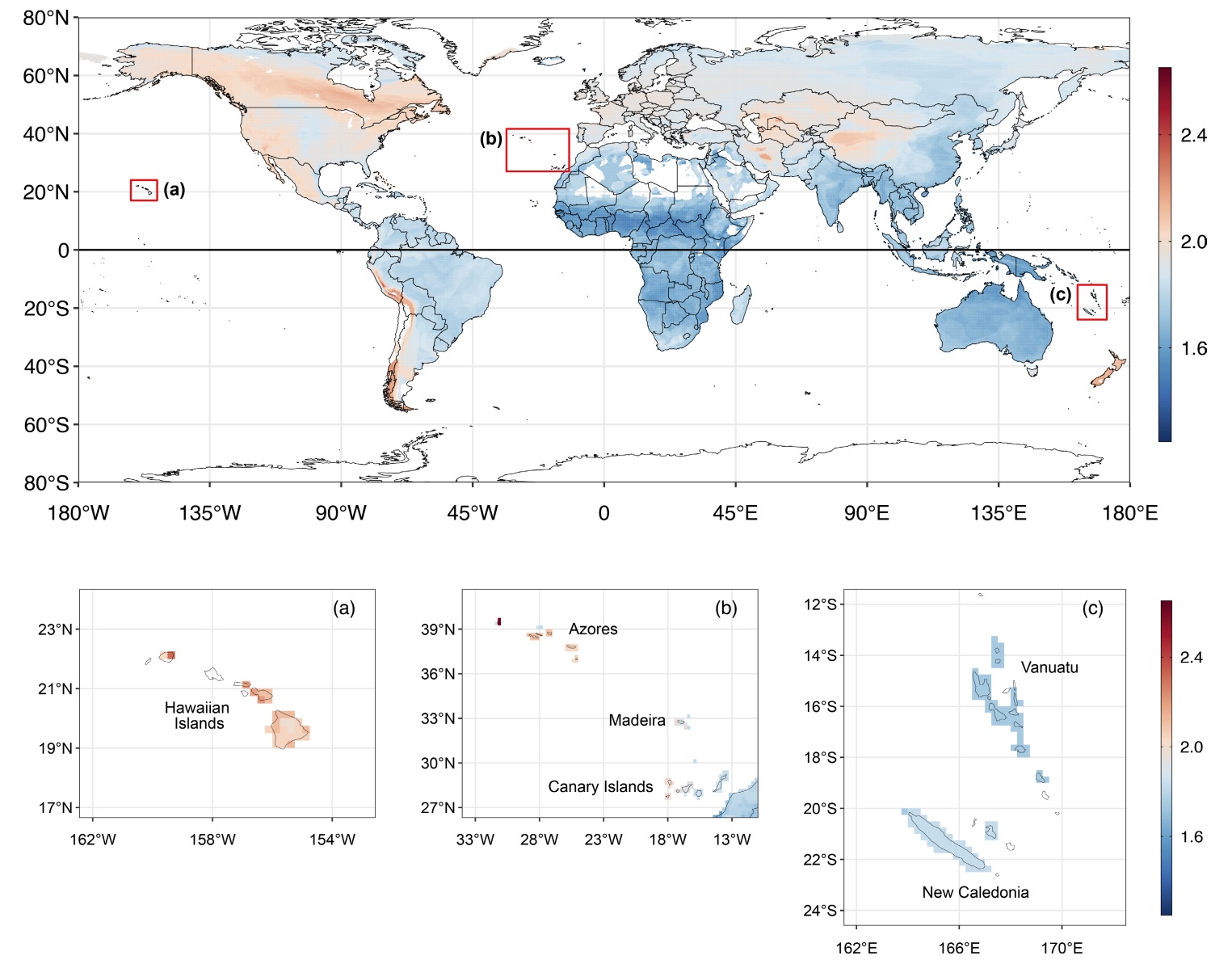
(Top) Diagram showing how outer and inner nests were defined. (Bottom) Geographical distribution of the mean outer nest volume divided
by body mass per 0.25° grid cell (n = 827 species). Grid cells
with fewer than four species were removed from the visualization and appear
blank. Subpanels (ac) illustrate variation in nest size in different island groups, with all grid cells containing at least one species displayed. The
same color scale has been retained for all panels to ensure comparability; values along the y- and x-axes correspond to latitude and longitude, respectively.
Variation in size of cup nests among passerine birds. Vanadzina et al. (2023) examined global variation in nest size and found that, after accounting for phylogeny, both inner and outer components of nest structure were strongly correlated with the body size of the species (i.e., larger birds have larger nests), but additonal variation is explained by two different sets of factors. First, the outer volume of nests is greater for birds in colder climates (providing greater insulation to help keep eggs and nestlings warm), species on islands, and species that nest on cliffs or rocks. Second, inner cup volume is only associated with average clutch size, increasing with the number of chicks raised in the nests. The larger nests of island birds and species that nest on cliffs or rocks (and, therefore, likely less accessible to predators) support the hypothesis that reduced levels of predation can lead to increases in nest size. Additional analysis revealed that nests get smaller with increasing distance above the ground, possibly due to physical constraints on the size of above-ground nests (e.g., branches in trees get smaller in diameter with increasing distance above ground).

Source: http://www.sandiegozoo.org/wildideas/animal/hummingbird.html
Anna's Hummingbird building nest

Nesting sites and nests of the Blue Manakin (Chiroxiphia caudata) in the Brazilian Atlantic Forest.
(a) Example of the nesting habitat of Blue Manakins. (b) Blue Manakin nest with a tail of decaying materials.
Nests with tails. Blue Manakins, small birds found in southeastern Brazil, build nests that look fairly messy, with long dangling structures (nest tails) woven from strands of moss, fibers and other debris. Martins et al. (2026) found that these nest tails help protect eggs and nestlings from predators. The investigators collected 50 manakin nests over two breeding seasons (once they had been abandoned) and placed them in similar locations. They left tails on half of the nests and removed them from the other half. They also placed realistic-looking eggs made of plasticine in each, and used infrared cameras to monitor the nests. The cameras revealed that nests without tails were more likely to be predated; only one nest with a tail was predated, whereas 11 nests where the tail had been removed were predated. All of the recorded predators were visually oriented, confirming the antipredatory adaptive function of avian nest tails, and corroborating the disruptive camouflage hypothesis (From: Arnold at Phys.org and Martins et al. 2026).
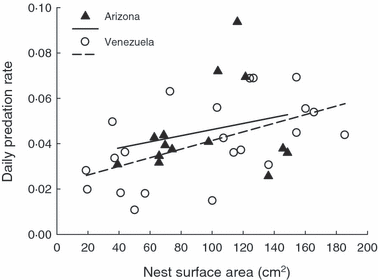
Nest surface area and nest predation rates for 36 altricial bird species
studied in tropical Venezuela
and temperate Arizona. Nest predation
increased with nest surface area at both
field sites.
Nest size and nest predation -- Latitudinal variation in clutch sizes of birds is a well described, but poorly understood pattern. Many hypotheses have been proposed, but few have been experimentally tested, and none have been universally accepted by researchers. The nest size hypothesis posits that higher nest predation in the tropics favours selection for smaller nests and thereby constrains clutch size by shrinking available space for eggs and/or nestlings in the nest. Biancucci and Martin (2010) tested this hypothesis with an experiment in a tropical forest and a comparative study between temperate and tropical field sites and, specifically, tested if: (i) predation rates increased with nest size, (ii) tropical birds had smaller nests controlled for body size, and (iii) clutch size was explained by nest size controlled for body size. Experimental swapping of nests of different sizes showed that nest predation increased with nest size in the tropical site. Moreover, nest predation rates were higher in species with larger nests at both sites. However, nest size, corrected for body mass and phylogeny, did not differ between sites and was not related to clutch size between sites. Hence, nest predation can exert selection on nest size as predicted by the hypothesis. Nest size increased with adult body mass, such that adult size might indirectly influence reproductive success through effects on nest size and nest predation risk. Ultimately, however, selection from nest predation on nest size does not explain the smaller clutch sizes typical of the tropics.
suspended cupped nests - nests not supported from below but from the rims, sides, or both:
pensile - nests suspended from the rims and sides; rather stiff, e.g., those of kinglets and vireos (like the Black-capped Vireo pictured below)

pendulous - nests suspended from the rims and sides; rather flexible and deep, like those of orioles

Oropendola nests
The pendent nests of some weaverbird and icterid species are among the most complex structures built by birds. The precarious attachments and extended entrance tunnels characteristic of these nests are thought to act as structural defences against invasion by nest predators, particularly tree-climbing snakes, but this hypothesis has yet to be systematically tested. Street et al. (2022) used phylogenetic comparative methods to investigate the relationship between nest structure and developmental period length, a proxy for offspring mortality, in weaverbirds (Ploceidae) and icterids (Icteridae), two bird families with highly elaborate pendent nests. Analysis revealed that more elaborate nests, particularly those with entrance tunnels, are associated with longer developmental periods, and suggest that elaborate nest structures can function as structural defenses, resulting in lower offspring mortality and slower development. More generally, constructing complex, protective nests may buffer against environmental hazards and reduce extrinsic mortality.
Baya Weavers (Ploceus phillipinus) nest in colonies of up to 20-30 pairs, usually in trees near freshwater and open ground.
Their nests hang from a branch and look like an upside-down flask. A long tube leads to a side entrance, making
it difficult even for snakes to enter the nest. Nests are made entirely out of strips of grass that the birds collect by cutting
a notch
in some
tall grass, then stripping off a 30-60 cm long piece.
A newly-made nest is green with fresh grass and turns
brown as the grass dries.
A bird may make up to 500 trips to complete a nest.
Hummingbird nest
adherent nests - cupped nests whose sides are attached by an adhesive substance (e.g., mud and/or saliva) to a vertical surface, like those of swifts and some swallows. Swallows (Hirundinidae), phoebes (Sayornis), and other mud nesters use a unique building material, a mixture of mud and their own saliva. During construction, mud nesters repeatedly pile a beakful of wet mud on the nest, and liquid bridges are formed in the nest due to evaporation. The capillary forces of liquid bridges provide temperary cohesion, but dehydrated saliva provides permanent cohesion after complete evaporation. Although building a nest may take several weeks, a transition from wet to dry structures can occur within a few hours. Mud itself cannot confer sufficient cohesion and adhesion in mud nests. The ability of mud nests to bear tensile loads originates from the gluing agent in the bird's saliva, which permeates into granules as a liquid and binds them as a solid after solvent evaporation. The gluing agent is called mucin, a family of large glycoproteins that are ubiquitous in animal organs and form a mucus gel with versatile functionality. However, because saliva primarily serves as a physiological fluid for predigestion, it is unlikely to have highly concentrated mucins. As a result, there is a size limit for mud-nesting birds that use saliva as glue, with the mass of most species with adherent nests ranging from about 1822 grams (From: Jung et al. 2021).
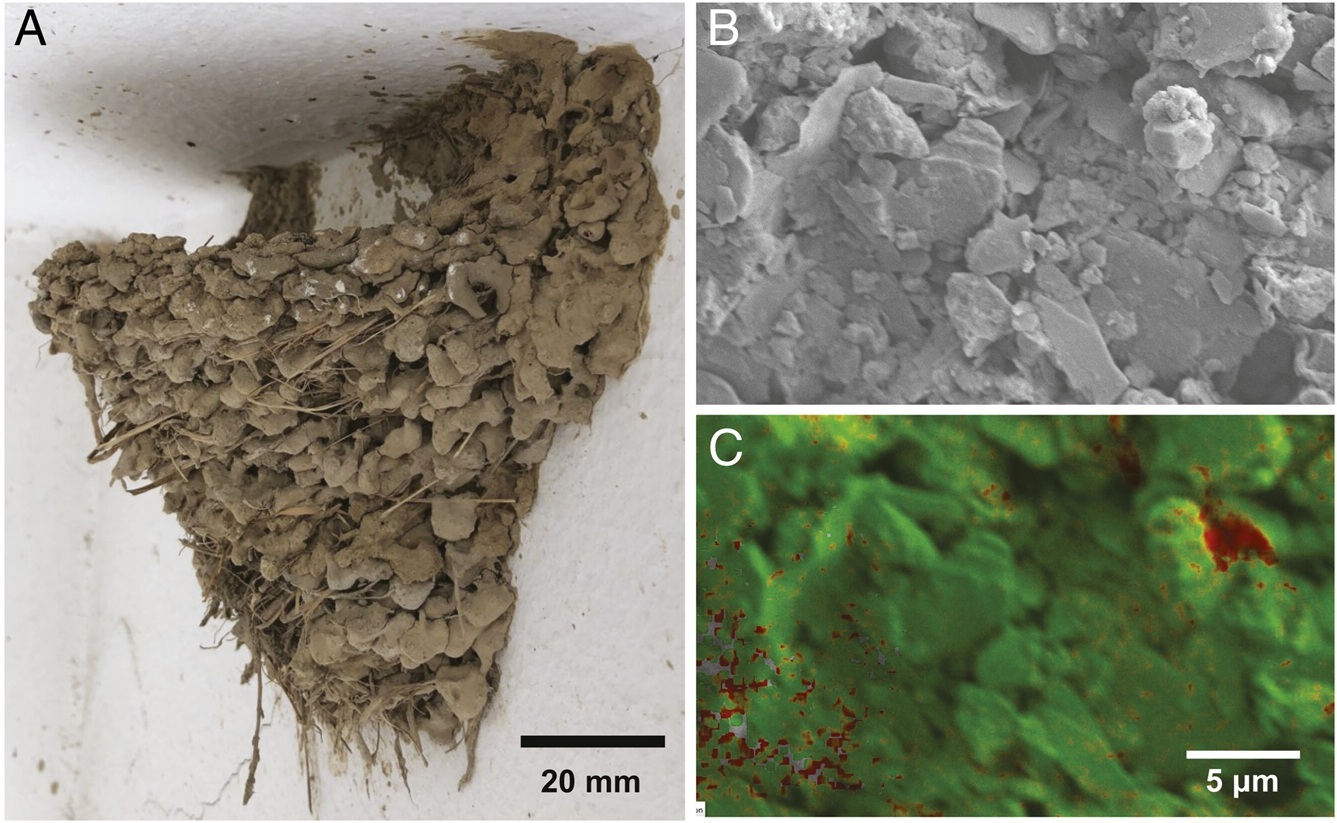
(A) A Barn Swallow nest. (B) Scanning Electron Microscope (SEM) image of the nest surface. (C) Chemical composition analysis of
the surface shown in B. The red area indicates a region containing mostly carbon atoms, which may originate from bird
saliva. The green area indicates a region containing mostly the silicon atoms of clay particles (From: Jung et al. 2021).

Chimney Swifts use saliva as glue to help construct and support their nests.

Saliva 'solidified' and forming arch over Chimney Swift nest to help hold it against the wall.
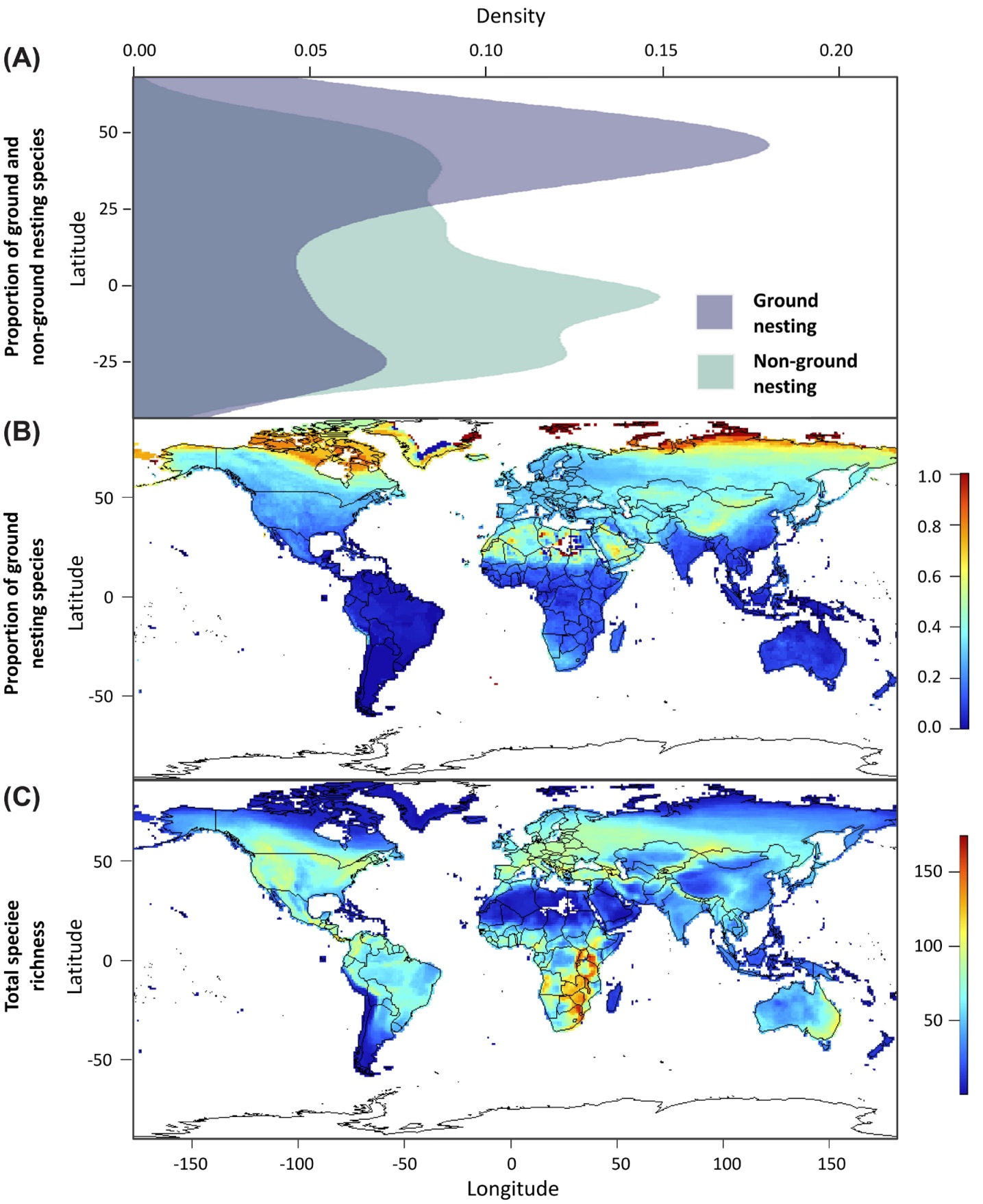
Biogeographical variation in the occurrence of ground-nesting passerines. (A) Latitudinal gradient in the proportion
of ground and non-ground nesting species, (B) Geographical distribution of the proportion of ground-nesting species,
and (C) Geographical distribution of total species richness (the number of ground and non-ground nesting passerine
species in the dataset; n = 1633) (From: Minias and Janiszewski 2023).
Nest location is one of the key components of avian reproduction and sharp transitions between different nest location strategies may have profound ecological and evolutionary consequences. Minias and Janiszewski (2023) used a phylogenetically-informed comparative framework to reconstruct the evolution of ground-nesting behavior in passerine birds, quantify its biogeographical and habitat variation, and test for its associations with life history traits. With a dataset of over 1600 species distributed worldwide and representing about 90% of all passerine families, analyses revealed a scattered phylogenetic distribution and moderate phylogenetic signal in the occurrence of ground nesting in passerines. Non-ground nesting was identified as a likely ancestral state at the root of passerine phylogeny. Occurrence of ground nesting was clearly associated with habitat variation, with open habitats generally having a greater proportion of ground-nesting species than non-open habitats (forests). Among the major habitat categories a greater proportion of ground nesters were found in boreal and temperate than subtropical and tropical zones. This reflected a general latitudinal gradient in the occurrence of ground nesting, with a higher proportion of ground nesting species at higher latitudes (especially in the Northern Hemisphere). Ground nesting was also associated with fast life histories, with ground nesters having larger clutches, shorter incubation periods, and faster chick development rates than non-ground nesters. This variation was possibly driven by higher predation rates on nestlings and adult birds in ground-nesting species.
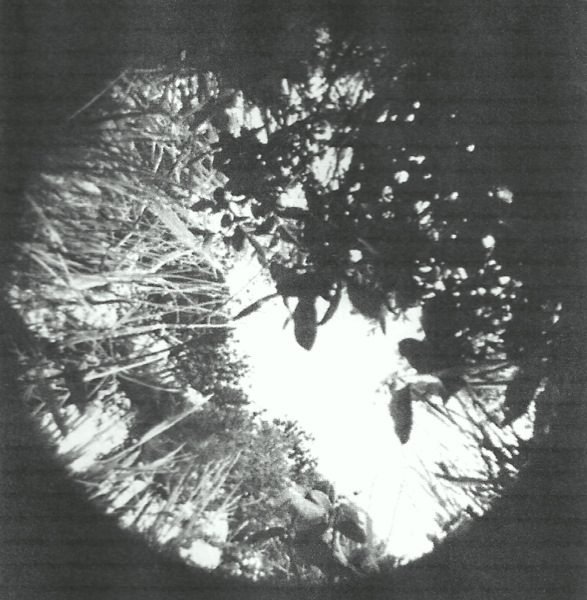
Hemispherical photo of the vegetative canopy over a White-crowned Sparrow nest. Nests often have less vegetative cover in the
eastern sky, allowing more solar radiation to strike nests in the morning when ambient temperatures are cooler (From: Walsberg and King 1978).
Birds nesting on the ground
Domed nests - the upper portion is enclosed by a constructed roof, including globular structures and pendulous nests with side entrances. Some domed nests have long entrance tunnels.
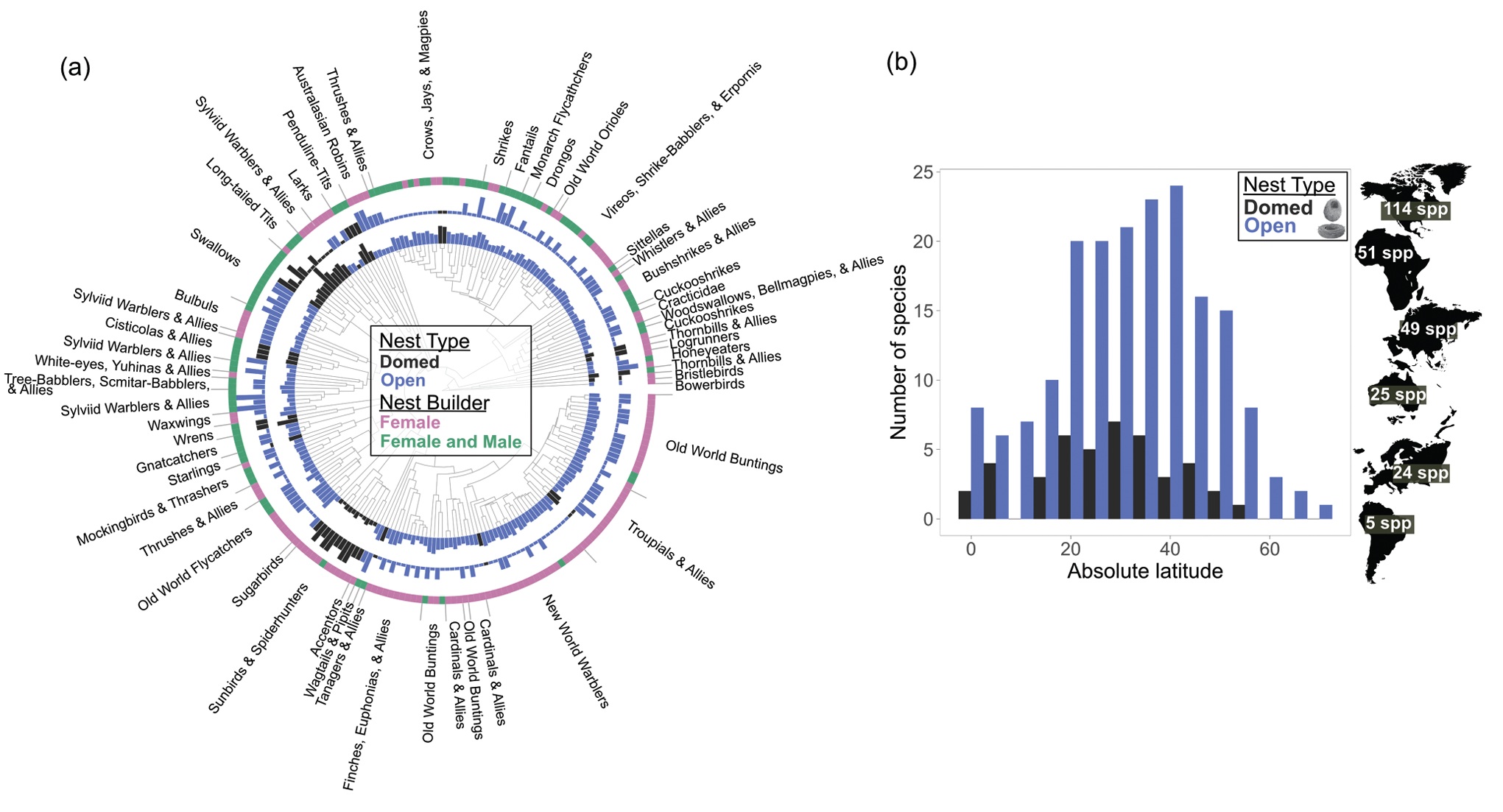

Rock cavity nests and eggs of: (A) Geositta rufipennis (Rufous-banded Miner), (B) Cinclodes oustaleti (Gray-flanked Cinclodes),
(C) Muscisaxicola maclovianus (Dark-faced Ground-tyrant), (D) Muscisaxicola albilora (White-browed Ground-tyrant),
and (E) Pygochelidon cyanoleuca (Blue-and-White Swallow) (From: Altamirano et al. 2023).
Organisms living and breeding in alpine habitats must cope with severe environmental challenges such as temperature extremes, storms, resource limitations and, sometimes, hypoxia, resulting in short windows and reduced opportunities to breed. Despite this, ~12% of bird species breed in alpine habitats and some of these species nest in rock cavities. Factors that may contribute to the rock cavity-nesting behavior of alpine breeding birds include: (1) alpine habitats are strongly seasonal, with deep snow cover during winter, and rock substrates are likely the first to be exposed in spring as solar radiation warms the rock under the snow, contributing to faster snowmelt. Thus, rock cavities are likely available earlier than other potential nest sites and, therefore, more likely to be selected as a nesting site, especially given the short breeding seasons at high elevations, (2) rock cavity nests may provide better microclimates for raising young, with higher levels of humidity, more consistent temperatures, and reducing UV-B exposure compared to exposed alpine nests, and (3) many potential nest predators cannot access subterranean nests, potentially driving selection for rock cavity nests (From: Altamirano et al. 2023).
Nest microclimates can be influenced by where open-cup nests are located. Shrub-nesting White-crowned Sparrows (Zonotrichia leucophrys) appear to consider the vegetation canopy when determining where to build nests. Walsberg and King (1978) examined the distribution of vegetation located above nests at a high-altitude site (1890 m) in Oregon and found that most nests were positioned so that they were more exposed to sunlight (75% more on average) in the morning when ambient temperatures were lower than the afternoon when temperatures were higher. This difference in exposure to solar radiation likely helped warm nests and their contents during cool mornings and reduced the likelihood of overheating during warmer afternoons.
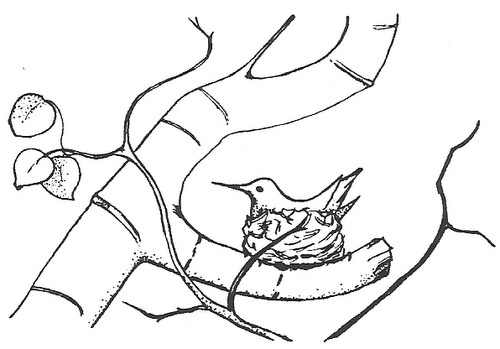
Typical nest location for a female Broad-tailed Hummingbird with a branch above the nest serving as a ‘roof’ (From: Calder 1973).
Nest microclimates may, in some cases, be as important for adult birds as for embryos and nestlings. Some hummingbirds nest at high latitude and high altitude locations where the combination of their high metabolic rates and sometimes cool ambient temperatures (particularly at night) may make balancing energy budgets difficult. Incubating and brooding female hummingbirds can, of course, conserve energy at night by entering torpor. However, even after allowing their body temperatures to drop, hummingbirds may still lose heat via radiation when ambient temperatures are lower than their body temperature. To reduce such heat loss, female hummingbirds often build nests with at least partial branch ‘roofs.’ Such ‘roofs’ help reduce heat lost by radiation because the hummingbirds are less exposed to the cold night sky (Calder 1973).
Experimental evidence that nest orientation influences microclimate in a temperate grassland. Birds exhibit an assortment of behavioral strategies to cope with variable environmental conditions during reproduction, including altering nest construction behaviors. Among species that build enclosed domed nests, the microclimate within nests is influenced not only by its structure and the surrounding vegetatio, but also by the orientation of the nest opening. Many grassland-dependent birds build dome-shaped nests with clear directionality of openings. Corimanya et al. (2024) studied two species in northeastern Kansas, United States, that typically orient their nests east to northeast in this region. However, in a drought year, both Grasshopper Sparrows (Ammodramus savannarum) and Eastern Meadowlarks (Sturnella magna) shifted orientations of their nests southward toward prevailing winds. Corimanya et al. hypothesized that this shift reduced the deleterious effects of heat stress on adults and developing young by reducing morning solar radiation and increasing cooling due to the prevailing southerly winds. To test this hypothesis, temperature, humidity, and wind speed were measured at pairs of unoccupied, field-collected sparrow and meadowlark nests, experimentally placed to face south or east (control) in a non-drought year. Nest orientation affected the daily microclimate patterns, with south-facing nests warming later in the day relative to east-facing nests. Temperature differences depended on humidity, with south-facing nests being relatively cooler under more humid conditions. These results provide experimental evidence for the benefits of plasticity in nest construction under challenging thermoregulatory conditions and show how ground-nesting birds may reduce thermoregulatory demands during incubation under climate variation.
| Tits weave fragrant nests. - Birds weave aromatic plants into their nests, apparently to keep their home clean and bug-free for raising chicks. Blue Tits on the fragrant Mediterranean island of Corsica can even smell when it's time to refresh fading fragments, ecologists have shown (Petit et al. 2002). Female blue tits gather lavender, yarrow, curry, mint and other scented plants for their nests shortly after laying eggs, and continue to do so until the chicks leave home. "They are real botanists and do a great job exploiting their environment to protect their chicks," says Marcel Lambrechts of the Centre for Functional Ecology and Evolution in Montpellier, France. The birds make a pot-pourri of 10 aromatic plants from the 250 species in their habitat. Many of the chemicals in these plants ward off bacteria, viruses, parasites, fungi and insects. Lambrechts's team removed the aromatic plants from 64 nests and then placed a hidden box containing lavender and yarrow underneath half of the nests. In the first 24 hours, only the birds with empty boxes replenished their herb supply. After 48 hours, the other half of the birds began to restock too, as the scent from the hidden herbs waned. "This field test directly shows that birds are attending to odour cues," says Larry Clark, who studies similar behaviour in European Starlings at the National Wildlife Research Center in Fort Collins, Colorado. The Blue Tits select for chemical diversity as well as high concentrations of chemicals, he points out, underlining the importance of olfaction in avian behavior. -- Kendall Powell, Nature Science Update | 
|

Denmark/photo59174.htm |
Birds Use Herbs To Protect Their Nests Researchers from Ohio Wesleyan University suggest that some birds may select nesting material with antimicrobial agents to protect their young from harmful bacteria. They presented their findings at the 2004 meeting of the American Society for Microbiology. "If the fresh herbs and plant materials that parent birds bring into the nest have a sufficient concentration of antimicrobial compounds, they could protect the nestlings from harmful bacteria," says researcher Jann Ichida. To find out if plants brought into the nest might prevent disease, Ichida and colleagues tested 12 different volatile plant materials against feather-degrading bacteria. Results showed that several types of plant materials and extracts including usnic acid, ascorbic acid, yarrow, and two oak species inhibited the growth of a number of harmful bacteria. "If the fresh herbs have a sufficient concentration of these chemicals, they could protect the nestlings from harmful bacteria," says Ichida. "By practicing medical botany, parent birds exercise effective home nest security and protect their offspring from select biodegrading microbes that affect the health of their young." - Science Daily |
| Using scat to reduce nest predation -- For many animals, nest construction is a prerequisite for successful breeding. The choice of nesting materials is an important component of nest construction, because material properties can influence nest design and, potentially, reproductive success. Common Waxbills are small African finches that select carnivore scat as a material to include in, on, and around their nests. Schuetz (2005) investigated the hypothesis that scat functions to reduce predation risk by documenting its use in a wild population of Common Waxbills and by conducting an artificial nest experiment. Among natural nests, scat was present in every nest that hatched young, and parents continued to add scat to nests throughout the nestling period. Among artificial nests, those that received experimental additions of carnivore scat survived at a significantly higher rate than did untreated nests, suggesting that scat functions to reduce predation risk. The mechanism by which nests are protected remains unclear, although it is likely that scat acts as an olfactory deterrent and/or camouflage. Researchers have long focused on the implications of nest site characteristics for avian life-history evolution. Results of the present study suggest that nest materials, similar to nest sites, may influence life histories of nest-building animals by affecting predation risk. |
|
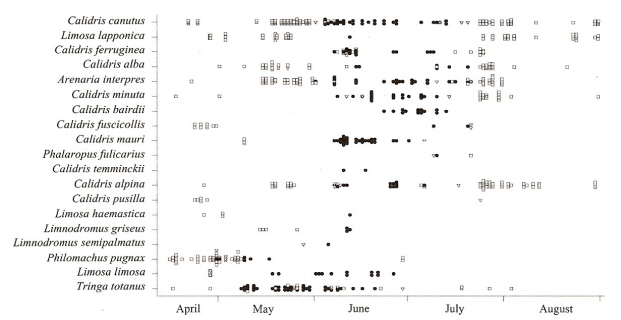
Seasonal changes in chemical composition of preen waxes of adults in 19 species of sandpipers. Species are listed from
top to bottom
on the basis of median latitude of their breeding range, with the northernmost species first. Squares = monoesters,
triangles = mixture of mono- and diesters, filled circles = diesters (From: Reneerken et al. 2002).
Nest construction
Although the specific methods certainly vary across species, the general process for building a nest typically follows five key stages: site selection, material gathering, foundation laying, wall construction, and lining.
1. Site Selection
The process begins with finding a location that balances safety from predators with proximity to food and water. Birds evaluate:
Protection: Hidden spots in dense foliage or high branches.
Stability: For tree- and shrub-nesting species, strong forks in branches or horizontal tree limbs to prevent collapse.
Microclimate: Shelter from wind and rain.
2. Gathering materials
Birds are resourceful engineers, collecting a variety of materials for nest constructions:
Structural: Twigs, coarse grass, bark, and roots for the frame.
Adhesives: Some species of birds use mud, saliva, or spider silk (e.g., hummingbirds) to bind materials together (but most species do not).
Insulation: e.g., moss, feathers, and fur may be incorporated into nests to keep eggs and young warm.
3. Laying the foundation
Once a site is selected, birds may begin the base.
Initial framing: Larger, sturdier twigs, (& for larger species) branches, or mud form a platform or anchor point.
Anchoring: Weaver birds, for example, knot strips of grass to a branch to create a secure ring.
Mechanical bonding: Most species of birds interweave twigs or branches relying on tension rather than glue.
4. Wall construction
This stage involves building the "cup" (for cup-nesting species).
Body molding: Species like American Robins use their own chests to press and shape mud and grass into a neat bowl, rotating as they work.
Weaving: Using only their beaks and feet, some species of birds pull material over and under to create a structure similar to a woven rug.
Reinforcement: Finer grasses may be woven into the rim to ensure the structure holds its shape.
5. Lining and final adjustments
The interior is finished with soft materials to protect the eggs and chicks.
Comfort: Many species use a lining of mammal fur, down feathers, or fine grass to provide cushioning.
Parasite deterrence: Some birds incorporate specific aromatic plants that may repel insects.
Repair: Birds may continue to reinforce or adjust the nest throughout the incubation and nestling periods.
Few studies have focused on the question of whether nest building by birds in an innate behavior or is, at least to some extent, learned. Two lines of evidence suggest that nest building has a large instinctive component: (1) the structure and composition of nests exhibit little intraspecific variation, and (2) birds raised in isolation tend to build species-specific nests. However, particularly for birds that build more complex nests, evidence indicates that birds learn by experience and, as a result, the quality of nests built by older birds may exceed that of nests built by younger, less experienced birds. For example, the first nests of young male Village Weavers (Ploceus cucullatus) are more loosely and crudely constructed than nests built by more experienced males.

Left, nest built by older, experienced male Village Weaver and, right, the first nest built by a young male
(Collias and Collias 1964).
Another way of examining the extent to which nest building is innate versus learned is to determine the degree of repeatability of nest morphology, with greater repeatability indicating a behavior with a greater genetic component. Among some species of birds, nest morphology has been found to be very repeatable. For example, Barn Swallows (Hirundo rustica) build cup-shaped nests using mud and straw. Characteristics of the nests of individual swallows were found to be consistent both within and between years and, in addition, the characteristics of nests were even similar across generations, suggesting that nest building by Barn Swallows has a heritable component and is a largely instinctive behavior (Møller 2006). Similarly, the nests of individual Penduline Tits (Remiz pendulinus) exhibit little variation across seasons (Schleicher et al. 1996). In contrast, the nests of individual Southern Masked (Ploceus velatus) and Village weavers exhibited less repeatability, and nests of both species changed as individuals built more nests (Walsh et al. 2010; Figure below). Such results suggest that, for many species of birds, nest building is primarily if not entirely instinctive. However, at least for species that build more complex nests, such as weavers (Ploceidae), nest construction involves both instinct (especially for first nests) and learning.

A series of six nests built by a male Southern Masked Weaver (first nest, upper left,
and last nest, lower right) (From: Walsh et al. 2010).
Weavers
These Birds Build Nests With Mud
The time needed to build a nest varies with the complexity of the nest and other factors such as time of year and weather. In temperate areas, construction of the first nest of the breeding season may take longer than nests later in the season (particular in resident species like Eastern Bluebirds and Northern Cardinals). Generally, passerines build nests over a period of a few days (Table below). Construction of the nests of some larger birds, such as raptors, may require as long as several weeks. Woodpeckers excavate nest cavities in tree trunks or branches, a process that, for species found in North America, typically takes anywhere from 6 to 36 days (Table below).
Time needed for representative songbirds to complete their open-cup nests.
Species |
Typical time to |
Reference |
Least Flycatcher |
5 – 7 days |
Tarof and Briskie 2008 |
Loggerhead Shrike |
6 – 11 days |
Yosef 2020 |
White-eyed Vireo |
3 – 5 days |
Hopp 2022 |
Clark’s Nutcracker |
5 – 8 days |
Schaming et al. 2024 |
Verdin |
about 6 days |
Webster 2020 |
Cactus Wren |
1 – 6 days |
Hamilton et al. 2020 |
Black-tailed Gnatcatcher |
2 – 4 days |
Farquhar and Ritchie 2020 |
Ruby-crowned Kinglet |
about 5 days |
Swanson et al. 2021 |
Northern Wheatear |
2 – 7 days |
Dunn et al. 2022 |
American Robin |
5 – 7 days |
Vanderhoff et al. 2020 |
Gray Catbird |
5 – 6 days |
Smith et al. 2020 |
Cedar Waxwing |
3 – 9 days |
Witmer et al. 2020 |
Northern Yellow Warbler |
4 – 10 days |
Valdez-Juárez et al. 2025 |
Eastern Towhee |
up to 5 days |
Greenlaw 2020 |
Chipping Sparrow |
2 – 8 days |
Middleton 2020 |
Scarlet Tanager |
2 – 7 days |
Mowbray 2020 |
Northern Cardinal |
3 – 9 days |
Halkin et al. 2021 |
Indigo Bunting |
2 – 10 days |
Payne 2020 |
Red-winged Blackbird |
3 days |
Holcomb and Twiest 1968 |
Orchard Oriole |
6 days |
Scharf and Kren 2022 |
Pine Siskin |
5 - 6 days |
Dawson 2020 |
Time needed for representative woodpeckers to create nest cavites.
Species |
Typical time to |
Reference |
Black-backed Woodpecker |
21 – 28 days |
Tremblay et al. 2020 |
Downy Woodpecker |
7 – 20 days |
Ritchison 1999 |
Golden-fronted Woodpecker |
6 – 12 days |
Husak and Maxwell 2020 |
Hairy Woodpecker |
7 – 21 days |
Bent 1939 |
Pileated Woodpecker |
21 – 36 days |
Bull and Jackson 2020 |
Red-bellied Woodpecker |
~14 days |
Miller et al. 2020 |
Red-headed Woodpecker |
12 – 17 days |
Frei et al. 2020 |
White-headed Woodpecker |
21 – 28 days |
Kozma et al. 2025 |
These engineers really soar -- If you have any doubt that birds are master builders, try this: Get a bunch of thin twigs and grass and try making a nest yourself. And no fair using your hands birds, remember, only use their beaks. Douglas Causey of Harvard University's Museum of Comparative Zoology has asked young students to do just that. You can imagine the result. But if you think making a simple nest is difficult, imagine building a more elaborate nest. Take, for example:
A South American ovenbird (Rufous Hornero, Furnarius rufus), which may take months to fashion one nest from clay or mud mixed with bits of straw, hair, and fibers. The tropical sun bakes the walls brick-hard.


Or Bald Eagles that use sticks, some two inches thick and several feet long, to make nests sturdy enough to support a human adult. They may look like a jumble of materials, but the sticks are usually placed in layers, beginning with a triangle, followed by more rotated, triangular layers.

Birds are capable of grand engineering feats. But are they engineers? Not in the way you might think. Just as birds know how to fly, they know how to build a nest without instructions or apprenticeship. It's a matter of instinct. "They are 'hard-wired,' " Causey says, "sort of like robots." Birds craft their nests without consciously thinking about it. How then did some species of birds develop such well-engineered, elaborate nests? One possible explanation involves natural variation and evolution. If a particular bird happens to build a nest that is stronger or more predator-safe, that bird's offspring are more likely to survive and pass along this trait to succeeding generations. Another possibility is that when females choose mates based on the quality of the nests they build, this means the best nest builders are more likely to breed. Nest-building, therefore, may still be evolving, but so slowly that no one really detects any change. This makes nest-building one of the most difficult bird behaviors to understand.
Generally, in temperate areas, nest-building coincides with the arrival of spring. That's when birds' internal "clocks" tell them it's time to mate and raise a brood. In the tropics, though, some birds build nests and breed year-round. The nest, of course, is where females lay eggs and brood young. Which bird actually does the building the male or female varies by species. In some cases, both collect material to build the nest and join in its construction. One of the more peculiar routines is that of the male Marsh Wren (Cistothorus palustris), which migrates north before the female and builds as many as 10 dummy nests in his territory. These dummy nests are not lined with soft materials; lining is added by the female only to the chosen nest. Dummy nests have been shown to have adaptive importance, with active nests built near larger numbers of dummy nests being more successful than those near fewer dummy nests (Leonard and Picman 1987).
We usually think of nests in tree branches. But some birds build nests on the ground, some in bushes. Others might attach their nests to the sides of cliffs. Barn Swallows have an affinity for barns. Chimney Swifts, as the name implies, favor chimneys and other man-made enclosures. Carolina Wrens will nest in almost any cavity, from an empty can to a coat pocket. To keep nests together and secured in place, birds need good adhesives. They use a variety of natural substances to do the job, including mud, saliva, spiderwebs, caterpillar silk, leaf mold, and certain plant fibers. Materials that make up the nest are intertwined, and with the weaver bird, are actually woven or thatched together using grass, strips of leaves, and twigs.
Birds can make hundreds of trips to collect materials. And while they seem to prefer natural objects (helpful as camouflage), some use almost anything that works and that they can carry. Candy wrappers, cellophane, shredded money even barbed wire have shown up in nests. But however coarse the outside of the nest is, the inside is usually lined with soft materials to make it comfy.
One theory about why birds build open nests is that they had to. Larger species shooed them out of the tree holes. Birds intent on avoiding bullies and predators may build nests that have coverings or that hang from the end of tree branches. Less aggressive birds may even build near more aggressive species, for protection. Although some large birds (eagles, hawks, and sea birds) return to nests they've made, most birds rarely do. Eagles may return annually to the huge platform nests they build in the treetops. Some measure as much as 6 feet in diameter. On the flip side are hummingbirds, which fashion tiny cup-like nests not much bigger than a thimble.
Nest-collecting was popular from about 1870 to 1920, says Douglas Causey, ornithologist at Harvard University. Most of the nests in the museum's collection date to that period. Back then, collectors didn't have the ecological awareness that people have today. They would climb trees and saw off branches to "collect" a nest with its eggs intact. Once museums had at least one of each, searching for more made no sense. From about 1920 to 1970, nest and egg collections gathered dust. "They just took up space, and no one knew what to do with them," Causey says. Some were thrown away. During the 1970s, though, interest in bird eggs came back. Scientists noticed that eggshells of some birds had become thinner and more breakable. The insecticide DDT was found to be the cause. Researchers compared the thickness of old eggshells with new. Their findings helped persuade Congress to ban the pesticide. Without eggs from 100 years ago, no comparisons could have been made. Egg and nest collections are important time capsules. They tell of environmental conditions at particular times. This, Causey says, is a good reason to collect more today. Researchers are now examining the old plant material in nests to determine how much carbon monoxide was in the atmosphere decades ago.
In the United States, it is unlawful to collect the nests of migratory birds, unless for approved scientific study. Although not strictly enforced, the law exists to protect birds in the wild from disturbance. When ornithologists study a nest, they know that if it has been unattended for several days, it's usually safe to conclude that the birds have gone and won't be back. -- Ross Atkin (Copyright © 2002 The Christian Science Monitor).
Literature Cited:
Altamirano et al. 2023. Rock cavity nesting as the norm: Breeding songbirds of the temperate High Andes. Ecology 104: e3931.
Bent, A. C. 1939. Life histories of North American woodpeckers. U.S. National Museum Bulletin 174, Washington, D.C.
Biancucci, L., and T. E. Martin. 2010. Can selection on nest size from nest predation explain the latitudinal gradient in clutch size? Journal of Animal Ecology 79: 1086-1092.
Bull, E. L., and J. A. Jackson. 2020. Pileated Woodpecker ( Dryocopus pileatus), version 1.0. In: Birds of the World (A. F. Poole, ed.). Cornell Lab of Ornithology, Ithaca, NY.
Calder, W. A. 1973. Microhabitat selection during nesting of hummingbirds in the Rocky Mountains. Ecology 54: 127-134.
Chia et al. 2023. A global database of bird nest traits. Scientific Data 10: 923.
Chiappe, L. M. 2009. Downsized dinosaurs: the evolutionary transition to modern birds. Evolution: Education and Outreach 2:248-256.Collias, E. C., and N. E. Collias. 1964. The development of nest-building behavior in a weaverbird. Auk 81: 42-52.
Corimanya, J. L., E. B. Smith, and W. A. Boyle. 2024. Experimental evidence that nest orientation influences microclimate in a temperate grassland. Journal of Field Ornithology 95:2.
Darolova, A., H. Hoi, and B. Schleicher. 1997. The effect of ectoparasite nest load on the breeding biology of the Penduline Tit Remiz pendulinus. Ibis 139: 115-120.
Dawson, W. R. 2020. Pine Siskin ( Spinus pinus). In: Birds of the World (A. Poole, ed.). Cornell Lab of Ornithology, Ithaca, NY.
Dial, K. P. 2003. Evolution of avian locomotion: correlates of flight style, locomotor modules, nesting biology, body size, development,and the origin of flapping flight. Auk 120:941-952.
Dunn, E. H., D. J. T. Hussell, J. Kren, and A. C. Zoerb. 2022. Northern Wheatear ( Oenanthe oenanthe). In: Birds of the World (P. G. Rodewald and B. K. Keeney, eds.). Cornell Lab of Ornithology, Ithaca, NY.
Farquhar, C. C., and K. L. Ritchie. 2020. Black-tailed Gnatcatcher ( Polioptila melanura). In: Birds of the World (A. F. Poole and F. B. Gill, eds.). Cornell Lab of Ornithology, Ithaca, NY.
Frei, B., K. G. Smith, J. H. Withgott, P. G. Rodewald, P. Pyle, and M. A. Patten. 2020. Red-headed Woodpecker ( Melanerpes erythrocephalus). In: Birds of the World (P. G. Rodewald, ed.). Cornell Lab of Ornithology, Ithaca, NY.
Ghent, A.W. 2001. Regular spatial patterns of Bank Swallow (Riparia riparia) tunnel entrances, with some possible evolutionary implications. American Midland Naturalist 146: 414-423.
Greenlaw, J. S. 2020. Eastern Towhee ( Pipilo erythrophthalmus). In: Birds of the World (P. G. Rodewald, ed.). Cornell Lab of Ornithology, Ithaca, NY.
Halkin, S. L., Daniel P. Shustack, M. Susan DeVries, Jodie M. Jawor, and Susan U. Linville. 2021. Northern Cardinal ( Cardinalis cardinalis). In: Birds of the World (A. Poole, ed.). Cornell Lab of Ornithology, Ithaca, NY.
Hamilton, R. A., G. A. Proudfoot, D. A. Sherry, and S. L. Johnson. 2020. Cactus Wren ( Campylorhynchus brunneicapillus). In: Birds of the World (A. Poole, ed.). Cornell Lab of Ornithology, Ithaca, NY.
Hartman, C.A. and L. W. Oring. 2003. Orientation and microclimate of Horned Lark nests: the importance of shade. Condor 105:158-163.
Holcomb, L. C., and G. Twiest. 1968. Ecological factors affecting nest building in Red-winged Blackbirds. Bird-Banding 39: 14-22.
Hopp, S. L. 2022. White-eyed Vireo ( Vireo griseus). In: Birds of the World (P. G. Rodewald and B. K. Keeney, eds.). Cornell Lab of Ornithology, Ithaca, NY.
Husak, M. S., and T. C. Maxwell. 2020. Golden-fronted Woodpecker ( Melanerpes aurifrons). In: Birds of the World (A. F. Poole and F. B. Gill, eds.). Cornell Lab of Ornithology, Ithaca, NY.
Jung et al. 2021. Avian mud nest architecture by self-secreted saliva. Proceedings of the National Academy of Sciences 118: e2018509118.
Kozma, J. M., T. J. Lorenz, M. G. Raphael, K. L. Garrett, R. D. Dixon, G. M. Kirwan, and S. M. Billerman. 2025. White-headed Woodpecker ( Picoides albolarvatus), version 2.2. In: Birds of the World (P. G. Rodewald, B. K. Keeney, and M. G. Smith, eds.). Cornell Lab of Ornithology, Ithaca, NY.
Leite et al. 2024. Nest attachment, rather than nest type, correlateswith passerine bird brain size. Ibis 166: 814825.
Leonard, M.L. and J. Picman. 1987. The adaptive significance of multiple nest building by male Marsh Wrens. Animal Behaviour 35:271-277.
Mainwaring et al. 2023. The evolution of nest site use and nest architecture in modern birds and their ancestors. Philosophical Transactions of the Royal Society B 378: 20220143.
Martins et al. 2026. Why do birds construct nest tails? A test of disruptive camouflage in the Blue Manakin. Biology Letters 22: 20250453.
Medina et al. 2022. Nest architecture is linked with ecological success in songbirds. Ecology Letters 25: 1365-1375.
Merino, S. and J. Potti. 1996. Weather dependent effects of nest ectoparasites on their bird hosts. Ecography 19: 107–113.
Middleton, A. L. 2020. Chipping Sparrow ( Spizella passerina). In: Birds of the World (A. F. Poole and F. B. Gill, eds.). Cornell Lab of Ornithology, Ithaca, NY.
Møller, A. P. 2006. Rapid change in nest size of a bird related to change in a secondary sexual character. Behavioral Ecology 17: 108-116.
Minias, P. and T. Janiszewski. 2023. Ground nesting in passerine birds: evolution, biogeography and life history correlates. Oikos 2023: e09870.
Mowbray, T. B. 2020. Scarlet Tanager ( Piranga olivacea). In: Birds of the World (A. F. Poole and F. B. Gill, eds.). Cornell Lab of Ornithology, Ithaca, NY.
Myers, P., R. Espinosa, C. S. Parr, T. Jones, G. S. Hammond, and T. A. Dewey. 2005. The Animal Diversity Web (online). Accessed November 27, 2005 at http://animaldiversity.org.
Payne, R. B. 2020. Indigo Bunting ( Passerina cyanea). In: Birds of the World (A. F. Poole and F. B. Gill, eds.). Cornell Lab of Ornithology, Ithaca, NY.
Petit, C., M. Hossaert-McKey, P. Perret, J. Blondel & M.M. Lambrechts. 2002. Blue Tits use selected plants and olfaction to maintain an aromatic environment for nestlings. Ecology Letters 5: 585 - 589.
Pettingill, O.S., Jr. 1985. Ornithology in Laboratory and Field, Fifth ed. Academic Press, New York, NY.
Proctor, H. and I. Owens. 2000. Mites and birds: diversity, parasitism and coevolution. Trends in Ecology and Evolution 15: 358–364.
Reneerkens, J., T. Piersma, J. S. Sinninghe Damsté. 2002. Sandpipers (Scolopacidae) switch from monoester to diester preen waxes during courtship and incubation, but why? Proc. Roy. Soc. B 269: 2135-2139.
Schaming, T. D., D. F. Tomback, and T. J. Lorenz. 2024. Clark's Nutcracker ( Nucifraga columbiana). In: Birds of the World (N. D. Sly, ed.). Cornell Lab of Ornithology, Ithaca, NY.
Scharf, W. C., and J. Kren. 2022. Orchard Oriole ( Icterus spurius). In: Birds of the World (P. G. Rodewald, ed.). Cornell Lab of Ornithology, Ithaca, NY.
Sheard et al. 2023. Nest traits for the world's birds. Global Ecology and Biogeography 33: 206-214.
Street et al. 2022. Convergent evolution of elaborate nests as structural defences in birds. Proceedings of the Royal Society B 289: 20221734.
Taylor et al. 2024. The fecundity costs of building domed nests in birds. Journal of Evolutionary Biology 37: 13781385.
Tremblay, J. A., Rita D. Dixon, Victoria A. Saab, Peter Pyle, and Michael A. Patten. 2020. Black-backed Woodpecker ( Picoides arcticus). In: Birds of the World (P. G. Rodewald, ed.). Cornell Lab of Ornithology, Ithaca, NY.
Valdez-Juárez, S. O., P. E. Lowther, C. Celada, N. K. Klein, C. C. Rimmer, P. Pyle, G. M. Kirwan, S. M. Billerman, P. F. D. Boesman, and A. J. Spencer. 2025. Northern Yellow Warbler ( Setophaga petechia). In: Birds of the World (S. M. Billerman, ed.). Cornell Lab of Ornithology, Ithaca, NY.
Vanadzina et al. 2023. Global drivers of variation in cup nest size in passerine birds. Journal of Animal Ecology 92: 338351.
van der Hoek et al. 2017. The diversity, distribution and conservation status of the tree- cavity- nesting birds of the world. Diversity and Distributions 23: 11201131.
Walsh, P. T., M. Hansell, W. D. Borello, and S. D. Healy. 2010. Repeatability of nest morphology in African weaver birds. Biology Letters 6: 149-151.
Webster, M. D. 2020. Verdin ( Auriparus flaviceps). In: Birds of the World (A. F. Poole and F. B. Gill, eds.). Cornell Lab of Ornithology, Ithaca, NY.
Welty, J.C. and L. Baptista. 1988. The life of birds, fourth ed. Saunders College Publishing, New York, NY.
Witmer, M. C., D. J. Mountjoy and L. Elliot. 2020. Cedar Waxwing ( Bombycilla cedrorum), version 1.0. In: Birds of the World (A. F. Poole, ed.). Cornell Lab of Ornithology, Ithaca, NY.
Yosef, R. 2020. Loggerhead Shrike ( Lanius ludovicianus). In: Birds of the World (A. F. Poole and F. B. Gill, eds.). Cornell Lab of Ornithology, Ithaca, NY.