Ornithology
Digestive System: Food & Feeding Habits

Hit 'Reload' or 'Refresh' to View Again!
Ornithology Digestive System: Food & Feeding Habits |
 Hit 'Reload' or 'Refresh' to View Again! |
Because of their high metabolic rates, birds must consume more food in proportion to their size than most animals. For example, a warbler might eat 80 percent of its body weight in a day. As a group, birds consume just about any type of food you can imagine, including amphibians, ants, buds, carrion, crustaceans, fish, fruit, grass, insects, larvae, leaves, molluscs, nectar, other birds, pollen, reptiles, rodents, roots, sap, seeds, suet, snails, wax, & worms. Birds acquire these food items in a wide variety of ways.

Major foraging guilds of birds. Vertivores prey primarily on vertebrates and invertivores on invertebrates, granivores are primarily seed eaters,
frugivores feed primarily on fruit, scavengers feed on carrion, nectarivores primarily on nectar, herbivores on plants, and aquatic predators on a wide variety of aquatic prey
(Figure from Pigot et al. 2020).

(a) Seven major foraging niches used by terrestrial invertivores, and (b) six major foraging niches used by aquatic predators (Figure from Pigot et al. 2020).
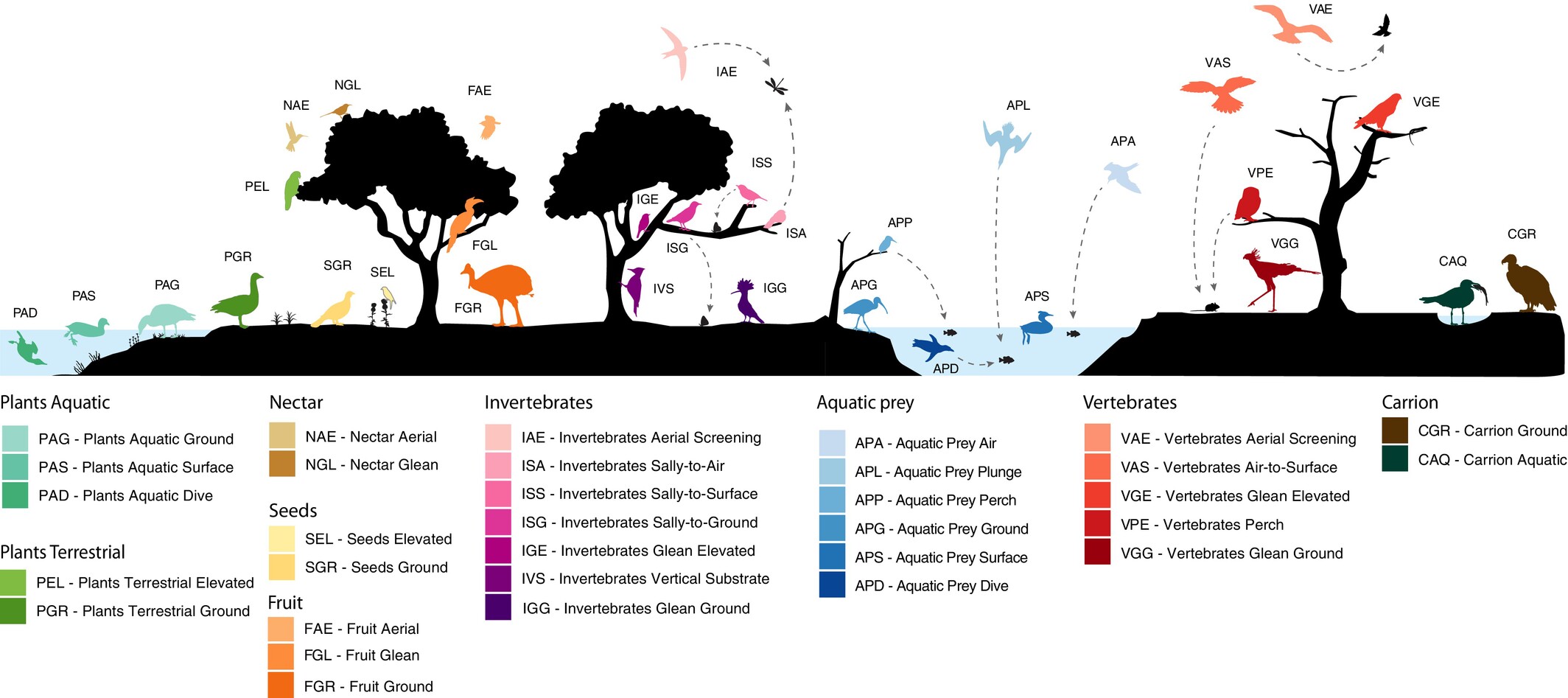
More detailed view of 32 foraging niches of birds. Plants aquatic=species obtaining food resources from plant materials in aquatic systems, including algae
and aquatic plant leaves. Plants terrestrial=species obtaining food resources from other plant materials in non-aquatic systems, including leaves,
buds, whole flowers, etc. Aquatic prey=species obtaining food resources from vertebrate and invertebrate animals in aquatic systems, including fish,
crustacea, molluscs, etc. (From: Sayol et al. 2026).
For birds that fly, mass affects flying ability so, in general, they need to keep their weight at some optimal level. That means that, except perhaps prior to migration, there is a limit to the amount of fat a bird can store. 'Efficient' means that birds must locate, ingest, & digest food as quickly and efficiently as possible.
|
of representative reptiles, birds, & mammals (Based on: Stevens and Hume 1998).
|
In general, typical mean retention times are 30 - 50 minutes for
avian
nectarivores, 40 - 100 minutes
for granivores, and 15 - 60 minutes for frugivores (Karasov 1990,
Klasing
1998).
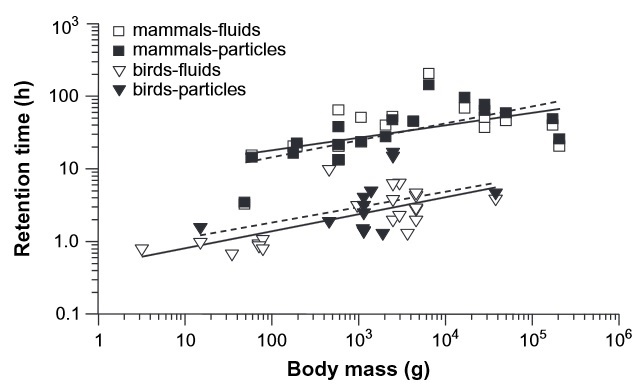
Retention times of fluids (solid lines) and particulate materials (dashed lines) markers in the gastrointestinal system of birds (triangles)
is shorter than that of mammals (squares). Even with shorter retention times, the digestive efficiency (energy extracted relative to food intake)
of birds
is similar to
that of mammals (Figure from McWhorter et al. 2009).
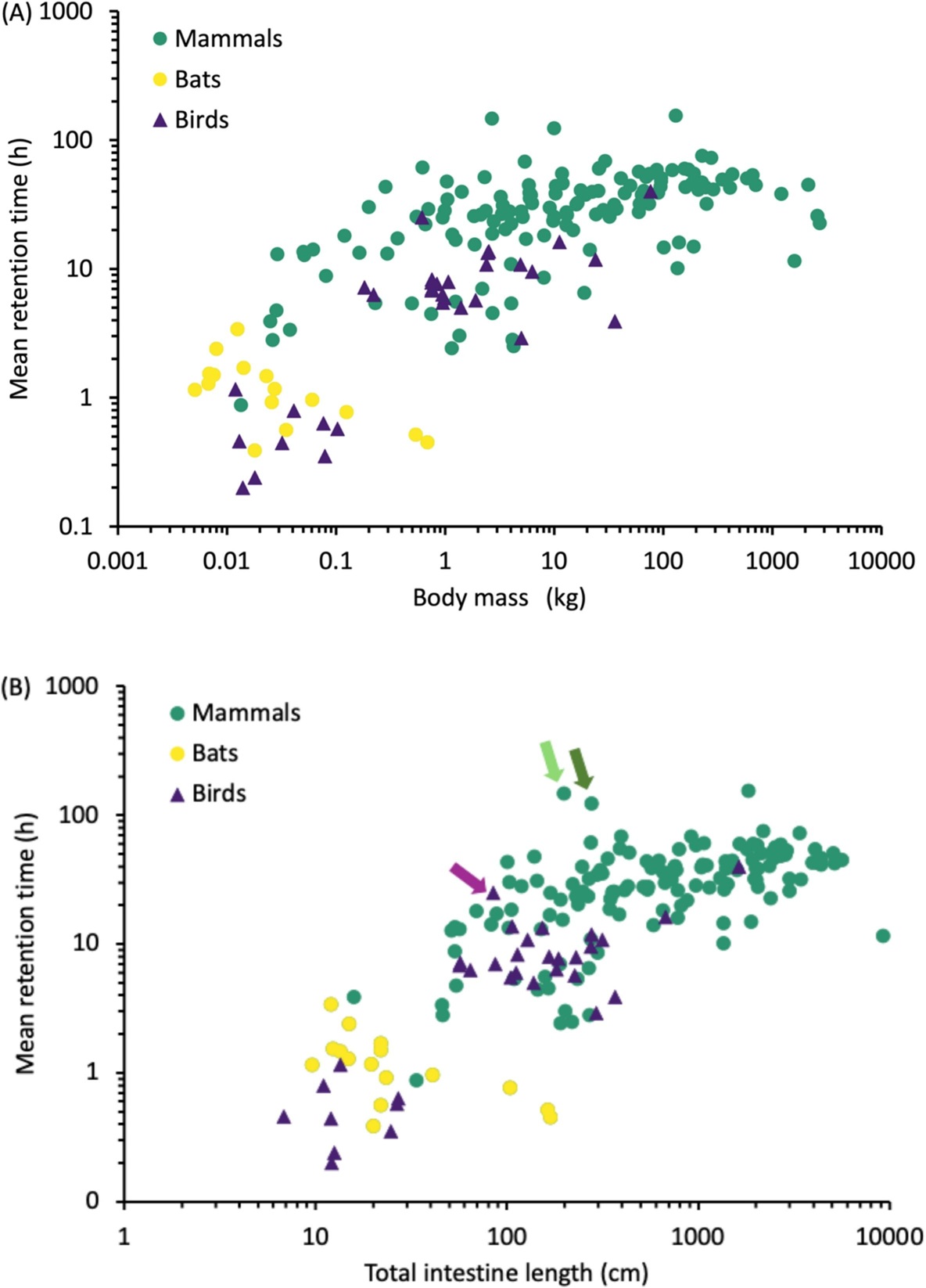
Relationships in mammals (including bats) and birds of the digesta mean retention time in the digestive
tract with (A) body mass or (B) total intestine length. In B, the bird Hoatzin (Opisthocomus hoazin, light
purple arrow) and the sloths (mammal; the three-toed sloth, Bradypus tridactylus - light green arrow,
and the two-toed sloth, Choloepus didactylus - dark green arrow) are marked by arrows as examples of
species where a large foregut fermentation chamber is associated with long mean retention times despite
comparatively short intestinal tracts. Note that retention times were generally shorter for most birds and
bats than for terrestrial mammals, with smaller size (i.e., lower body mass) and, more importantly, shorter
total intestinal length being contributing factors (From: Duque-Correa et al. 2025).
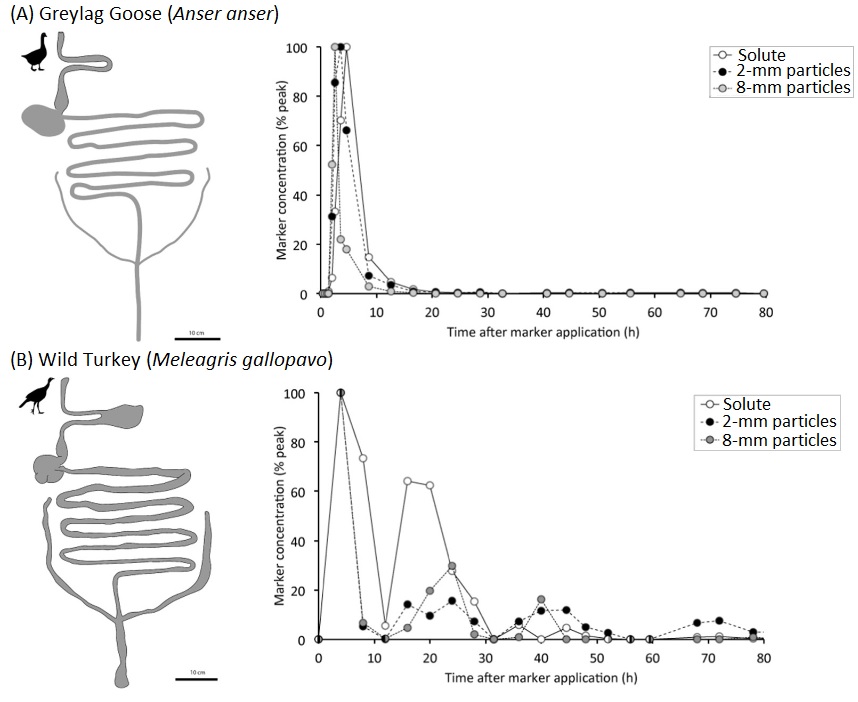
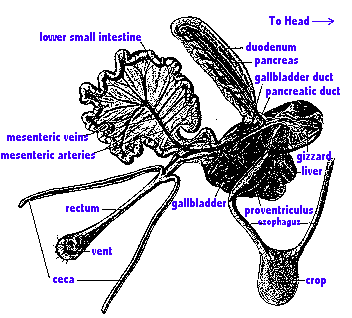
Here's one example of an avian digestive system:

Digestive system of a Domestic Chicken. The major components of the avian digestive system are the
alimentary
canal plus several accessory structures.
The 'canal' includes
the oral
cavity, pharynx,
esophagus (which includes a crop in some birds),
stomach
(proventriculus & gizzard), small intestine, & large intestine.
The large intestine then empties
into the cloaca. Important accessory
structures
include the beak, salivary glands, liver, & pancreas.
A bird's bill consists of a bony framework covered by a tough layer of keratin (called the rhamphotheca). The keratin layer is continuously replaced throughout the life of a bird & is just as continuously worn down by eating and manipulating objects. Birds use their bills in a wide variety of ways, including preening, vocal modulation, thermoregulation, nest construction, tool use (for a few species), and, of course, food acquisition. Most birds have fast gapes and relatively weak bites, with bills serving as versatile, tweezer-like clamps (Navalon et al. 2018). Fewer birds have slower gapes and high biting force (Navalon et al. 2018). Examples of the latter include parrots and other seed-eating birds, plus some birds that often kill prey with their bills (e.g., falcons and shrikes).
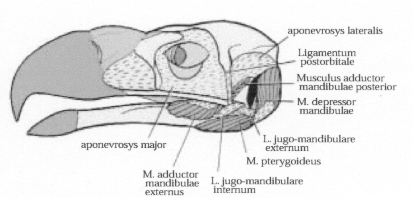
Ligaments & muscles on the skull of a Steller's Sea Eagle.
Muscles that close the jaw include the adductor mandibulae externus, adductor mandibulae posterior, & pterygoideus.
The depressor mandibulae opens the jaw (Source: Ladyguin 2000)
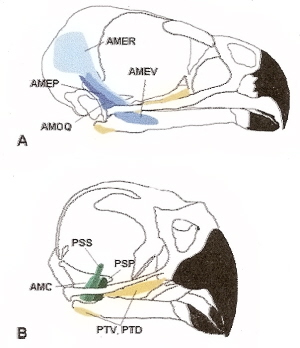
Muscles involved in jaw closure for a Cooper's Hawk. A. Lateral view showing adductor mandibulae group, including
adductor mandibulae externus pars profunda (AMEP), adductor mandibulae ossis quadrati (AMOQ), adductor mandibulae
externus pars rostralis (AMER), adductor mandibulae pars ventralis (AMEV). B. Antero-lateral view showing the
pseudotemporalis group, including adductor mandibulae caudalis (AMC), pseudotemporalis profundus (PSP),
pseuodotemporalis superficialis (PSS), pterygoideus pars dorsalis (PTD), and pterygoideus pars ventralis (PTV)
(Sustaita 2008).
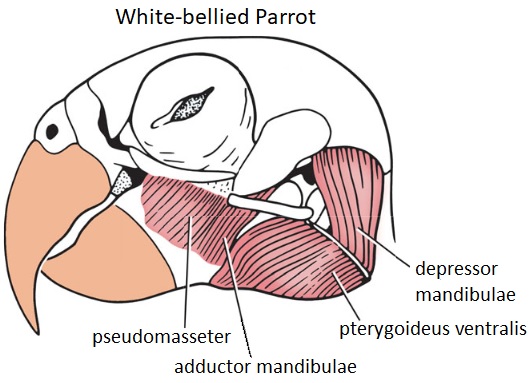
Jaw musculature of a White-bellied Parrot
(Figure modified from Sereno et al. 2010)
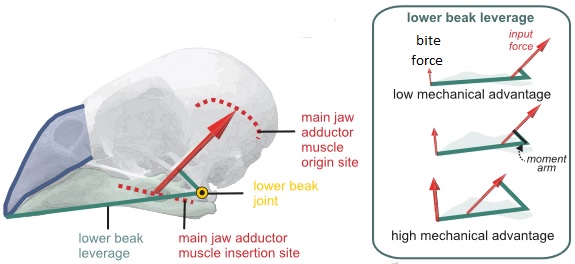
High bite forces are generated when the moment arm of the muscles’ input force is relatively high
(Figure from Van Wassenbergh and Baeckens 2019).
Variation in bill morphology
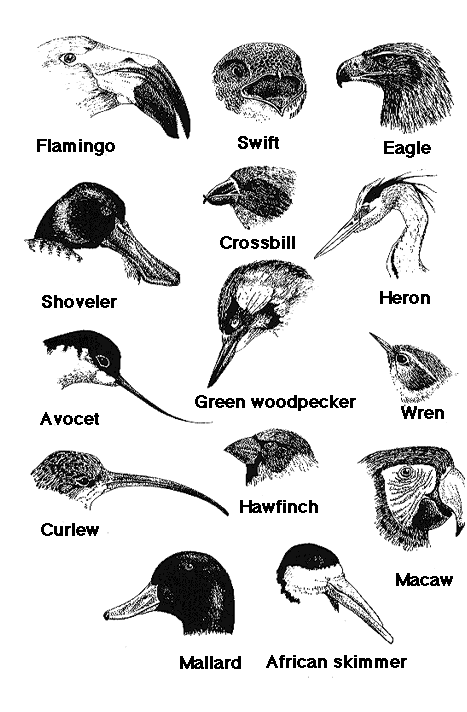
|
|
Great Egret
African Fish Eagle
Golden Eagle vs. a goat
Harris's Hawks hunt a jackrabbit
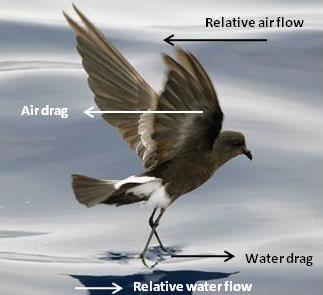
Sea-anchor 'soaring' by storm-petrels -- As described by Pennycuick (2008), storm-petrels have a unique method for
picking up food from the water's surface. They glide into the wind with their body clear of the surface, but with their webbed feet
in
the water, helping to 'anchor' them. Aerodynamic (or air) drag on on wings (friction drag) and body (parasitic drag) push
them backward through the water so that the feet tend to slowly move backward through the water, and that creates forward-directed
hydrodynamic (or water) drag. When this water drag balances the aerodynamic (air) drag, the storm-petrels remain in place, suspended above
the water's surface at a height that allows them to pick small food items from the surface (Photo source: Wikipedia).
Pileated Woodpecker

A Pileated Woodpecker striking a wood substrate with its bill (times are in milliseconds). Note how the force of the impact causes
the woodpecker’s crest to move forward (from 63.1 to 68.1 ms), after which the crest feathers return to their original position (87.5 ms) (From Ross 2014).
Woodpecker shock-absorbing system -- Woodpeckers are known to drum hard woody surfaces of trees at a rate of 18 to 22 times per second with a deceleration of 1200 g (humans can lose consciousness at g-forces as low as 4 to 6 g). Woodpeckers have four structures that help absorb mechanical shock and prevent brain damage: (1) a hard, but elastic, beak, (2) a hyoid apparatus that rigidly supports the tongue, (3) an area of spongy bone located at the contrecoup position from the beak, and (4) skull bone with cerebrospinal fluid (CSF). A woodpecker's hyoid extends posteriorly from the floor of the oral cavity, goes behind the neck, divides into two bands, goes around the back of the skull, and inserts at the front of the skull. This allows woodpeckers to extend their tongues well beyond the tip of the beak when foraging, but this arrangement also helps distribute mechanical vibrations when drumming. The spongy bone is thought to evenly distribute mechanical vibrations before they propogate to the brain. Finally, woodpeckers have a very narrow space for CSF between the skull and brain, and little CSF helps reduce transmission of vibrations into the brain through the CSF (Source: Yoon and Park 2011).
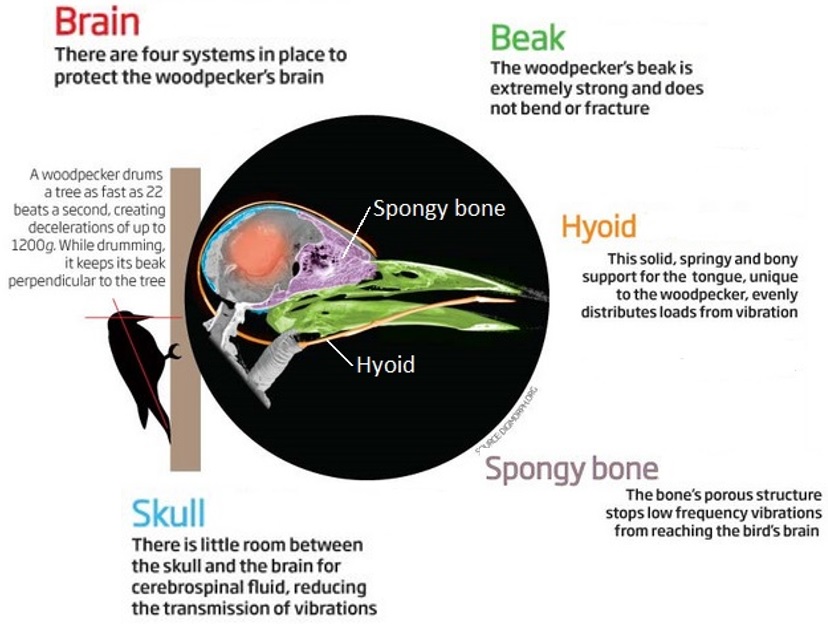
Links:
Woodpecker's head inspires shock absorbers
Compare with humans
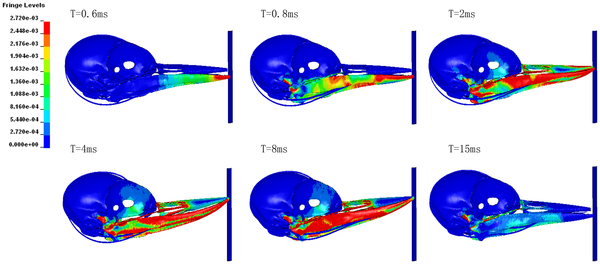
The unequal length of the upper and lower parts of woodpecker beaks (the lower being longer) directs the force of impact downwards,
away from the brain, when it hits the tree (red coloration indicates the greatest force or stess; blue indicates the least force or stress). Time after impact proceeds from upper left (0.6 milliseconds after impact) to lower right (15 milliseconds after impact).

The hyoid of woodpeckers loops over top of the skull to completely surround their skulls. The hyoid helps direct the stress of impact below and around the skull (and brain) and also acts like a 'safety belt', helping to keep the skull and brain in place. It is the movement of the brain inside the skull during impact, more than the blow itself, that causes concussions. If the brain is held in place, injury risks are greatly reduced. As in the above figure, time after impact proceeds from upper left to lower right (Figures from Wang et al. 2011).
More recently, the results of a study by Van Wassenbergh et al. (2022) suggest that woodpecker brains do not require a shock-absorbing system (see video below)
(https://www.cell.com/current-biology/pdf/S0960-9822(22)00855-7.pdf). However, it may be the case that, over a woodpecker's lifetime, the various mechanisms to minimize
shock described above may still prove beneficial in minimizing damage to woodpecker brains.
The capillary ratchet mechanism
Surface tension transport of prey by feeding shorebirds: the capillary ratchet -- Prakash et al. (2008) found that phalaropes and other shorebirds take advantage of surface interactions between their beak and water droplets to propel bits of food from the tip of their long beaks to their mouth. They peck at the surface, picking up droplets of water with prey inside. Because their beaks point downward when feeding, gravity must be overcome to get those droplets from the tip of the bird's long beak to its mouth. This feeding strategy depends on surface tension. As the beak opens and closes, each movement propels the water droplet one step closer to the bird's mouth. Specifically, when the beak closes, the drop's leading edge moves toward the mouth, while the trailing edge stays put. In this stepwise ratcheting fashion, the drop travels along the beak at a speed of about 1 meter per second. The efficiency of the process, called the "capillary ratchet," depends on beak shape, and long, narrow beaks, like those of phalaropes, are best suited to this mode of feeding. - MIT News
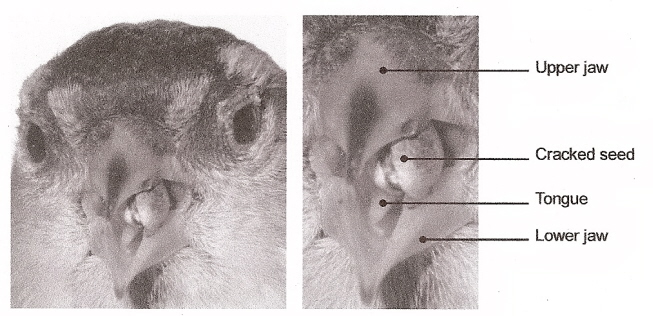
| Finches do not simply bite the seeds; instead; the lower mandible is moved toward the tip of the bill in a slicing motion. When most of the coat has been cracked or removed, the lower mandible is moved from side to side to remove the rest of the shell, thus releasing the kernel. Some large finches also have raised hard surfaces in the upper palate that function as anvils so large seeds can be held firmly while the lower mandible slices and cracks the sides of the seed. As tricky as nutcracking sounds, most birds accomplish it rapidly, shelling small seeds in a few seconds and large finches can crack open and devour a large seed or nut in less than 20 seconds. | 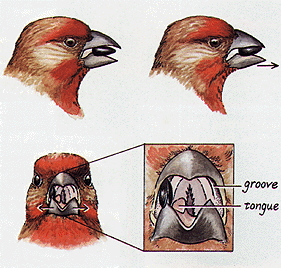 |
Serin (Serinus serinus) with a seed positioned in its bill. Note how the tongue is used to hold the seed in position
(From: van der Meij 2005).
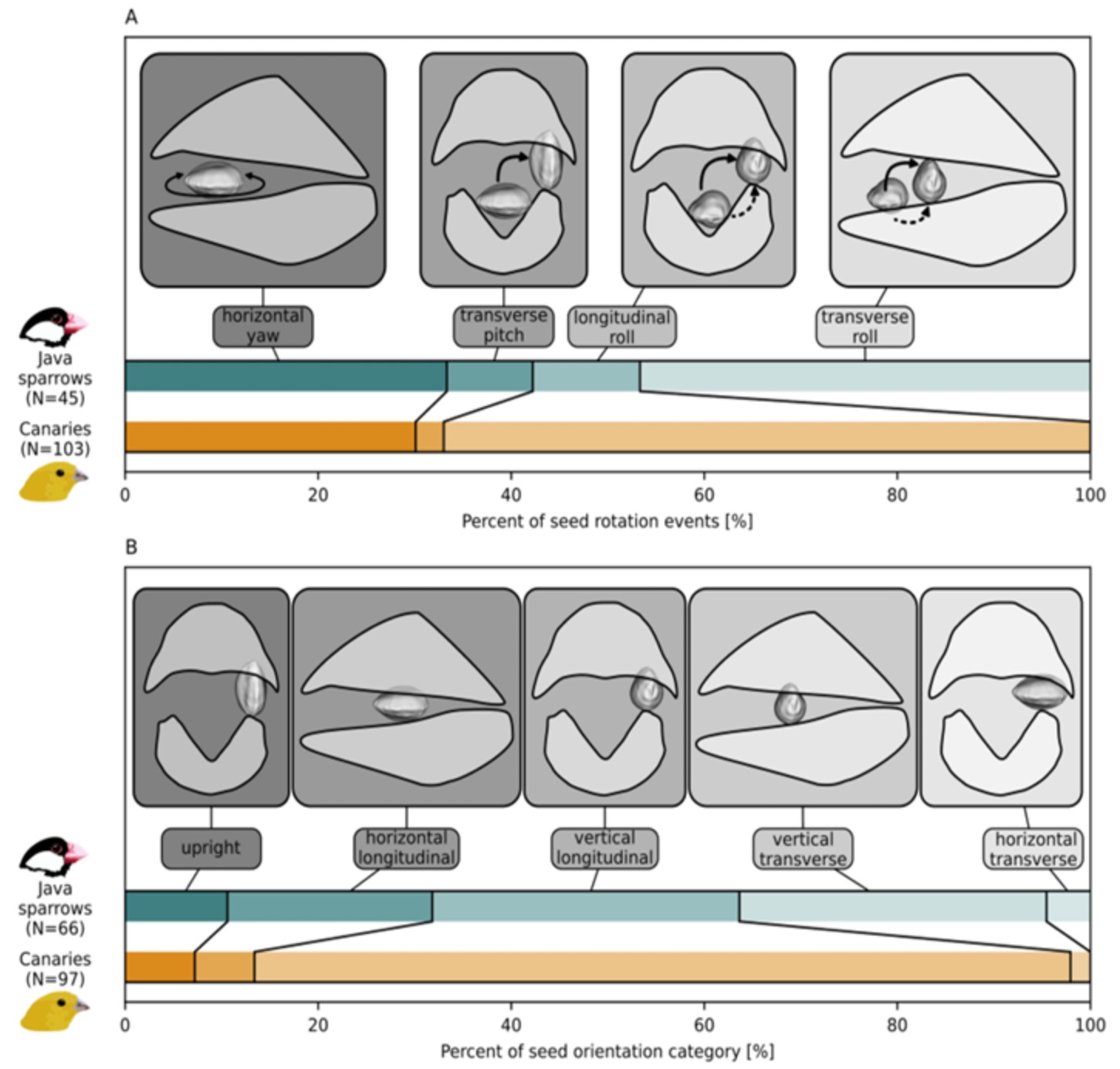
Birds use distinct types of seed rotation to position and crack open seeds. A) Four dominant types of seed
rotation in Domestic Canaries and Java Sparrows. Solid arrows in (A) indicate approximate typical trajectories
of seeds during rotation, dashed arrows indicate alternative options of trajectories that the birds used only
occasionally. B) Five dominant types of seed orientation during biting in canaries and Java Sparrows.
Upright refers to a special case of transverse orientation, with the long axis of the seed lying parallel to
the sagittal plane of the beak. In A+B, longitudinal and transverse refer to the long axis of the seed being parallel
and orthogonal to the long axis of the beak, respectively. Seed rotations and orientations are visualized
on only one side for clarity. Seeds and beaks are not drawn to scale. N denotes the sample size (number of
analyzed seed rotation or biting events). Wide boxes show beaks in lateral view, narrow boxes show
beaks in frontal view (From: Mielke et al., bioRxiv preprint; doi.org/10.64898/2026.04.22.720076).
Chickadee cracking open and eating safflower and sunflower seeds (with slow motion)

Photo source: http://www.homepages.mcb.net/wormwell/Gannet130604.htm
The plunge dive of the Cape Gannet -- Gannets have one of the most spectacular prey-capture behaviors of all marine predators,
plummeting from up to 30 m into the water, where they seize fish with their razor-sharp beaks. Ropert-Coudert et al. (2004) monitored
the biomechanics of plunge diving in 25 free-ranging Cape Gannets Morus capensis using a rapid-sampling acceleration and depth
recorder. Their data provide the first detailed description of this highly specialized foraging technique. They recorded no or a very low
deceleration when Gannets entered the water, which underlines the remarkable streamlining of this large bird. Birds use their momentum
to travel underwater at an average descent rate of 2.87 m/s (sd = ±1.53) before actively braking once they attain the desired depth (range:
0.3–9.7 m). Ropert-Coudert et al. (2004) showed that Gannets sometimes used either their wings or feet for underwater propulsion during
the course of 9.4% of the dives that had undulations in their depth profiles. After chasing prey, birds developed an upward momentum before
gliding passively back to the surface, making use of their buoyancy to complete the dive at the lowest possible energy cost.
(Check the Gannet videos at ARKive).
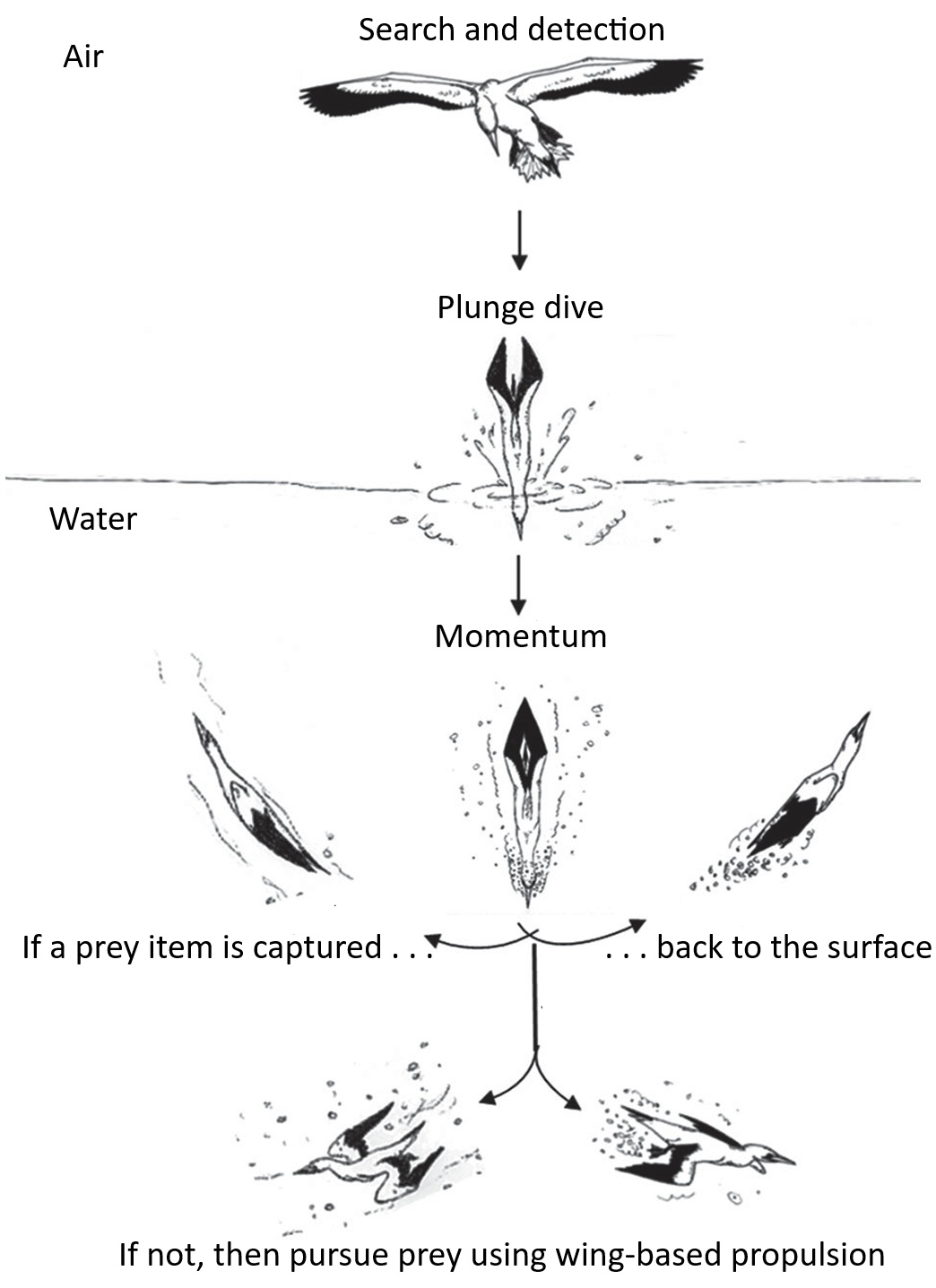
Plunge-diving Australasian Gannets. Gannets (Morus spp., Sulidae) capture pelagic prey (fish and squid)
by plunge diving into the sea from heights often exceeding 5 meters. Once submerged using their momentum,
they either adopt a V-shaped dive profile (if they capture a prey item), where they surface immediately after
the plunge (sometimes using a short phase of wing flapping). If no prey item is captured, then gannets
actively pursue prey using wing-based location. Thus, gannets have flexible underwater hunting tactics, where
they use their speed and momentum to quickly capture prey or, if not, switch their feeding style to active
pursuit (From: Machovsky-Capuska et al. 2012).
Northern Gannets
Feet and talons
Although not part of the digestive system in an anatomical sense, some
birds, like hawks and owls,
use their feet and talons to capture prey. Typically, raptor
prey are killed by the talons of the contracting foot being driven
into their bodies; if required, the hooked bill is used to kill prey
being
held by the talons.
The raptor digital tendon locking mechanism -- Digital tendons form a mechanical-locking mechanism in many birds that must maintain a degree of grip force, including perching, hanging, tree-climbing, and raptorial species. In raptors, powerful hindlimb muscles produce a strong grasp, and a tendon
locking mechanism (TLM) helps sustain grip force. The components of the digital TLM include a 'textured' pad on the ventral
surface of each flexor tendon that contains thousands of minute, rigid,
well-defined projections called tubercles (see figure below). The neighboring portion of the surrounding tendon sheath contains a series of transversely running plicae (folds) that often have a proximal slant (i.e. towards the base of the toe). When the flexor tendons are pulled taut, and the digits flexed, the tubercle pad moves proximally over the stationary plicae on the sheath. When resistance to digital flexion is met, the locking elements intermesh and engage and the friction produced prevents slippage of the tendons. This permits digital flexion to be maintained with little or no muscular involvement (Einoder and Richardson 2006). In sum, differences in grip
force & the
hunting behavior of owls and hawks suggest at least
a partial basis for resource partitioning in the
eastern deciduous forests of North America. Each raptor
has a
unique force production, along with a different time of activity, that
would allow for a degree of prey specialization.
The silent flight of owls -- Noise is generated by vortices produced when air flows over a bird's wing and larger vortices produce more noise. Wings with small saw-toothed projections (vortex generators), like those on the leading edge of owl wings, generate many small vortices instead of large vortices and produce less aerodynamic noise. In addition, the fringe feathers at the trailing edge of the wing (with fewer hooklets at the ends of the barbs) help to break up the sound waves that are
generated as air flows over the top of their wings and forms downstream wakes, and the soft down feathers located elsewhere on the wings and legs of owls
absorb the remaining sound frequencies above 2,000 hertz and make owls completely
silent to their prey. As a bonus, with high angles of attack and at slow speeds, vortex generators stick out of the stagnant air near
the surface of the wing, and into the freely moving air outside the boundary layer. This surface layer is typically quite thin, but dramatically reduces speed of the airflow towards the rear of the wing. The vortex generators mix the free stream with the stagnant air to get it moving again, providing considerably more airflow at the rear of the wing and helping to prevent stalling. This process is referred to as 're-energizing the boundary layer.'
Unpredictable predators -- The use of space by predators in relation to their prey is a poorly understood aspect of predator-prey interactions. Classic theory suggests that predators should focus their efforts on areas of abundant prey, that is, prey hotspots, whereas game-theoretical models of predator and prey movement suggest that the distribution of predators should match that of their prey's resources. If, however, prey are spatially anchored to one location and these prey have particularly strong antipredator responses that make them difficult to capture with frequent attacks, then predators may be forced to adopt alternative movement strategies to hunt behaviorally responsive prey. Roth and Lima (2007) examined the movement patterns of bird-eating Sharp-shinned Hawks (Accipiter striatus) in an attempt to shed light on hotspot use by predators. Their results suggest that these hawks do not focus on prey hotspots such as bird feeders but instead maintain much spatial and temporal unpredictability in their movements. Hawks seldom revisited the same area, and the few frequently used areas were revisited in a manner consistent with unpredictable returns, giving prey little additional information about risk. But why wouldn't Sharp-shinned Hawks focus their hunting on the areas with the most potential prey (bird feeders)? One possibility is that behaviorally responsive prey diminish the "hotspot" quality of feeders. Although feeder hotspots are sources of abundant prey, the individuals at such feeders generally benefit from group vigilance as a result of these higher densities. As a result, the vulnerability of the prey may actually be lower at feeders than at other locations. In addition, unpredictable movement may reflect a sort of "prey management" by predators, whereby predators spread their hunting activity over multiple areas in an effort to avoid inflating the antipredator behavior of their prey. This hunting strategy may be effective when prey are anchored to high-resource areas such as feeders and use antipredator behaviors, such as high vigilance, that reduce a predator's attack success if it attacks frequently and predictably. Vultures: Obligate avian scavengers Vultures and condors are obligate scavengers that feed on meat as do other predators, but with the difference that they do not kill their prey. As such, vultures and condors need to find animal carcasses that have previously died from different causes including diseases, starvation, accidents, predation, or game hunting. According to phylogenetic DNA analyses, vultures have evolved in three independent evolutionary lineages. The three clades are:
Clade 1: New World vultures (five genera in the family Cathartidae with a total of seven species).
Clade 2: Subfamily Gypaetinae: Three monotypic genera in the Old World with three extant species: Bearded Vulture (Gypaetus barbatus), Egyptian Vulture (Neophron percnopterus), and Palm-nut Vulture (Gypohierax angolensis). The three species occur in Africa, but are not sympatric in their distributions.
Clade 3: Subfamily Aegypiinae with Griffon vultures (genus Gyps with eight species) and close relatives (clade 3b) as well as Cinereous Vultures (Aegypius monachus) and relatives (clade 3a; with four monotypic genera Sarcogyps, Trigonoceps, Aegypius, and Torgos). Griffon Vultures (genus Gyps) share ancestry with Hooded Vultures (Necrosyrtes monachus). All the species are distributed in the Old World. Most vultures are considered obligate scavengers, feeding mostly on medium-size and large carcasses of wild and domestic ungulates. Such carcasses occur wherever large herds of livestock are kept and in the savannahs of Africa that have high densities of antelopes, giraffes, zebras, and buffaloes. In Europe and Asia, livestock and wild ungulates constitute the main source of their diet. Few vulture species prey occasionally on small live animals (e.g. Egyptian Vultures, Palm-nut Vultures, and Bearded Vultures). In general, many vultures are considered polyphagous and food opportunists. Most vultures that feed on large carcasses have extensive areas of bare skin on the head and neck. This can be seen as an adaptation to avoid soiling and sticking of the head feathers when they tear out meat and soft tissues from the inside of a carcass. Vulture skull morphology is linked to their feeding behavior and can predict a vultures feeding strategy, with three main categories:
Rippers: Species with strong beaks and robust skulls, allowing them to tear through tough hides and access muscles (i.e. Cinereous Vultures). Gulpers: Species with elongated skulls and narrow beaks, adaptations that enable them to delve deep into carcasses to consume soft tissues (Griffon Vultures).
Scrappers: Smaller vultures that exhibit slender beaks suited for picking residual bits of flesh from bones (i.e., Hooded Vultures and Egyptian Vultures). Since carrion of large animals is a limited resource that occurs at random and is rare, vultures have had to develop extraordinary sensory and social skills to find their food. Most vultures use social information to find carrion. For example, when Griffon Vultures gather at a carcass, this is often a signal to other species that there is something to eat; they join in to feed on the carrion as well. Only three species of New World vultures of the genus Cathartes (Greater Yellow-headed Vultures, Turkey Vultures, and Lesser Yellow-headed Vultures) can detect carcasses using their sense of smell (From: Wink et al. 2026). Frugivores Bird frugivores are species that primarily or exclusively consume fruit as their main food source. Most frugivorous birds (50%) are passerines (Passeriformes), but a significant number of woodpeckers and allies (Piciformes), parrots (Psittaciformes) and pigeons (Columbiformes) are also frugivorous species (915%). Frugivore richness is highest in the Neotropics, and current climate explains more variance in species richness and proportion of frugivores than of non-frugivores. Neotropical birds that eat fruit come in a variety of body form and sizes. Guans and curassows (Cracidae) are primarily frugivorous and include the largest avian frugivores ranging from chicken to turkey-sized. Many parrots, macaws, and parakeets (Psittaciformes) are also frugivores, and the daily
movements of their flocks in Neotropical landscapes are
often guided by their fruit-tracking behavior. Cotingas (Cotingidae) are probably the most iconic endemic bird family of Neotropical frugivores of varied sizes and spectacular plumages and ornaments that are
highly effective seed dispersers. Because of
the large variety of seeds they disperse, cotingas have been
found to be key elements of bird-plant mutualistic networks. Fleshy fruits provide birds with nutrients such as water,
carbohydrates (sugars), lipids, protein, vitamins, and pigments (antioxidants), with the nutrient composition varying across fruiting species. For example,
most fruits have low
protein content so most fruit-eating birds have mixed diets
that include invertebrates (From: Carlo et al. 2022).
Obligate frugivores: These birds rely almost entirely on fruit year-round. Examples include Hornbills, Fruit-pigeons, Toucans, and Cotingas.
Partial/Facultative frugivores: These species consume fruit as a major part of their diet but supplement it with insects or other food, often depending on the season. Many Thrushes, Bulbuls, and Tanagers fall into this group.
Opportunistic frugivores: These birds eat fruit only occasionally when it is abundant or other food sources are scarce. To thrive on a fruit-heavy diet, frugivorous birds have evolved specialized physical and biological traits:
Morphology: Beak shape and gape size limit the size of fruit a bird can swallow whole.
Digestive systems: Obligate frugivores often have shorter intestines or faster gut-retention times to rapidly process large amounts of fruit pulp while passing seeds quickly.
Color vsion: Frugivores are highly visual and often prefer bright colors like red and black, which contrast sharply with green foliage and indicate ripeness. Kleptoparasitism, or "parasitism by theft," is a specialized foraging strategy where one bird steals food already procured by another. Kleptoparasitism can occur during both intra- and interspecific interactions. This behavior is most prominent in approximately 200 species of birds across 33 families, and particularly among seabirds and raptors.
Specialized Marine Kleptoparasites:
These families are "obligate" or regular kleptoparasites, often relying on theft for a significant portion of their diet. Great Frigatebird (Fregata minor): Regularly targets boobies and terns.
Magnificent Frigatebird (Fregata magnificens): Targets pelicans, gulls, and cormorants.
Lesser Frigatebird (Fregata ariel): Often seen targeting terns. Skuas and Jaegers (Stercorariidae): Highly aggressive "pirates" of the open ocean.
Great Skua (Stercorarius skua): Specifically targets gannets and gulls.
Arctic Jaeger (Stercorarius parasiticus): Frequently steals from terns and puffins.
Pomarine Jaeger (Stercorarius pomarinus): Known to target gulls. Opportunistic Kleptoparasites:
Many species use theft as a secondary tactic when the opportunity arises, such as when food is scarce.
Gulls (Laridae): Famous for stealing from humans, each other, and other species like pelicans or ducks.
Herring Gull (Larus argentatus): Known to steal from oystercatchers.
Black-headed Gull (Chroicocephalus ridibundus): Often targets terns. Charadriiformes: Sheathbills (Chionis) are well known for being highly opportunistic kleptoparasites. Sheathbills use their kleptoparasitic skills primarily to target nesting seabird colonies:
Targeting penguins: They are famous for harassing penguins, especially when parents are attempting to feed their chicks. Sheathbills will fly at or bump into penguins to make it drop or regurgitate krill and fish, which the sheathbill then quickly consumes.
Other victims: They also steal from Imperial Cormorants and various species of petrels and terns. Raptors (Accipitridae & Falconidae): Large birds of prey often harass smaller ones for their kills.
Bald Eagle (Haliaeetus leucocephalus): Frequently harasses Ospreys to drop their fish.
Golden Eagle (Aquila chrysaetos): Noted for stealing from other raptors.
Peregrine Falcon (Falco peregrinus): Although usually hunters, they may steal from or be stolen from by other falcons. Corvids (Corvidae): Highly intelligent birds that use deception and teamwork to steal.
Carrion Crow (Corvus corone): Steals from shorebirds like oystercatchers.
Fork-tailed Drongo (Dicrurus adsimilis): Uses false alarm calls to scare other birds (and meerkats) away from their food.
How Do Brown Pelicans Survive Their Death-Defying Dives?
Caspian Tern diving for fish
Common Kingfishers diving for fish
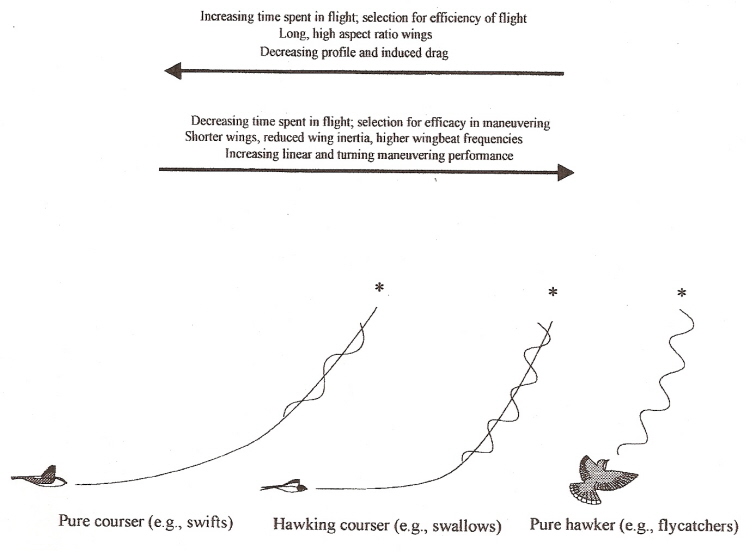
Aerial insectivores -- Swifts depend on flying efficiently and maintaining high speed. Hawking insectivores, like flycatchers, depend on perches
located near prey, but they must be able to accelerate rapidly and be very maneuverable. Swallows combine these two strategies; they are fast,
maneuverable
and able to accelerate when necessary (Warrick 1998).
Watch the Masterful Flight of Tree Swallows
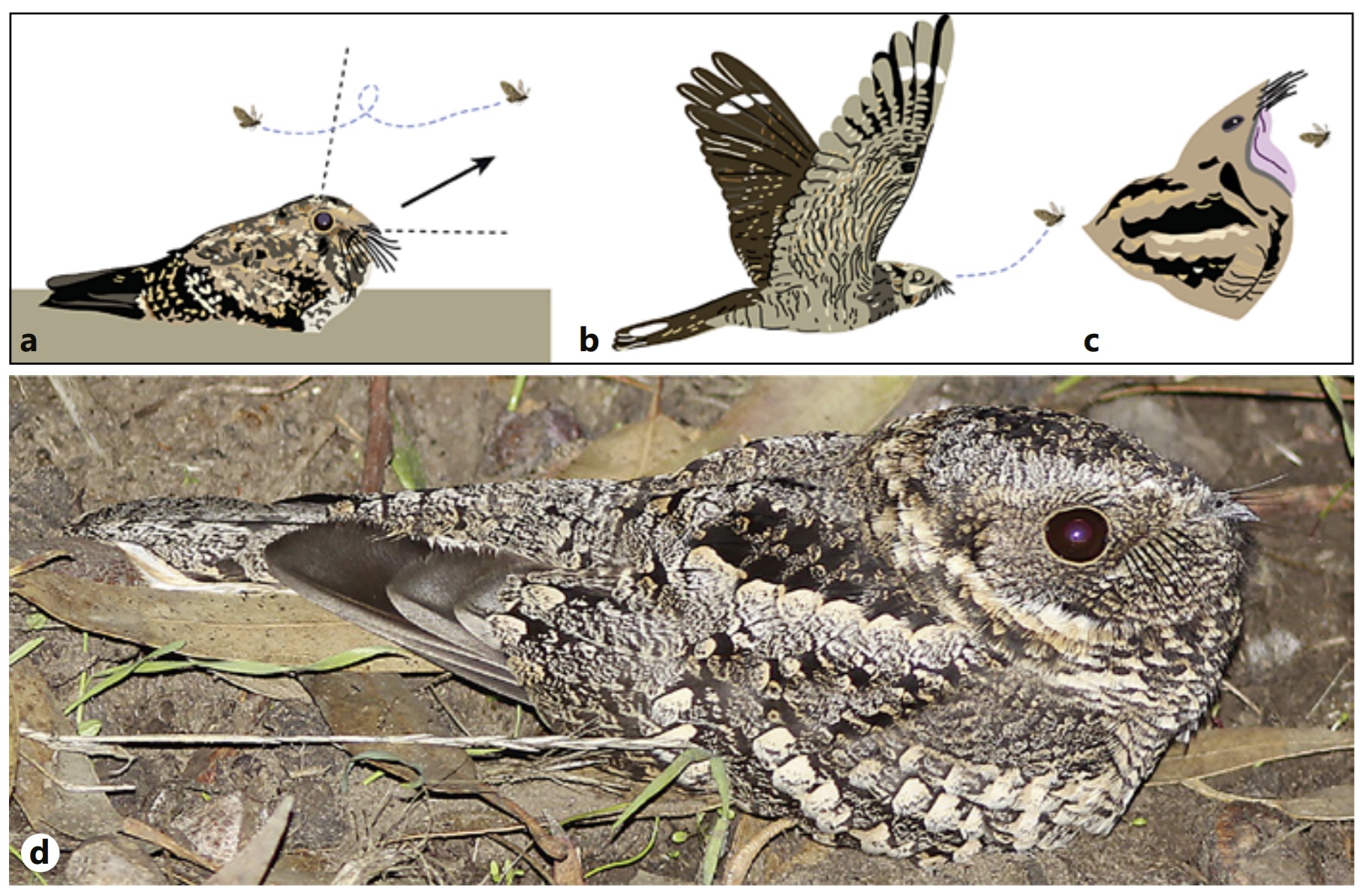
Foraging behavior of nightjars. Among nightjars (Caprimulginae), the most
common foraging strategy is a
sit-and-wait tactic, with a behavioral sequence that has
been described for several nightjar species.
(a) From the ground, nightjars wait
until a prey item is spotted. The aerial pursuit of flying insects
is triggered by their pass over
the dorsal visual field. (b) Attack of prey occurs from below. (c) Final stage
of hunting: the prey is engulfed in the notoriously wide
mouth. Note that when the mouth is open,
the nightjars
eyes no longer face forwards,
but upwards, relative to prey location. (d) A nightjar (Band-winged
Nightjar,
Systellura longirostris) in its usual
cryptic resting position prior to prey detection (From: Salazar et al. 2020).
Bird of Prey, the multi award-winning feature-length documentary from the Cornell Lab of Ornithology, weaves a remarkable story of the world's rarest eagle species and the heroic individuals working tirelessly to save it. Since its release, Bird of Prey has been viewed by audiences around the world and throughout the Philippines where the film has become an invaluable tool for raising awareness and support for conservation of the critically endangered Philippine Eagle.
To learn more about Philippine Eagles and how you can help support their conservation visit:
https://www.philippineeaglefoundation.org.
Harpy Eagle
The Slow Motion Beauty and Skill of an Ospreys Dive


Talons of (left > right): Harpy Eagle, Golden
Eagle,
Bald Eagle,
Great Horned Owl, Red-tailed Hawk, & Peregrine Falcon

Talons of five species of owls. (A) Blakiston's Fish-Owl (fish specialist), (B) Socotra Scops-Owl
(insect specialist), (C) Great Gray Owl (small mammal specialist), (D) Great Horned Owl (generalist
predator), and (E) Snowy Owl (small mammal specialist with highly curved talons). In general, talon
curvature varies with prey type, with small mammal and insect specialists having straighter talons.
Generalists exhibit pronounced talon curvature, and fish specialists have particularly thick and robust
talons. Highly curved talons are likely essential for capturing
and retaining large prey, but can still be used,
though maybe not as
efficiently, to capture small mammals, whereas the reverse might
not be true (From:
Cameron et al. 2023).

Action of the avian digital TLM: Top, digital extension, Bottom, digital flexion. This shows the movement of the talon, flexor and extensor tendons,
and the movement of the ventrally located tubercle pad relative to the stationary plicated sheath and phalangeal bone
(From: Einoder and Richardson 2006).
Accipitriformes (hawks & eagles) and
Strigiformes
(owls) differ in morphology, talon force, & hunting behavior --
Ward et al. (2002) examined the hindlimb morphology of six raptors
(listed
below) to determine if resource partitioning might be explained, at
least
in part, by morphological differences. One difference is that the digit
pattern in Strigiformes is zygodactylous (see photo to the right), a
pattern
that may reduce the chance of prey escaping by maximizing the area of
the
foot & may allow owls to better subdue larger prey than
similar-sized
hawks. The morphology of owls (shorter & wider tarsometatarsus; see
photo below right) also appears to be associated with a stronger grip,
while the hindlimbs of hawks & eagles (relatively long and
gracile)
seem adapted for high-velocity movements.
The force produced by
talons
may be related to time of activity. Owls hunt when light levels are low
so if an attacking owl misses its prey, relocating it may be difficult.
Hawks are diurnal hunters and can use visual cues during and after an
attack.
If unable to subdue prey initially, they can relocate prey visually and
catch it. Given the morphological differences and hunting
behaviors
of these raptors, how well do those characteristics relate to prey-size
selection?
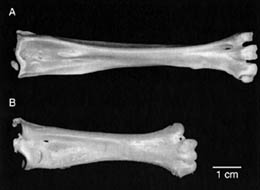
Tarsometatarsi of a similarly-sized hawk & owl.
(A) Red-tailed Hawk. (B) Great Horned Owl.
The incredible hunting techniques of eagles

Barred Owl primary - leading edge below and trailing edge above
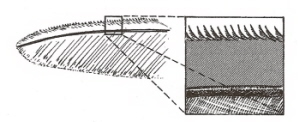
The serrated leading-edge feather of an owl
(Norberg 2002)
.
Eurasian Sparrowhawk

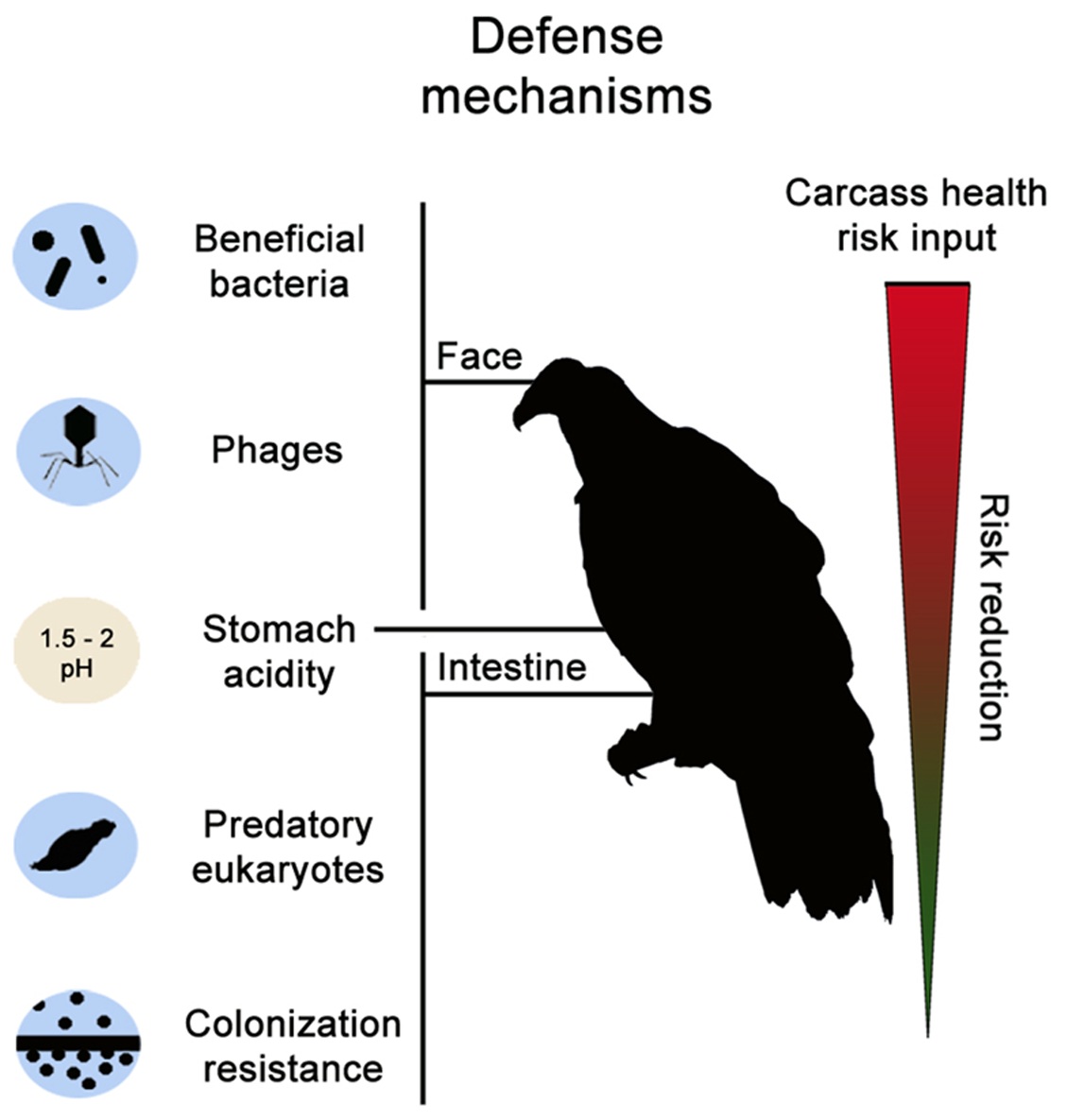
Vultures are exposed to a variety of pathogens, such as those that
cause anthrax, pneumonia, brucellosis, and food poisoning. Factors
that help protect vultures from these pathogens include (1) beneficial
bacteria present on facial skin that prevent colonization by potentially
harmful bacteria (via biosynthesis of antibiotics), (2) phages (viruses
that infect bacteria, also called bacteriophages) on facial skin and in
the digestive system can infect and eliminate potentially pathogenic
bacteria, (3) acid in the stomach kills some (but not all) potential
pathogens, (4) microbial eukaryotes in the intestine may feed on
bacteria (additional study is need to confirm this), and (5) assemblages
of beneficial microbes (biofilms) lining the walls of the intestine
may inhibit colonization by potential pathogens (From: Mendoza
et al. 2018).
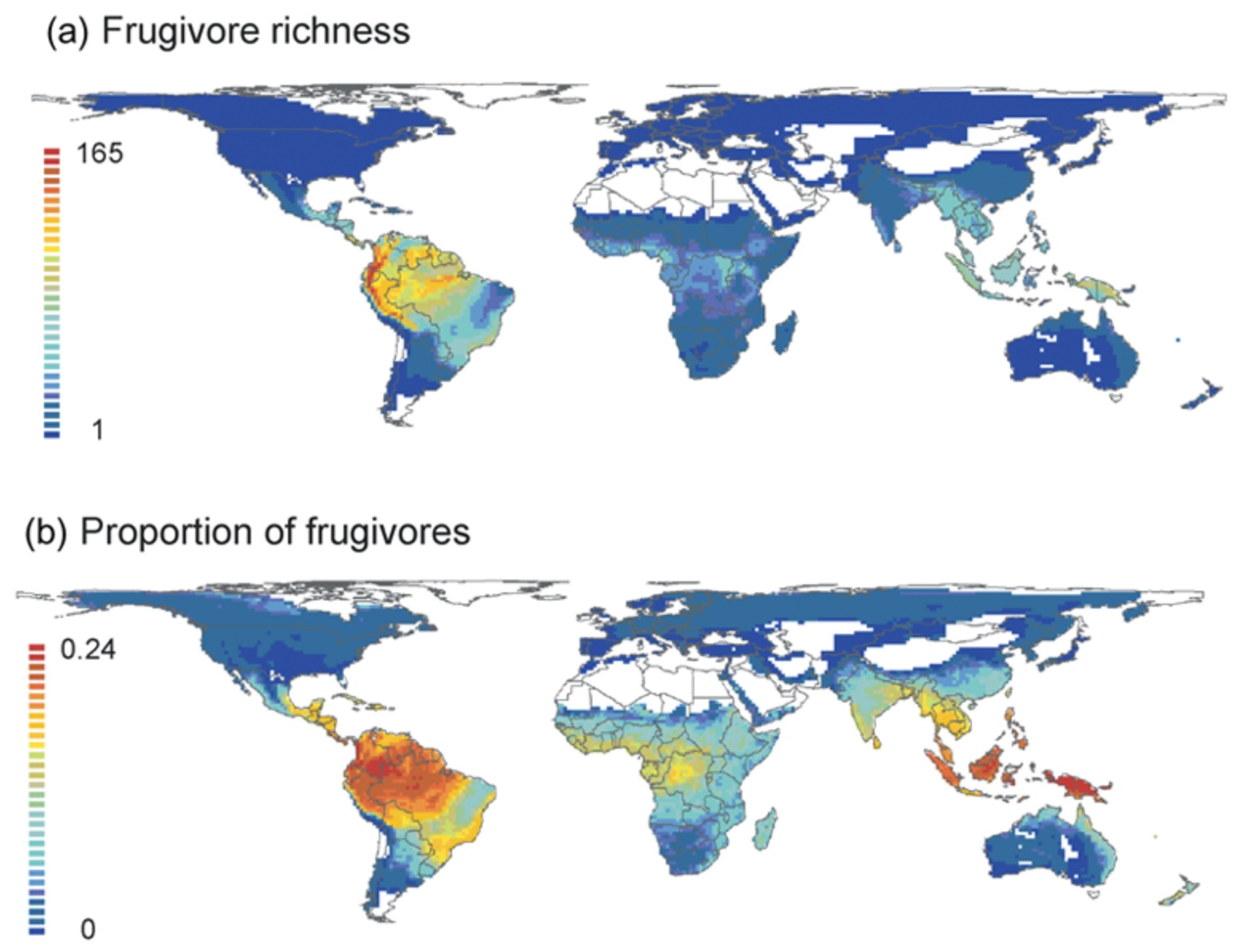
Global geographical patterns of avian frugivory. (a) Species richness of avian frugivores.
Proportion of birds that are frugivorous (Figure from: Kissling et al. 2009).
Birds can be categorized based on how much they rely on fruit:
Kleptoparasitic frigatebirds
Frigatebirds (Fregatidae): Known for chasing other seabirds to force them to regurgitate their catch.
| Bristles are stiff and hairlike,
consisting of
a central rachis without vanes, and provide both protective and
sensory
functions. Bristles occur most prominently around the eyes
("eyelashes"),
the lores, the nostrils, and around the rictus (corners) of the mouth.
Not all birds have bristles. Rictal bristles are prominent in many
insectivorous
birds, particularly aerial insectivores like nightjars (Order
Caprimulgiformes)
and flycatchers (Family Tyrannidae), and may be used as sensory organs
to help locate and capture prey, much like mammals use whiskers. In addition, bristles around the mouth may help protect the eyes from food items a bird is trying to capture (Conover and Miller 1980). The photo to the right shows the rictal
bristles of a Hooded Warbler.
(Source: www.westol.com/~banding/Pictorial_Highlights_080511_2002.htm) |
 |

Seabirds are choking on ocean plastic (video)
;
The avian tongue:
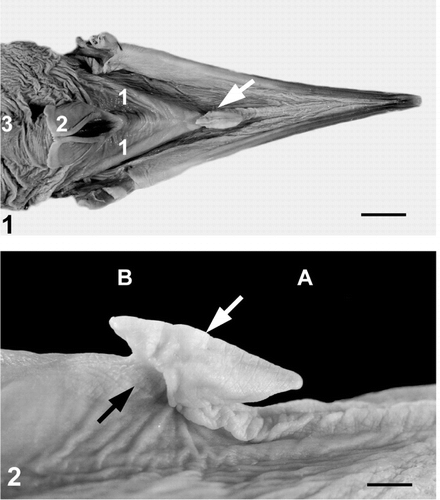 |
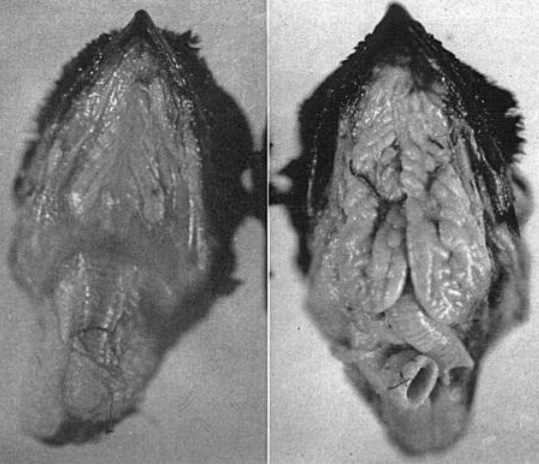
The tongues of cormorants and other fish-eating species are small because these species swallow prey whole and tongues are not needed to manipulate or position food in the oral cavity. Fig. 1. Dorsal view of the surface of the lower bill of a Great Cormorant (Phalacrocorax carbo). Arrow shows the tongue with sharpened tip. 1, hyoid bones; 2, laryngeal sulcus with ostium; 3, esophagus. Scale bar, 12 mm. Fig. 2. Lateral view of the cormorant tongue. The tongue and the small anterior and posterior areas of the mucosa of the bill are covered by white keratinized epithelium. Black arrow shows short base of the tongue. White arrow shows the median crest on the dorsal surface of the tongue. A, anterior; B, posterior. Scale bar, 3 mm (Source: Jackowiak et al. 2006). |

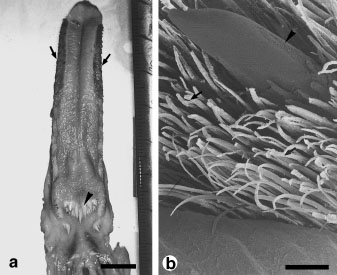
Detailed view of the horny tip (left) of the Guadeloupe Woodpecker tongue in vivo position (Villard and Cuisin 2004).


(a) Skull of Great Spotted Woodpecker, (b) hyoid apparatus of a Great Spotted Woodpecker, and
(c) hyoid apparatus of a non-woodpecker (Eurasian Hoopoe) (From: Wang et al. 2011).
For more detailed information about bird tongues, check this paper: Function-related morphological characteristics and specialized structures of the avian tongue
| Goose tongue -- The dorsal
surface of the tongue of Middendorff's Bean
Goose (Anser fabalis middendorffii) has an anterior region
that
extends for five-sixths of its length plus a posterior region. Large
conical
papillae (indicated by arrowhead to the right) are located in a row
between
the anterior and posterior regions. On both sides of the anterior
region,
lingual papillae are compactly distributed, and small numbers of large
conical papillae are found between the lingual papillae. The dorsal
surface
of the tongue is covered by numerous fine processes, which help hold
food
on the tongue's surface.
The taste buds of birds may be located in the upper beak epithelium, in the anterior mandible, and the mandibular epithelium posterior to the tongue. Some taste buds are also located ventrolaterally on the anterior tongue. -- From: Iswasaki (2002). |
 Surface structure and histology of the dorsal epithelium of the tongue of Middendorff's Bean Goose. (a) Macroscopic dorsal view of the tongue. Arrows show lingual hairs on the lateral sides). (b) Scanning electron micrograph of the lateral side of the tongue. Lingual papillae (arrows) are compactly distributed on the tongue, and large conical papillae (arrowhead) are scattered among them. Scale bars = 10 µm (a) & 500 µm (b). (From: Iwasaki 2002). |



Penguins also have backward-pointing papillae on the roof of their oral cavities (From: Penguins International).
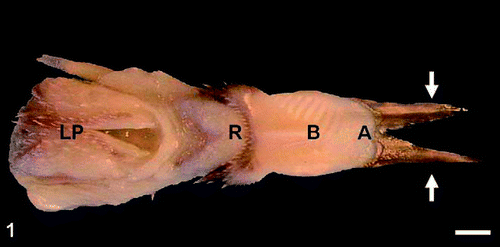 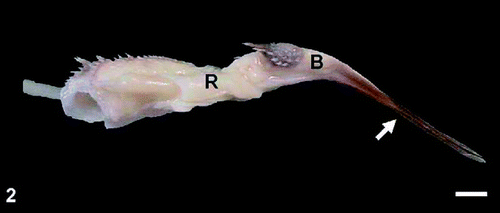 |
Fig. 1. Dorsal view of the tongue of the Spotted Nutcracker (Nucifraga caryocatactes). Arrows show two elongated processes of the apex. A, apex, B, body, R, root, LP, laryngeal prominence. Scale bar, 3 mm. Fig. 2. Lateral view of the tongue of the nutcracker. Arrow shows elongated processes, pointed diagonally, B, body, R, root. Scale bar, 3 mm. |
Nutcracker tongue -- Jackowiak et al. (2010) found that a characteristic feature of the Spotted Nutcracker's tongue is the unique structure of the anterior part of the tongue, which has two long and highly keratinized processes — a product of the renewable keratinized layer of the epithelium covering the ventral surface of the tongue. These dagger-like processes, which are a modified “lingual nail,” help nutcrackers lever up and shell seeds, which are transported over the short sulcus-shaped body of the tongue. Another unique feature of the nutcracker's tongue is the groove separating the body from the root. Two rows of highly keratinized, mechanical, conical papillae are located at the junction of the body and the root. These papillae are mechanically protective elements for passing food particles in the form of seeds. The nutcracker tongue is an efficient tool resembling a lever that is helpful in shelling seeds.
Nutcrackers break or partially break off the outer scales of the husk of the cone with their beak and then strip the remaining parts of the husk with the lower part of the beak, exposing the seeds that adhere to the base of the husk. Using the keratinized processes of the lingual apex, they then lever the seeds out of the claw-like base of the seed wing.

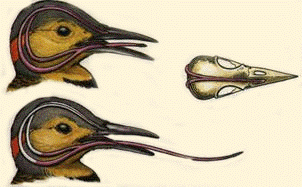
Source: http://www.zoobooks.com/newFrontPage/animals/virtualZoo/animals/h/humingbirds/head2.htm
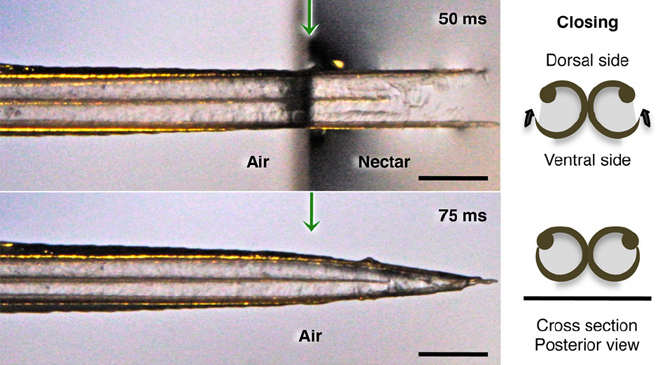
Hummingbird tongues are fluid traps, not capillary tubes -- Hummingbird tongues pick up a liquid, calorie-dense food that cannot be grasped, a physical challenge that has long inspired the study of nectar-transport mechanics. Existing biophysical models predict optimal hummingbird foraging on the basis of equations that assume that fluid rises through the tongue in the same way as through capillary tubes. Rico-Guevara and Rubega (2011) found that hummingbird tongues do not function like a pair of tiny, static tubes drawing up floral nectar via capillary action. Instead, the tongue tip is a dynamic liquid-trapping device that dynamically traps nectar by rapidly changing their shape during feeding. High-speed video observations showed that an entire tongue transformation cycle occurs in as little as 1/20th of a second. In addition, the tongue–fluid interactions are identical in both living and dead birds, demonstrating that this mechanism is a function of the tongue structure itself, and therefore highly efficient because no energy expenditure by the bird is required to drive the opening and closing of the trap. These results rule out previous conclusions from capillarity-based models of nectar feeding and highlight the necessity of developing a new biophysical model for nectar intake in hummingbirds.
Links:
Hummingbird tongue tips twist to trap nectar
How the hummingbird tongue really works (with videos)
|
|
Biomechanical hypothesis for suction feeding by sunbirds. Sagittal sections of the bill and tongue of a feeding sunbird (left) and transverse sections of the bill and tongue during feeding (right). These sections combine anatomical information, hypothetical motions, and deformations inferred from external high-speed video observations. (A) Initial position of the tongue at the start of the first feeding cycle. (B) Tongue extension, with the base pressed dorsally into the ventral surface of the maxilla before ventral motion of the tongue. (C) The tongue continues to move ventrally while the lateral tongue edges are sealed against the upper bill. (D) Partial retraction of the tongue, and the proximal portion of the tongue lowered from the upper bill. The hermetic seal is released, and nectar can move from the proximal end of the tongue to the proximal oral cavity and throat. (E) The tongue has completed a feeding cycle. Some nectar often remains in the tubular portion of the tongue from the previous feeding cycle. (F) During re-extension for the second feeding cycle, the nectar left over from the previous cycle is drawn proximally into the tongue via suction as the bird begins to generate a pressure differential across the length of the tongue before the tongue tips make contact with the nectar reservoir. |
Nectarivory has independently evolved many times among birds, yet little is known about the diversity of
feeding mechanisms
that enable specialized taxa to efficiently collect this energy-rich resource. Multiple
avian groups have converged on evolving
elongated bills and tube-like tongues adapted for nectar extraction. Old World sunbirds (family Nectariniidae) stand out as having
the greatest degree of convergence in
bill and tongue morphology with the well-studied and highly specialized New World
hummingbirds (family
Trochilidae), which fill their tongues via fluid trapping and expansive filling. However, using museum
specimens, high-speed video, and fluid modeling, we show that sunbirds use a unique drinking mechanism not
found in any other
animal: intralingual suction. Using tube-like tongues, sunbirds are able to move nectar
from flowers to their mouths by generating
a pressure differential along the length of the tongue, a remarkable
feat for animals without lips or cheeks (From: Cuban et al. 2026).
Energy and nitrogen balance in a
hummingbird --
Keeping
fit and healthy on a low-fat, fiber-free diet isn't easy, but despite
the
nutritional disadvantages of life on a liquid lunch, hummingbirds
flourish
by supplementing their nectar intake with tiny arthropods. But the
beneficial
snacks come at a high metabolic price; flies don't sit still, so
hummingbirds
work hard chasing their protein. Just how much nitrogen a hummingbird
extracts
from the protein in its  diet,
or the amount of effort needed to gather it, wasn't clear, so
López-Calleja
et al. (2003) began tempting the tiny birds with nitrogen laced nectar
and found that although their protein requirements were relatively
meager,
the tiny creatures' metabolic demands were colossal: 43 kJ day-1! diet,
or the amount of effort needed to gather it, wasn't clear, so
López-Calleja
et al. (2003) began tempting the tiny birds with nitrogen laced nectar
and found that although their protein requirements were relatively
meager,
the tiny creatures' metabolic demands were colossal: 43 kJ day-1!
López-Calleja et al. (2003) trapped almost 40 Green-backed Firecrowns in central Chile, before transporting them to an aviary in Santiago ready to test out their metabolism. Back in the lab, the team prepared nectar solutions with different concentrations of amino acids to see how much protein the birds needed to maintain a stable body weight. By filming the birds as they sipped from feeders, they measured the amount of energy and nitrogen that the birds consumed. To calculate the bird's nitrogen uptake, they also needed to know how much waste nitrogen the birds lost. So, they collected all of the birds' feces, making sure that none dried out, and measured the nitrogen content. Not surprisingly, the birds that were fed small amounts of protein began losing weight quickly, even though they were able to sip as much high-energy nectar as they wanted. However, the birds that were fed 1.82% nitrogen or more, held their weight. López-Calleja et al. (2003) calculated that the tiny aeronauts need at least 10 mg nitrogen per day to maintain a stable body mass, or else they waste away. What does that translate to in terms of flies? López-Calleja et al. (2003) provided the birds with 500 fruit flies to snack on while offering them either an unlimited nectar supply, a restricted nectar intake, or no nectar at all. After five days of access to flies and nectar, the birds were fit and healthy, catching around 150 flies a day, sufficient to supply them with 5% nitrogen. The birds that had a reduced nectar supply also maintained a stable weight, although they went into torpor overnight to conserve energy. But the birds fed flies alone began losing weight, no matter how hard they worked to feed themselves. Fernández, one of the co-authors was surprised that `flies are not a complete food source for hummingbirds'. She suspects that although the flies should supply all of the hummingbirds needs, the birds simply have to work too hard to catch flies to rely on them as their soul food source. -- Kathryn Phillips, Journal of Experimental Biology |
Flush–pursuit foragers use exaggerated
and animated
foraging movements to flush potential insect prey that are then pursued
and captured in flight. The Myioborus
redstarts
comprise 12 species of flush–pursuit warblers found in montane
forests
of the American tropics and subtropics. All members of the genus have
contrasting
black-and-white tail feathers that are exposed by spreading the tail during foraging. Mumme (2002)
examined
plumage pattern and tail-spreading behavior to see how they affected
flush–pursuit
foraging performance of the Slate-throated Redstart (Myioborus
miniatus)
in Costa Rica. Although flycatching was the most common foraging tactic
used by Slate-throated Redstarts, flush–pursuit prey attacks occurred
more
frequently following hops in the spread-tail foraging posture than hops
in more typical warbler-like posture, suggesting that tail-spreading
behavior
assists in startling and flushing potential insect prey. The hypothesis
that the white tail feathers enhance flush–pursuit foraging was tested
by means of a plumage-dyeing experiment. After locating nests, Mumme
(2002)
captured the male and female and assigned one member of each pair to
the
experimental treatment group; its mate served as a control. For
experimental
birds, a permanent marker was used to blacken the white tips of the
three
outer retrices. For sham-darkened controls,the naturally black tips of
the three inner retrices were also ‘‘blackened.’’ Experimental birds
with
darkened tail feathers were significantly less successful in
flush–pursuit
foraging, showed a significantly lower overall rate of prey attack, and
fed their nestlings at a significantly lower rate than did their
sham-darkened
mates. For experimental birds, only 7.6% of hops in the spread-tail
posture
were followed by an attack on a prey item, compared to 20.9% of hops
for
controls. These results indicate that white tail feathers are
critically
important in startling potential prey. that are exposed by spreading the tail during foraging. Mumme (2002)
examined
plumage pattern and tail-spreading behavior to see how they affected
flush–pursuit
foraging performance of the Slate-throated Redstart (Myioborus
miniatus)
in Costa Rica. Although flycatching was the most common foraging tactic
used by Slate-throated Redstarts, flush–pursuit prey attacks occurred
more
frequently following hops in the spread-tail foraging posture than hops
in more typical warbler-like posture, suggesting that tail-spreading
behavior
assists in startling and flushing potential insect prey. The hypothesis
that the white tail feathers enhance flush–pursuit foraging was tested
by means of a plumage-dyeing experiment. After locating nests, Mumme
(2002)
captured the male and female and assigned one member of each pair to
the
experimental treatment group; its mate served as a control. For
experimental
birds, a permanent marker was used to blacken the white tips of the
three
outer retrices. For sham-darkened controls,the naturally black tips of
the three inner retrices were also ‘‘blackened.’’ Experimental birds
with
darkened tail feathers were significantly less successful in
flush–pursuit
foraging, showed a significantly lower overall rate of prey attack, and
fed their nestlings at a significantly lower rate than did their
sham-darkened
mates. For experimental birds, only 7.6% of hops in the spread-tail
posture
were followed by an attack on a prey item, compared to 20.9% of hops
for
controls. These results indicate that white tail feathers are
critically
important in startling potential prey. |
American Redstart
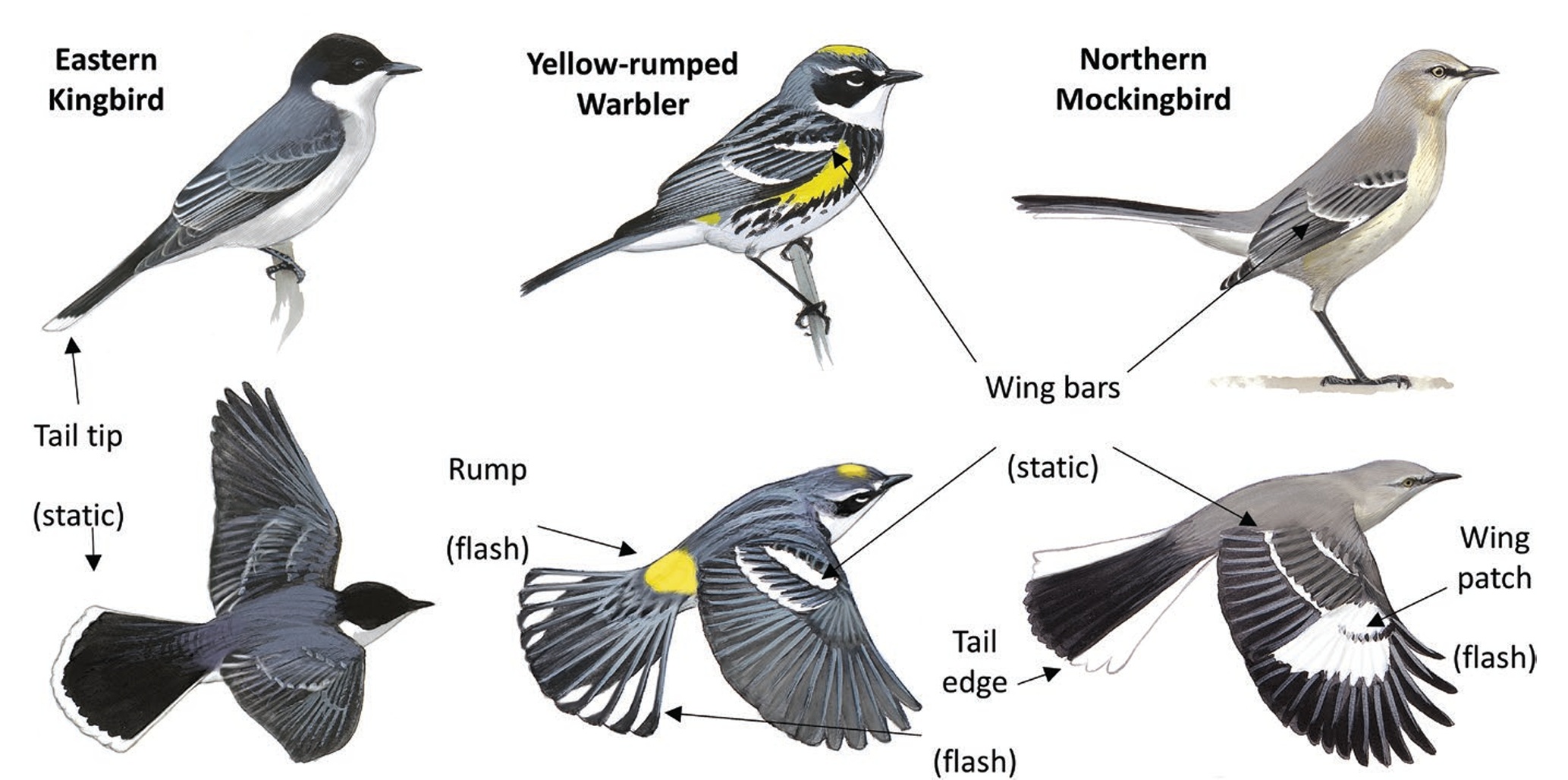
Examples of static (tail tip, wing bar) and flash (tail edge, rump, wing patch) plumage patches on 3 species of North American passerines.
Close encounters with possible prey . . . counterfeit predators -- You are a 12-gram, insectivorous, tropical rainforest bird, foraging in shady, tangled, dappled, rustling foliage where edible caterpillars and other insects are likely to shelter. You want to live 10–20 years. You are peering under leaves, poking into rolled ones, searching around stems, exploring bark crevices and other insect hiding places. Abruptly an eye appears, 1–5 cm from your bill. The eye or a portion of it is half seen, obstructed, shadowed, partly out of focus, more or less round, multicolored, and perhaps moving. If you pause a millisecond to ask whether that eye belongs to acceptable prey or to a predator, you are likely to be—and it takes only once—someone’s breakfast. Your innate reaction to the eye must be instant flight, that is, a “startle” coupled with distancing. The bird that must learn to avoid what appears to be a predator’s eye is not long for this world. Now, a safe few meters away, are you going to go back to see whether that was food? No. You, like billions of other individuals and hundreds of other species for millions of years, have just been a player in an act of natural selection favoring mutations that lead to the multitudes of “false eye” color patterns, “eyespot” patterns, or “facsimiles of eyes” and “faces” adorning tropical caterpillars and pupae. Associated body patterns often suggest other head and facial features, which in turn enhance the eye-like nature of the spots. None of these patterns exactly matches the eyes or face of any particular species of predator; but, even when quickly and partially glimpsed, all give the illusion of an eye or face. These false eyes are mimicking the eyes and faces of such predators of insect-eating birds as snakes, lizards, other birds, and small mammals, as perceived at close range by the insectivorous birds in their natural world. These color patterns, and the birds’ reactions to them, are the evolutionary footprints of predator/prey encounters as shallow as today and as deep as the first terrestrial vertebrate eyes - From: Janzen et al. (2010). |
 Representative caterpillar false eyes and faces |
Buccal, or oral, cavity:

Source: http://animal.discovery.com/fansites/jeffcorwin/carnival/flyingace/swiftlet.html
Esophagus:
Anhinga swallowing a large fish
Note the distended throat of this Peregrine Falcon
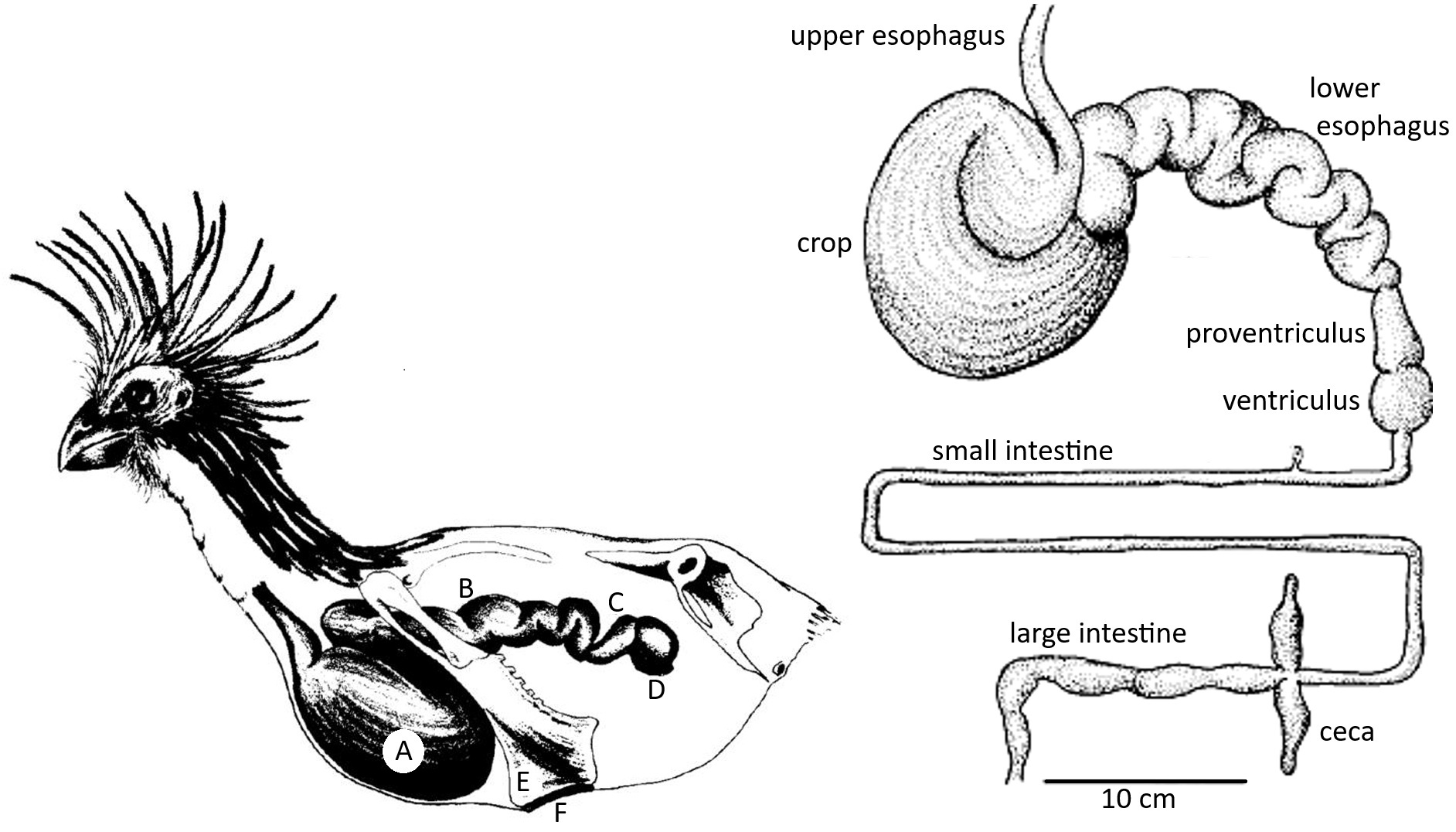
Schematic representations of the digestive system of an adult Hoatzin. Left: Anterior
portion of the digestive system showing the (A) crop, (B) posterior esophagus, (C) proventriculus, and (D) gizzard.
The anterior sternum is reduced in size to make room for large fermentation chambers in the crop, resulting in a reduction in area
available for flight-muscle attachment to (E) sternal keel, (F) "resting" pad at bottom of the sternum used while perching with full crop. Right: Drawing
the Hoatzin digestive system (From: Grajal 1995).
Hoatzins are the only bird with a fully-functional foregut fermentation system where the crop serves as the major site of microbial fermentation. The large crop and posterior esophagus have become functional fermentation chambers and also have a storage function. The large crop and lower esophagus represent 77% of the total gut capacity. The crop is composed of two interconnected chambers and the lower esophagus is a multichambered organ. The two-crop chambers are connected through a constricted zone with circular muscles. The interior linings of the crop and esophagus have longitudinal ridges covered by cornified epithelium. A combination of abrasion and microbial action effectively reduce particle size to aid in digestion (From: Grajal 1995).
Hoatzin's specialized crop
The non-‘lactating’ pigeon crop (A) has a completely different appearance from that of the
‘lactating’ crop (B). The lactating crop is
more than
twice the size of the non- ‘lactating’ crop, with a thickened wall and two very obvious lateral lobes. When the
‘lactating’ crop is
opened (C) the
pigeon ‘milk’ is seen as a bed of close-packed discrete
rice-shaped pellets that are closely associated with the mucosal surface
of the
tissue (From: Gillespie et al. 2011).
Crop 'milk' -- Both male and female Rock Pigeons (Columba livia) produce pigeon ‘milk’ that is fed to their young. Pigeons generally lay two eggs one day apart, which hatch 18 days after they are laid. Two days before the first egg hatches, pigeon ‘milk’ begins to be produced in the crop of the parent birds. A similar substance is produced by flamingos and male Emperor Penguins. The normal function of the crop is food storage. During the process of pigeon ‘lactation’, a curdlike substance is regurgitated from the crop and fed to young pigeons. This 'milk’ is 60% protein and the remainder is mostly fat (32-36%) with a small amount of carbohydrate (1-3%), in addition to the mineral (calcium, potassium, sodium, and phosphorus) content. Pigeon 'milk' also contains IgA antibodies and antioxidants (carotenoids).
Owls
set beetle trap with dung - Levey
et
al. (2004) compared what Burrowing Owls ate when there was a typical
litter
of dung at the entrances to their nest burrows with their diet when the
dung was removed. The owls ate 10 times more beetles when the dung was present, suggesting the waste did not build up by accident.
Burrowing
Owls make their nests in small tunnels, and place a variety of debris,
including dung, at the entrance. After finding that Burrowing
Owls
also had a high concentration of dung beetles in their diet, Levey et
al.
(2004) proposed that the owls might be using dung as bait to attract
the
beetles. To test this hypothesis, they cleared all nest entrances at
two
colonies of owls of debris, then one owl colony had a typical littering
of dung applied while the other was left bare. After four days each
entrance
was again completely cleared and the situation was reversed. Analysis
of
the owls' waste clearly showed that when dung was present, the owls
feasted
on ten times more dung beetles. As Levey says, "this experiment
demonstrates
that tool use makes a difference to a wild animal". Although it may be
tempting to conclude the owls are clever enough to devise this trap,
Levey
explained: "I don't believe these burrowing animals are aware of the
link
between the dung they bring in and the beetles they catch". Instead,
the
baiting may simply have evolved, as owls who happened to collect more
dung
had a better diet and therefore bred more successfully. -- Peter
Wood, BBC News Online dung was present, suggesting the waste did not build up by accident.
Burrowing
Owls make their nests in small tunnels, and place a variety of debris,
including dung, at the entrance. After finding that Burrowing
Owls
also had a high concentration of dung beetles in their diet, Levey et
al.
(2004) proposed that the owls might be using dung as bait to attract
the
beetles. To test this hypothesis, they cleared all nest entrances at
two
colonies of owls of debris, then one owl colony had a typical littering
of dung applied while the other was left bare. After four days each
entrance
was again completely cleared and the situation was reversed. Analysis
of
the owls' waste clearly showed that when dung was present, the owls
feasted
on ten times more dung beetles. As Levey says, "this experiment
demonstrates
that tool use makes a difference to a wild animal". Although it may be
tempting to conclude the owls are clever enough to devise this trap,
Levey
explained: "I don't believe these burrowing animals are aware of the
link
between the dung they bring in and the beetles they catch". Instead,
the
baiting may simply have evolved, as owls who happened to collect more
dung
had a better diet and therefore bred more successfully. -- Peter
Wood, BBC News Online |
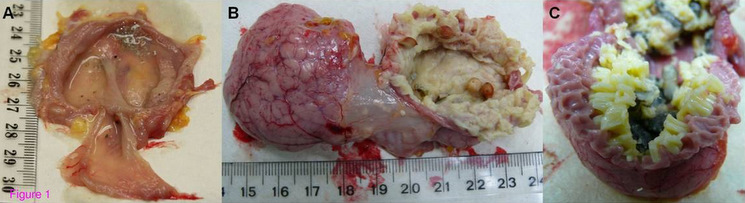
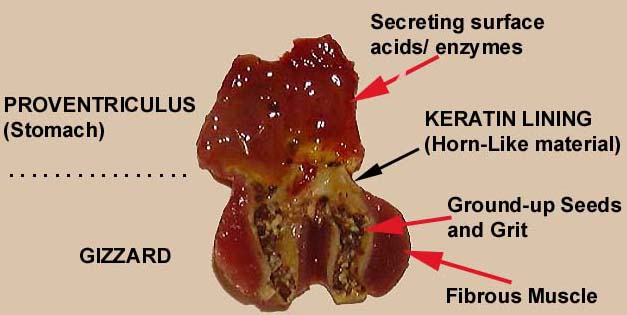
The avian stomach is divided into 2 parts:
Photomicrograph (50X) of a cross section through the proventriculus showing folds of mucous membrane (P);
deep proventricular glands (GP); capsule (connective tissue) around the glands (arrow head); muscle layer (m); serosa
(connective tissue) with blood vessels (S), and the lumen (L) (From: Catroxo et al. 1997).
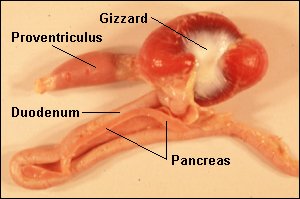
Source: http://arbl.cvmbs.colostate.edu/hbooks/pathphys/digestion/herbivores/birds.html
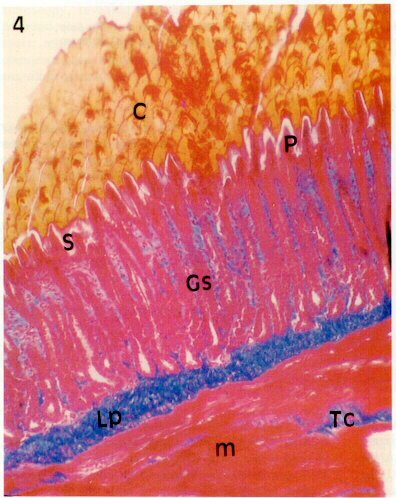
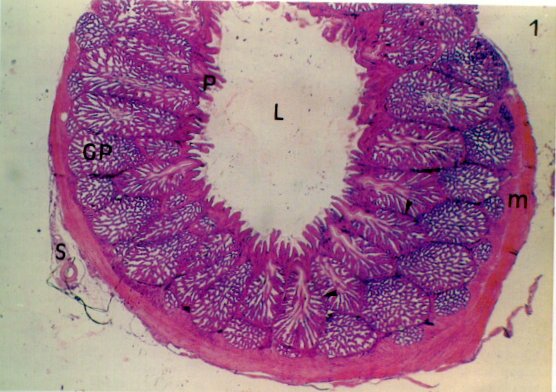
Photomicrograph (210X) of longitudinal section of the gizzard showing folds of mucous membrane lined by simple
prismatic epithelium (P); simple tubular glands (Gs) in the lamina propria constituted by connective tissue (Lp);
secretion of glands (S) that are
continuous with the cuticle (or koilin); (C), part of muscle layer (m), interpersed with bundles of
connective tissue (Tc) (From: Catroxo et al. 1997).
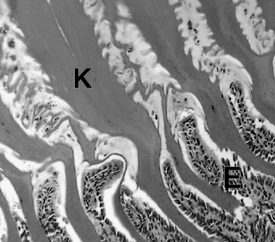
Photomicrograph (400X) of the koilin of an Eclectus Parrot (Eclectus roratus).
Note the regular, columnated structure of the koilin layer (K) and its
association with the
glandular epithelium (E) of the ventriculus (From: De Voe et al. 2003).
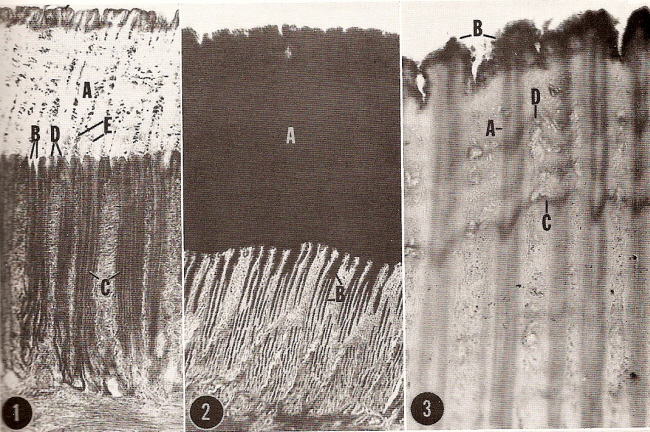
(1) Section through inner lining of a chicken gizzard. A, koilin, B, crypts, C, glands that secrete koilin, D, epithelial surface, E, desquamated epithelial cells,
(2) Mucosa of the gizzard. A, koilin, B, secretion in gland lumens and crypts, and (3) Koilin layer. A, secretion column, B, koilin-layer surface,
C, horizontal stripe indicating a 'pause' in secretion of the koilin, D, cellular debris. (From: Eglitis and Knouff 1962).
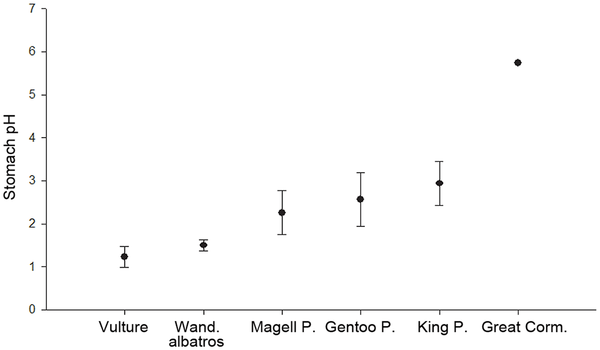
Stomach pH (mean and standard deviation) of White-backed Griffon Vultures, Wandering Albatrosses, Magellanic and Gentoo penguins
(Spheniscus magellanicus and Pygoscelis papua, King Penguins (Aptenodytes patagonicus), and Great Cormorants (Phalacrocorax carbo).
Vultures of the seas -- Animals are primarily limited by their capacity to acquire food, yet digestive performance also conditions energy acquisition, and ultimately fitness. Optimal foraging theory predicts that organisms feeding on patchy resources should maximize their food loads within each patch, and should digest these loads quickly to minimize travelling costs between food patches. Grémillet et al. (2012) tested the prediction of high digestive performance in Wandering Albatrosses (Diomedea exulans) that can ingest prey of up to 3 kg, and feed on highly dispersed food resources across the southern ocean. GPS-tracking of 40 Wandering Albatrosses from the Crozet archipelago during the incubation phase confirmed foraging movements of between 475–4705 km, giving the birds access to a variety of prey, including fishery wastes. Using miniaturized, autonomous data recorders placed in the stomach of three birds, the first-ever measurements of gastric pH and temperature in procellariformes were obtained. These revealed surprisingly low pH levels (mean = 1.50 ± 0.13), markedly lower than in other seabirds, and comparable to those of vultures that feed on carrion. Such low stomach pH gives Wandering Albatrosses a strategic advantage because it allows a rapid chemical breakdown of ingested food and rapid digestion. This is useful for feeding on patchy, natural prey, but also on fishery wastes, which might be an important additional food resource for Wandering Albatrosses. It is likely that this physiological characteristic evolved as a response to a diet largely composed of squid, and to a patchy distribution of this food resource resulting in large, infrequent meals. The strategy of Wandering Albatrosses is to cover long distances rapidly and at low costs to increase the probability of encountering dispersed prey patches whose distribution is unpredictable.
| A Price Worth Paying -- Birds don't
need teeth
to grind their food; they solve the mashing problem with a powerful
gizzard.
But not all gizzards are equal. In fact, Red Knots' gizzards grow
larger
when the birds put on weight preparing for migration. But they also
change
size throughout the year. What causes such changes in gizzard size?
van Gils et al. (2003) served knots that had large and small gizzards (as determined by ultrasonography) a selection of hard intact molluscs and soft mollusc meat and filmed the birds as they ate. Knots with large gizzards consumed far more molluscs with shells than the birds with smaller gizzards. van Gils et al. (2003) also offered the birds a shell-heavy diet, but even the birds with the largest gizzards needed to feed for 16 hours a day to sustain their weight! Birds with smaller gizzards simply couldn't feed fast enough. By allowing them to crush more shell per gizzard-full, larger gizzards gave birds the edge. Thus, even though it is energetically costly for the knots to maintain a larger gizzard, when the bird needs to get the most out of its crunchy diet, it's a price worth paying. So, the birds' gizzards enlarge as they fatten for migration. van Gils et al. (2003) also found the knot's gizzards enlarged when the molluscs begin shrivelling (as their winter food supply dwindles). Because the molluscs' shells stay the same size as the molluscs shrink, the amount of shell a bird must process to eat its fill also increases. But with their larger gizzards, the birds can still make the most of even the crunchiest winter diet! -- Kathryn Phillips, Journal of Experimental Biology |

|
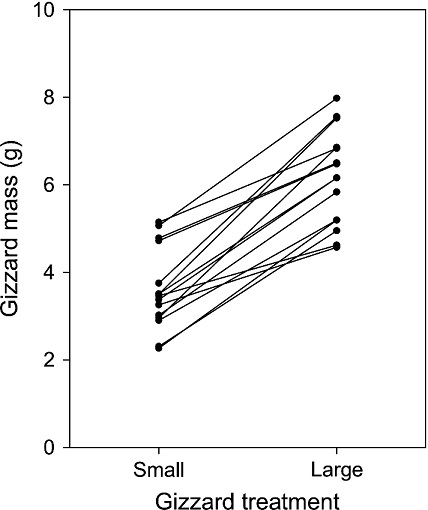
Effect of three-week long gizzard mass treatment on gizzard mass at the start of diet choice experiments. Lines connect estimated gizzard mass
from the same individual Red Knots in the two treatment categories. During the small gizzard treatment, birds received a high digestive quality diet of the flesh from open mussels,
and during the large gizzard treatment birds received a low digestive quality diet of gastropod mudsnails, Hydrobia ulvae, that required processing large shell volumes in the gizzard (Mathot et al. 2017)..
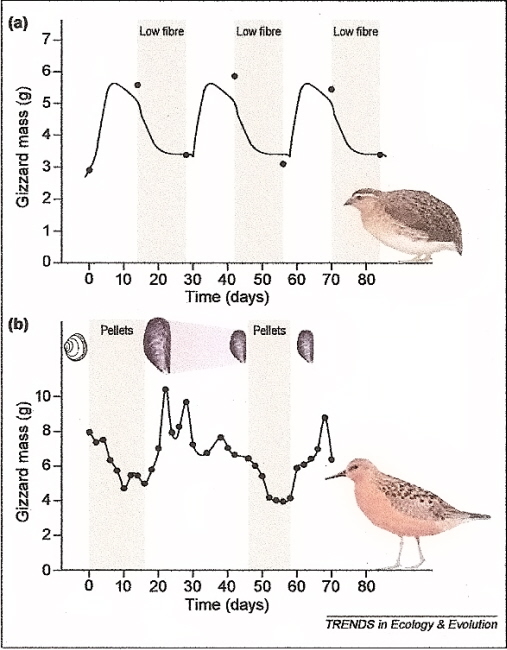
Reversible size changes in the gizzards of adult Japanese Quail (Coturnix japonica) (a) and Red Knots (Calidris canutus) (b).
Quail were given a diet of alternatively low or high non-digestible fiber content (3% vs. 45%). Within 14 days, they showed a doubling
of the size of their gizzards. Red Knots have strong muscular gizzards for feeding on molluscs. With a change in diet from medium-small
mussels (Mytilus edulis) to a diet of soft food pellets, gizzard mass was reduced by almost 50% in about 8 days. A shift back to a mussel diet
induced about a doubling in gizzard mass in just a few days. As the knots were fed progessively smaller mussels (day 22 to day 46) that are easier
to crush, gizzard mass again declined. A switch back to a soft food pellet diet caused a further decline in gizzard mass. Finally, a switch back to a
mussel diet again cause a rapid increase in gizzard mass (From: Piersma and Drent 2003).
Canary stomach
Source: http://members.madasafish.com/~grahamwhite/download/gizzard.htm

Ostrich (Struthio camelus) stomach. Note how particle size of material in the gizzard (ventriculus) is smaller than in the proventriculus due to the grinding action of the muscular walls plus small pebbles (gastroliths). The capacity to reduce particle size is related to the metabolic demands of a species. Therefore, particle size reduction is often considered the key digestive difference between ecto- and endotherms that allows endotherms to rely on shorter digesta retention times without losing digestive efficiency, and hence facilitate the high level of food intake necessary to meet their increased metabolic requirements. Oes, esophagus; Prov, proventriculus; Gizz, gizzard; SI, small intestine (From: Fritz et al. 2011).
Gizzards vs. teeth -- A gizzard system is advantageous when the organismal design demands that the site of particle size reduction be close to the center of gravity; as such, the use of a gizzard is usually linked to the primary characteristic of herbivorous birds, flight. In contrast, adaptations for chewing intrinsically increase the weight of the head. The use of the gizzard system has the potential advantages that intake rate is not limited by chewing, that no investment in dental tissue is necessary, and that dental wear is not a determinant of senescence as observed in mammals. The absence of age-dependent tooth wear might even be a contributing factor to the slower onset of senescence in birds as compared to mammals. On the other hand, the use of a gizzard requires the intake of suitable grit or stones—an action that represents, in the few studies where this has actually been quantified in birds, a relevant proportion of feeding time (Fritz et al. 2011).
| Long-term preservation of stomach contents in incubating King Penguins -- Male King Penguins (Aptenodytes patagonicus) are able to store undigested food in their stomach for up to 3 weeks during their incubation fast. Such an adaptation ensures hatchling survival if their mate's return is delayed. Using small electronic recorders, Thouzeau et al. (2004) studied the change in gastric pH, motility and temperature during the first week of food storage. The pH could be maintained at values as high as 6 throughout the incubation fast, a pH unfavorable for avian gastric proteinase activity. Gastric motility was markedly reduced for most of the incubating birds, with lower motility probably associated with a better conservation of stomach content. Stomach temperature was maintained at around 38°C. The fact that stomach temperature of incubating birds did not show a daily rhythmic fluctuation as seen in non-breeding birds could be due to temperature constraints on embryo development. Thus, this study demonstrates substantial adjustments of pH and gastric motility in incubating King Penguins, which may contribute to the inhibition of digestive gastric processes. Mechanisms underlying these adjustments are probably complex, including a combination of neuronal, humoral, and/or hormonal factors. |  |
Penguins fast with food in the stomach
Intestine:
Small intestine:

Gastrointestinal tracts of a carnivorous hawk, an
omnivorous
chicken, and 4 herbivorous birds.
Note the larger size of the crops in omnivores and herbivores,
and particularly in the Hoatzin. Ceca are small in hawks and
relatively large in grouse. Although ceca are
relatively
small in Hoatzins, Emus, and Ostriches, an expanded foregut
(Hoatzins), a much longer midgut (Emus), or a much longer
colon (Ostriches) compensates for this (From: Stevens and Hume 1998).
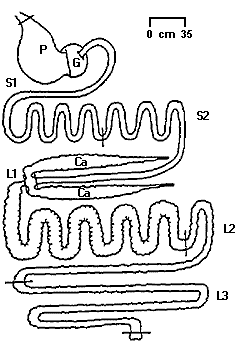
GI tract of an Ostrich chick
(P = proventriculus; G = gizzard; S1 = small intestine;
Ca = caeca; L1, L2, & L3 = sections of
large intestine)
(Source: http://www.ianr.unl.edu/pubs/animals/nf251.htm)
Glucose transport in birds -- In
contrast with
regulation of intestinal glucose transport in mammals, amphibians and
fish,
intestinal glucose transport does not change with dietary carbohydrate
in most birds. This is interesting, because the diets of many birds
change
with seasons, and the levels of carbohydrate in those diets also vary
with
season. Nevertheless, intestinal glucose transport rates do not vary
with
dietary carbohydrate levels in American Robins, House Sparrows, and
Yellow-rumped 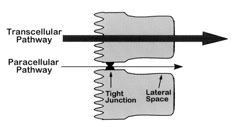 Warblers.
The absence of dietary modulation of glucose transport in birds may be
due to the predominance of passive glucose transport, probably
occurring
through the paracellular pathway (i.e., between cells rather than
through
cells via active transport). If transport were largely passive and
dependent
on transepithelial concentration gradients, then there would not be any
need for specific changes in carrier-mediated (active) transport. For
example,
passive absorption of nutrients such as fat-soluble vitamins is not
subject
to modulation by diet. Warblers.
The absence of dietary modulation of glucose transport in birds may be
due to the predominance of passive glucose transport, probably
occurring
through the paracellular pathway (i.e., between cells rather than
through
cells via active transport). If transport were largely passive and
dependent
on transepithelial concentration gradients, then there would not be any
need for specific changes in carrier-mediated (active) transport. For
example,
passive absorption of nutrients such as fat-soluble vitamins is not
subject
to modulation by diet.
Over-reliance on the passive pathway provides metabolic advantages and ecological constraints. It does provide birds with an absorptive process that can deal with rapid and large changes in intestinal sugar concentrations. The passive pathway is also energetically inexpensive to maintain and modulate. However, passive absorption through the paracellular pathway is dependent on concentration gradients. In the absence of a transport system that selects which materials to absorb, this non-discriminatory pathway may also increase vulnerability to toxins, and thus constrain foraging behavior and limit the breadth of the dietary niche of the birds. Another problem is that when luminal sugar concentrations are lower than those in plasma, glucose may diffuse back into the lumen. -- Source: Ferraris (2001). |
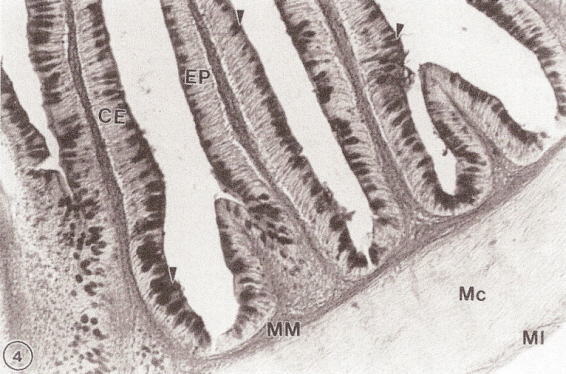
Cross-section of the intestine (ileum) of a Spotted Tinamou (Nothura maculosa).
Villi are lined with columnar epithelium (EP), including goblet cells (arrows) that secrete mucus. The muscle
layer includes longitudinal fibers (MI) on the perimeter, circular fibers (Mc), and additional longitudinal fibers
at the base of the villi
(muscularis muscosae; MM) (From: Chikilian and de Speroni 1996).
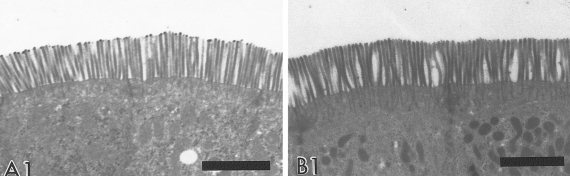
Intestinal microvilli ('brush border') of a (A) House Sparrow and (B) Savannah Sparrow. Scale bar = 0.5 µm
(From: Casotti 2001).
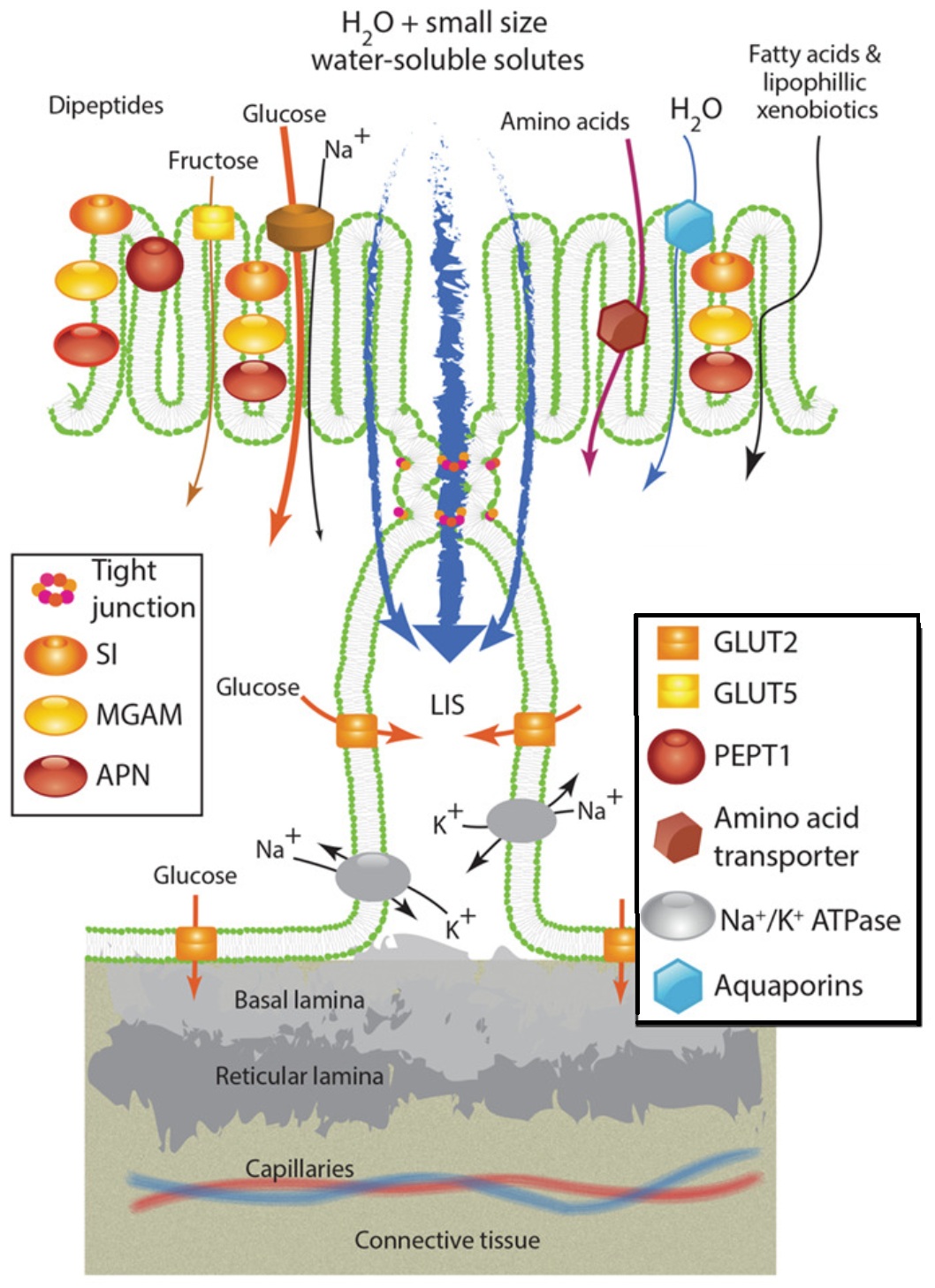
Absorption of materials across intestinal villi. The small intestinal epithelium plays a role in the absorption
of nutrients through transcellular (nutrients passing through the cells) transport, facilitated and active diffusion
(nutrients passing through the membrane via transport proteins), and paracellular transport (nutrients passing
through the tight junction between cells). Ions, solutes, and water, as well as glucose, are absorbed via tight
junctions. SI, MGAM, and APN are enzymes that break down larger molecules into smaller ones, e.g., lipases (break down
lipids into fatty acids/glycerol), proteases (proteins to amino acids), and glycosidases (carbohydrates), that can be
absorbed by tranport proteins. GLUT2 and GLUT5 are glucose transporters that facilitate the transport of glucose across
the plasma membrane via a process called facilitated diffusion. PEPT1 transports proteins across the cell membranes. Amino acid
transporter tranports amino acids across the cell membrane. Na+/K+ ATPase is an active transport mechanism for transporting sodium
and potassium ions into and out of cells. Aquaporins aid in the transport of water into and out of cells (Figure from Karasova and Caviedes-Vidal 2021).
Diagram illustrating some of the functions of the Ostrich small intestine. Intestinal digestion and nutrient absorption
in Ostriches are mediated by villi, microvilli, and pancreatic enzymes found within the intestinal lumen. Mucus is a
critical barrier within which is found abundant commensal microbiota. More generally, the intestine is the key
organ for digestion and absorption. Additionally, it can metabolize toxic substances and eliminate them from the body.
The intestines of birds are typically shorter than those of mammals of the same body volume, which is thought to be
an adaptation to flight evolution. As a result, birds have shorter food retention times in the digestive tract, and their
fiber digestive efficiency is typically lower than that of herbivorous mammals. To compensate for this low digestion
efficiency, birds usually have higher food intake and food absorption efficiency. Notably, the digestive system of
Ostriches differs from that of most other birds. Ostriches have well-developed intestines with longer food retention
times, and their fiber digestion efficiency is comparable to that of herbivorous mammals, distinguishing them from
other species of birds (Modifed from: Zhang et al. 2026).
Large intestine:
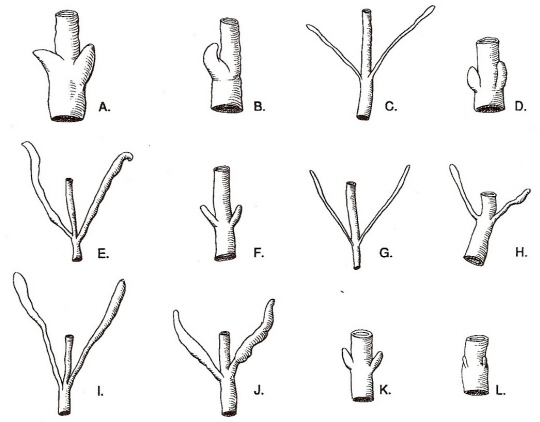
Avian ceca. (A) Little Cormorant, Phalacrocorax niger, (B) Cattle Egret, Bubulcus ibis, (C) Cotton Teal, Nettapus coromandelianus,
(D) Crested Serpent Eagle, Spilornis cheela, (E) Common Quail, Coturnix coturnix, (F) Indian Ring Dove, Streptopelia decaocto,
(G) Red-wattled Lapwing, Vanellus indicus, (H) Koel, Eudynamys scolopacea, (I) Spotted Owlet, Athene brama, (J) Indian Roller, Coracias benghalensis,
(K) Eastern Skylark, Alauda gulgula, & (L) Grey Wagtail, Motacilla caspica (From: Clench 1999).
| Avian geophagy -- In birds, geophagy (the intentional consumption of soil) is known for geese, parrots, cockatoos, pigeons, cracids, passeriforms, hornbills, & cassuaries. Brightsmith and Muñoz-Najar (2004) observed ten species of psittacids, three species of columbids, and two species of cracids consuming soil from banks of a river in Peru. They found that preferred soils were deficient in particles large enough to aid in the mechanical breakdown of food and help digestion. Percent clay content and cation exchange capacity (CEC), both predicted to correlate with adsorption of toxins, did not differ between used and unused sites as had been found in a similar study. Instead, preferred soils were more saline and had higher concentrations of exchangeable sodium. This suggests that the choice of soils at their study site was based primarily on sodium content. Experimental evidence has shown that soils are capable of adsorbing biologically relevant quantities of toxins in vitro and that soil consumption by parrots does reduce the absorption of toxins in vivo. Brightsmith and Muñoz-Najar (2004) did not find evidence that parrots choose soils with greater CEC or clay content, the characteristics that correlate with the capacity to adsorb toxins. Instead, they found that birds chose soils with higher concentrations of sodium. These two findings are not mutually exclusive but instead suggest that there may be a set of conditional rules for soil selection. In situations in which sodium concentrations are variable, the birds appear to choose soils that are highest in sodium (this study). In areas in which sodium concentrations are uniformly high, birds may choose the soils that have the largest ability to adsorb dietary toxins. |
 www.pbs.org/wnet/nature/realmacaw/photoessay6.html
|
Macaws at a clay lick
| Why are bird feces white? Unlike mammals, birds don't urinate. Their kidneys extract nitrogenous wastes from the bloodstream, but instead of excreting it as urea dissolved in urine as we do, they excrete it in the form of uric acid. Uric acid has a very low solubility in water, so it emerges as a white paste. This material, as well as the output of the intestines, emerges from the bird's cloaca. |
;
Pressures produced when penguins pooh -- Brooding Chinstrap (Pygoscelis antarctica ) and Adélie (P. adeliae) penguins do not leave their stony nest to defecate, but move to the edge of it, stand up, turn their back nest-outward, bend forward, lift their tail, and shoot The expelled material hits the ground an average of 40 cm away from the bird. Meyer-Rochow and Gal (2004) determined that the pressures involved could be approximated if they knew the (1) distance the feces traveled, (2) density and viscosity of the material, and (3) shape, aperture, and height of the anus above ground. They determined these parameters and calculated that penguins generated pressures of around 10 kPa (77 mm Hg) to expel watery material and 60 kPa (450 mm Hg) to expel material of higher viscosity similar to that of olive oil (forces well above those known for humans). How penguins choose the direction of defecation, and how wind direction factors into that decision, remain unknown. |
Accessory organs:
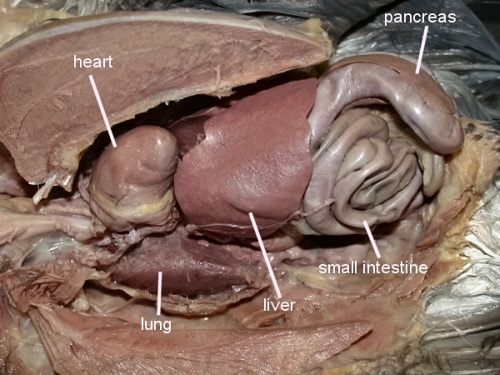
Pigeon liver and pancreas (& other organs)
(http://www.wtamu.edu/~rmatlack/pigeon_dissection/body_cavity.jpg)
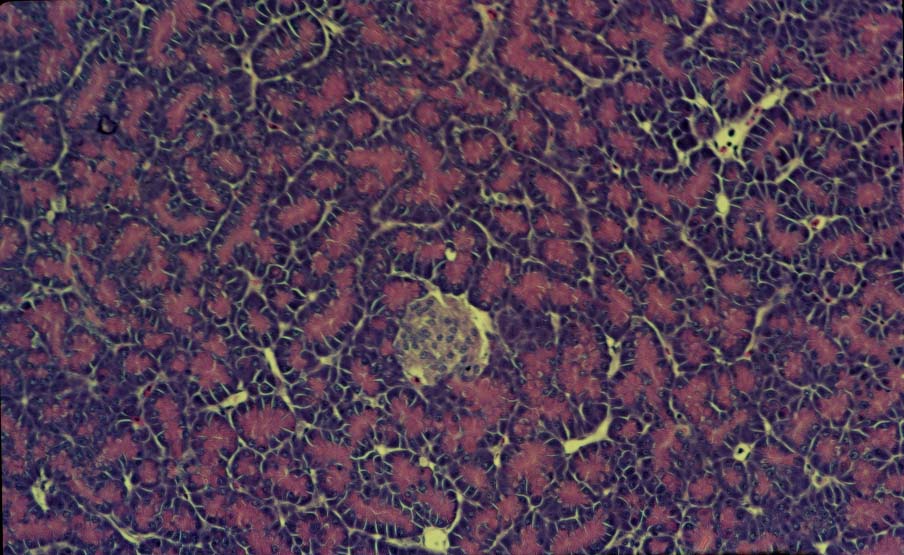
Avian Pancreas tissue
(Source: http://www.uga.edu/~poultry/faculty/compton/ps3800/ps3800slides.htm)
Literature Cited:
Benedict et al. 2024. Phylogenetic analyses support flush-pursuit foraging and f locking behaviors as evolutionary drivers of flash plumage signals in North American passerines. Ornithology 142: 114.
Beninger, P. G., and R. W. Elner. 2020. On the tip of the tongue: natural history observations that transformed shorebird ecology. Ecosphere 11: e03133.
Brightsmith, D. J. and R. A. Muñoz-Najar. 2004. Avian geophagy and soil characteristics in southeastern Peru. Biotropica 36: 534-543.
Cameron et al. 2023. Ecomorphological adaptations of owl feet and talons. Journal of Zoology 319: 285295.
Canham et al. 2023. The exploitation of biofilm by migrant western sandpipers (Calidris mauri). Heliyon 9: e17268.
Carlo et al. 2022. Functional ecology of Neotropical frugivorous birds. Ornithology Research 30: 139154.
Casotti, G. 2001. Luminal morphology of the avian lower intestine: evidence supporting the importance of retrograde peristalsis for water conservation. Anatomical Record 263: 289-296.
Catroxo, M. H. B., M. A. I. Lima, and C. E. M. P. D. M. Cappellaro. 1997. Histological aspects of the stomach (proventriculus and gizzard) of the Red-capped Cardinal (Paroaria gularis gularis). Rev. Chil. Anat. 15: 19-27.
Chikilian, M. and N. B. de Speroni. 1996. Comparative study of the digestive system of three species of tinamou. I. Crypturellus tataupa, Nothoprocta cinerascens, and Nothura maculosa (Aves: Tinamidae). Journal of Morphology 228: 77-88.
Clench, M. H. 1999. The avian cecum: update and motility review. Journal of Experimental Zoology 283: 441-447.
Conover, M. R. and D. E. Miller. 1980. Rictal bristle function in Willow Flycatcher. Condor 82: 469-471.
Cuban et al. 2026. Divergent nectar-feeding mechanisms evidenced by intralingual suction in sunbirds. Current Biology 36: 110.
De Voe, R., L. Degernes, and K. Karli. 2003. Dysplastic koilin causing proventricular obstruction in an Eclectus Parrot (Eclectus roratus). Journal of Avian Medicine and Surgery 17: 27–32.
Duke, G.E. 1994. Anatomy and physiology of the digestive system in fowl. Pages 46-49 in Proc. 21st Annual Carolina Poultry Nutrition Conference, Dec. 7-8, Charlotte, N.C.
Duque-Correa et al. 2025. Does intestine length explain digesta retention times in birds and mammals? Comparative Biochemistry and Physiology A 300: 111789.
Eglitis, I. and R. A. Knouff. 1962. An histological and histochemical analysis of the inner lining and glandular epithelium of the chicken gizzard. American Journal of Anatomy 111:49-65.
Einoder, L. and A. Richardson. 2006. An ecomorphological study of the raptorial digital tendon locking mechanism. Ibis 148: 515-525.
Ferraris, R. P. 2001. Dietary and developmental regulation of intestinal sugar transport. Biochem. J. 360:265-276.
Frei, S., S. Ortmann, M. Kreuzer, J.-M. Hatt, and M. Clauss. 2017. Digesta retention patterns in geese (Anser anser) and turkeys (Meleagris gallopavo) and deduced function of avian caeca. Comparative Biochemistry and Physiology A 204: 219-227.
Fritz, J., E. Kienzle, J. Hummel, O. Wings, W. J. Streich, and M. Clauss. 2011. Gizzard vs. teeth, it's a tie: food-processing efficiency in herbivorous birds and mammals and implications for dinosaur feeding strategies. Paleobiology 37: 577-586.
Gillespie, M. J., V. R. Haring, K. A. McColl, P. Monaghan, J. A. Donald, K. R. Nicholas, R. J. Moore, and T. M. Crowley. 2011. Histological and global gene expression analysis of the 'lactating' pigeon crop. BMC Genomics 12: 452.
Grajal A. 1995. Structure and function of the digestive tract of the Hoatzin (Opisthocomus hoazin): a folivorous bird with foregut fermentation. Auk 112: 20-28.
Grémillet, D., A. Prudor, Y. le Maho, and H. Weimerskirch. 2012. Vultures of the seas: hyperacidic stomachs in Wandering Albatrosses as an adaptation to dispersed food resources, including fishery wastes. PLoS ONE 7: e37834.
Hunt et al. 2019. phylogeny and herbivory are related to avian cecal size. Scientific Reports 9: 4243.
Iwasaki, S. 2002. Evolution of the structure and function of the vertebrate tongue. Journal of Anatomy 201:1-14.
Jackowiak, H., W. Andrzejewski, and S. Godynicki. 2006. Light and scanning electron microscopic study of the tongue in the cormorant Phalacrocorax carbo (Phalacrocoracidae, Aves). Zoological Science 23: 161-167.
Jackowiak, H., K. Skieresz-Szewczyk, Z. Kwieciński, J. Trzcielińska-Lorych, and S. Godynicki. 2010. Functional morphology of the tongue in the nutcracker (Nucifraga caryocatactes). Zoological Science 27: 589-594.
Janzen, D. H., W. Hallwachs, and J. M. Burns. 2010. A tropical horde of counterfeit predator eyes. Proceedings of the National Academy of Sciences USA 107: 11659-11665.
Johnstone, D. W. 1958. Sex and age characters and salivary glands of the Chimney Swift. Condor 60: 73-84.
Karasov, W.H. 1990. Digestion in birds: chemical and physiological determinants and ecological implications. Stud. Avian Biol. 13: 391–415.
Karasova, W. H. and E. Caviedes-Vidal. 2021. Adaptation of intestinal epithelial hydrolysis and absorption of dietary carbohydrate and protein in mammals and birds. Comparative Biochemistry and Physiology A 253: 110860.
Klasing, K.C. 1998. Comparative Avian Nutrition. CAB International, New York, NY.
Kobayoshi et al. 1998. Fine structure of the tongue and lingual papillae of the penguin. Archives of Histology and Cytology 61: 37-46.
Knudson, E. I. 2002. Instructed learning in the auditory localization pathway of the Barn Owl. Nature 417:322-328.
Ladyguin, A. 2000. The morphology of the bill apparatus in the Steller's Sea Eagle. Pp. 1 - 10 in First Symposium on Steller's and White-tailed Sea Eagles in east Asia (M. Veta and M.J. McGrady, eds.). Wild Bird Society of Japan, Tokyo.
Levey, D. J., R. S. Duncan, & C. F. Levins. 2004. Animal behaviour: Use of dung as a tool by burrowing owls. Nature 431: 1038-1039.
López-Calleja, M. V., M. J. Fernández, and F. Bozinovic. 2003. The integration of energy and nitrogen balance in the hummingbird Sephanoides sephaniodes. J. Exp. Biol. 206:3349 -3359.
Mathot, K. J., A. Dekinga, and T. Piersma. 2017. An experimental test of state–behaviour feedbacks: gizzard mass and foraging behaviour in Red Knots. Functional Ecology 31: 1111-1121.
Machovsky-Capuska et al. 2012. Visual accommodation and active pursuit of prey underwater in a plunge-diving bird: the Australasian gannet. Proceedings of the Royal Society B 279: 41184125.
McWhorter, T. J. and C. Martinez del Rio. 2000. Does gut function limit hummingbird food intake? Physiological and Biochemical Zoology 73:313-324.
Mendoza et al. 2018. Protective role of the vulture facial skin and gut microbiomes aid adaptation to scavenging. Acta Veterinaria Scandinavica 60: 61.
Meyer-Rochow, V. B. and J. Gal. 2004. Pressures produced when penguins pooh—calculations on avian defaecation. Polar Biology 27: 56-58.
Mumme, R. L. 2002. Scare tactics in a neotropical warbler: white tail feathers enhance flush-pursuit foraging performance in the Slate-throated Redstart (Myioborus miniatus). Auk 119:1024-1035.
Natale, R. 2023. Using ecomorphology to understand the evolutionary history and spatial biodiversity of wading birds. Ph.D. dissertaion, University of Chicago, Chicago, IL.
Navalon, G., J. A. Bright, J. Marugan-Lobon, and E. J. Rayfield. 2018. The evolutionary relationship among beak shape, mechanical advantage, and feeding ecology in modern birds. Evolution 73: 422-435.
Pennycuick, C. J. 2008. Gliding flight and soaring. In: Theoretical Ecology Series, vol. 5. Modelling the flying bird (C. J. Pennycuick, ed.), pp. 271-304. Elsevier Inc., Amsterdam, The Netherlands.
Norberg, U. M. L. 2002. Structure, form, and function of flight in engineering and the living world. Journal of Morphology 252: 52-81.
Pauw, A. 1998. Pollen transfer on birds' tongues. Nature 394:731-732.
Piersma, T. and J. Drent. 2003. Phenotypic flexibility and the evolution of organismal design. Trends in Ecology and Evolution 18: 228-233.
Pigot, A. L., C. Sheard, E. T. Miller, T. P. Bregman, B. G. Freeman, U. Roll, N. Seddon, C. H. Trisos, B. C. Weeks, and J. A. Tobias. 2020. Macroevolutionary convergence connects morphological form to ecological function in birds. Nature Ecology & Evolution 4: 230-239.
Prakash, M., D. Quéré, and J. W. M. Bush. 2008. Surface tension transport of prey by feeding shorebirds: the capillary ratchet. Science 320: 931-934.
Rico-Guevara, A., and M. A. Rubega. 2011. The hummingbird tongue is a fluid trap, not a capillary tube. Proceedings of the National Academy of Sciences USA 108: 9356-9360.
Ropert-Coudert, Y., D. Grémillet, P. Ryan, A. Kato, Y. Naito, and Y. Le Maho. 2004.
Between air and water: the plunge dive of the Cape Gannet Morus capensis.
Ibis 146: 281-290.
Ross, E. 2014. Woodpeckers and the biomechanics of concussion. M. S. thesis, University of British Columbia, Vancouver, BC, Canada.
Roth, T. C., II, and S. L. Lima. 2007. Use of prey hotspots by an avian predator: purposeful unpredictability? American Naturalist 169: 264-273.
Salazar et al. 2020. Anatomical Specializations Related to Foraging in the Visual System of a Nocturnal Insectivorous Bird, the Band-Winged Nightjar (Aves: Caprimulgiformes), Brain, Behavior and Evolution 94: 2736.
Sayol et al. 2026. AVONICHE: A Global Dataset of Dietary and Foraging Niches for Birds. Global Ecology and Biogeography, https://doi.org/10.1111/geb.70197.
Seki, Y., M. S. Schneider, and M. A. Myers. 2005. Structure and mechanical behavior of a toucan beak. Acta Materialia 53: 5281-5296.
Sereno, P. C., Z. Xijin, and T. Lin. 2010. A new psittacosaur from Inner Mongolia and the parrot-like structure and function of the psittacosaur skull. Proceedings of the Royal Society B 277: 199-209.
Shifferman, E. and D. Eilam. 2004.
Movement and direction of movement of a simulated prey affect the success rate in Barn Owl Tyto alba attack.
Journal of Avian Biology 35: 111-116.
Stevens, C. E. and I. D. Hume. 1998. Contributions
of Microbes in Vertebrate Gastrointestinal Tract to Production and
Conservation
of Nutrients. Physiological Reviews 78: 393-427.
Sustaita, D. 2008. Musculoskeletal underpinnings to differences in killing behavior between North American accipiters (Falconiformes: Accipitridae) and falcons (Falconidae). Journal of Morphology 269: 283-301.
Thouzeau, C., G. Peters, C. Le Bohec, and Y. Le Maho1. 2004. Adjustments of gastric pH, motility and temperature during long-term preservation of stomach contents in free-ranging incubating King Penguins. Journal of Experimental Biology 207: 2715-2724.
van der Meij, M. A. A. 2005. A tough nut to crack. Adaptations to seed cracking in finches. Ph. D. diss., Leiden University, Leiden, The Netherlands.
van Gils, J. A., T. Piersma, A. Dekinga, and M. W. Dietz. 2003. Cost-benefit analysis of mollusc-eating in a shorebird. II. Optimizing gizzard size in the face of seasonal demands. J. Exp. Biol. 206:3369 -3380.
Villard, P. and J. Cuisin. 2004. How do woodpeckers extract grubs with their tongues? A study of the Guadeloupe Woodpecker in the French West Indies. Auk 121:509-514.
Wang, L., J. T.-M. Cheung, F. Pu, D. Li, M. Zhang, and Y. Fan. 2011. Why do woodpeckers resist head impact injury: a biomechanical investigation. PLoS ONE 6: e26490.
Ward, A. B., P. D. Weigl, and R. M. Conroy. 2002. Functional morphology of raptor hindlimbs: implications for resource partitioning. Auk 119: 1052-1063.
Warrick, D. R. 198. The turning- and linear-maneuvering performance of birds: the costs of efficiency for coursing insectivores. Canadian Journal of Zoology 76: 1063-1079.
Wink et al. 2026. What, if anything, is a vulture? Phylogeny and trait evolution in obligate avian scavengers. Journal of Ornithology, https://doi.org/10.1007/s10336-025-02358-1.
Yanega, G. M., and M. A. Rubega. 2004. Hummingbird jaw bends to aid insect capture. Nature 428: 615.
Yoon, S.-H., and S. Park. 2011. A mechanical analysis of woodpecker drumming and its application to shock-absorbing systems. Bioinspiration & Biomemetics 6: 1-12.
Zaefarian et al. 2019. Avian liver: the forgotten organ. Animals 9: 63.
Zhang et al. 2026. Gut Health in Ostriches (Struthio camelus): Insights Into Intestinal Structure, Functions, Microbiome, and Improvement Strategies. Animal Research & One Health, https://doi.org/10.1002/aro2.70069.
|
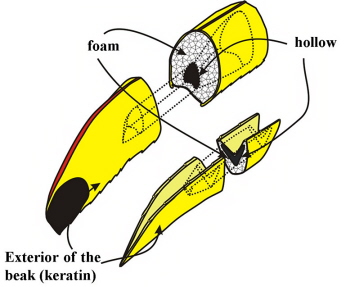 |
 The interior of the beak is rigid "foam" composed of bony fibers and drum-like membranes sandwiched between outer layers of keratin. The "foam" is covered with overlapping keratin tiles, each about 50 µm in diameter and 1 µm thick, glued together to form sheets.The closed, air-filled spaces reduce overall weight without loss of rigidity. |
Toucan bills: strong but light -- Toucans live in the jungle canopies of South America and feed on tree fruits growing at the ends of branches. They perch on sturdier portions of a branch and use their long beak to reach their food. So, the beak must be rigid enough to resist bending and twisting forces, but has to be light or the bird couldn't get off the ground. Incredibly, a toucan's beak that comprises about 1/3 of its body length, only makes up about 1/20 of a toucan's mass. The lightweight strength of the toucan's beak is due to a matrix of bony fibers and drum-like membranes sandwiched between an outer layer of keratin. Seki et al. (2005) found that the secret to the toucan beak's lightweight strength is an unusual bio-composite. The interior of the beak is rigid "foam" composed of bony fibers and drum-like membranes sandwiched between outer layers of keratin.
The relation between rate of success and direction of movement for a food item that was pulled forward (a), backward (b) and sideways (c). Direction of prey progression – dotted arrow (1), direction of owl flight – dashed arrow (2), and direction to which the owl had to move its head or trunk – solid arrow (3). Owl picture from Knudsen (2002). Movement and direction of prey affect raptor success rate -- Shifferman and Eilam (2004) tested a novel idea, that rather than maximizing their distance from a predator during close-distance encounters, prey species are better off moving directly or diagonally toward the predator in order to increase the relative speed and confine the attack to a single available clashing point. They used two tamed Barn Owls (Tyto alba) to measure the rate of attack success in relation to the direction of prey movement. A dead mouse or chick was used to simulate the prey, pulled to various directions by means of a transparent string during the owl's attack. Both owls showed a high success rate in catching stationary compared with moving food items (90% and 21%, respectively). Success was higher when the prey moved directly away, rather than towards the owls (50% and 18%, respectively). Strikingly, these owls had 0% success in catching food items that were pulled sideways. This failure to catch prey that move sideways may reflect constraints in postural head movements in aerial raptors that cannot move the eyes, but rather move the entire head in tracking prey. So far there is no evidence that defensive behavior in terrestrial prey species takes advantage of the above escape directions to lower rates of predator success. However, birds seem to adjust their defensive tactics in the vertical domain by taking-off at a steep angle, thus moving diagonally toward the direction of an approaching aerial predator. These preliminary findings warrant further studies in Barn Owls and other predators, in both field and laboratory settings, to uncover fine predator head movements during hunting, the corresponding defensive behavior of the prey, and the adaptive significance of these behaviors. |