
Living in Groups
Groups:
Are there adantages to being in such groups/flocks? If so, what are they??
Seminar - Ultimate and proximate benefits of social behavior in Superb Starlings by Sarah Guindre-Parker
Foraging groups:

2. Can reduce variance in an individual's feeding rate (Baker et al. 1981)
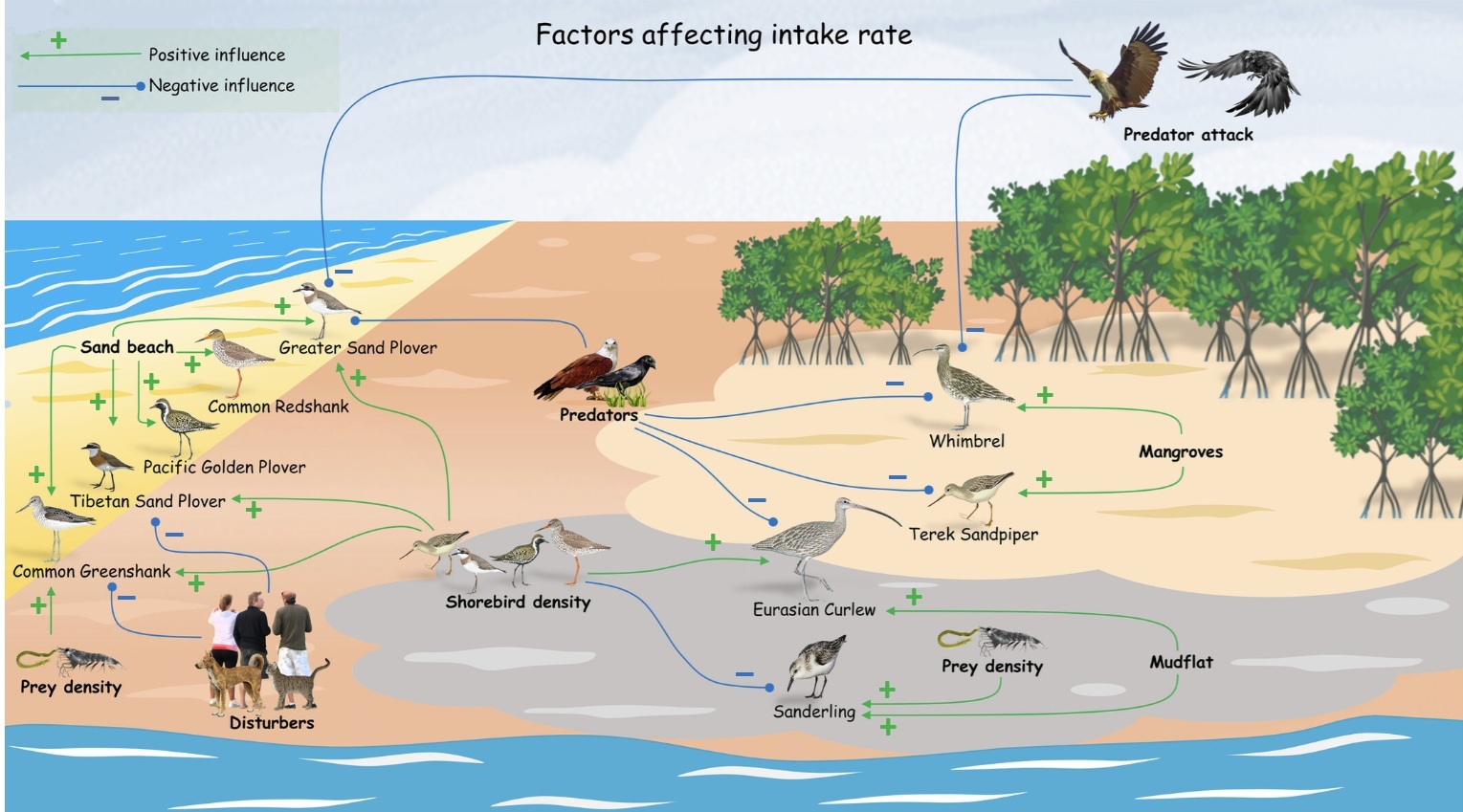
Factors influencing food intake rate of shorebirds. Food intake rates of most shorebird species in this study were positively affected by the density of
shorebirds and were also affected by habitat type. Intake rates of nearly half of the shorebird species were negatively affected by the density of predators or
predator attack rates. Human disturbance had a negative effect on intake rates of three species of shorebirds (Common Greenshanks, Tibetan Sand Plovers,
and [not shown in this figure) Eurasian Curlews). Intake rates were also influenced by habitat, with positive effects for several species of shorebirds on sand
beaches. Other habitats with positive effects on intake rates included mangroves (Whimbrels and Terek Sandpipers) and mudflats (Eurasian Curlews and Sanderlings).
Larger groups are more efficient problem solvers -- Group living commonly helps organisms face challenging environmental conditions. Although a known phenomenon in humans, recent findings suggest that a benefit of group living in animals generally might be increased innovative problem-solving efficiency. This benefit has never been demonstrated in a natural context, however, and the mechanisms underlying improved efficiency are largely unknown. Morand-Ferron and Quinn (2011) examined the problem-solving performance of Great and Blue tits at automated devices and found that efficiency increased with flock size. This relationship held when restricting the analysis to naive individuals, demonstrating that larger groups increased innovation efficiency. In addition to this effect of naive flock size, the presence of at least one experienced bird increased the frequency of solving, and larger flocks were more likely to contain experienced birds. These findings provide empirical evidence for the “pool of competence” hypothesis in nonhuman animals. The probability of success also differed consistently between individuals, a necessary condition for the pool of competence hypothesis. Solvers had a higher probability of success when foraging with a larger number of companions and when using devices located near rather than further from protective tree cover, suggesting a role for reduced predation risk on problem-solving efficiency. In contrast to traditional group living theory, individuals joining larger flocks benefited from a higher seed intake, suggesting that group living facilitated exploitation of a novel food source through improved problem-solving efficiency. Together these results suggest that both ecological and social factors, through reduced predation risk and increased pool of competence, mediate innovation in natural populations.
Links:
Birds of diverse feathers, problem-solving together
Feathered friends help wild birds innovate
Better problem-solving in groups

Social status and physiological costs -- For group-living animals, the maintenance of a position in the social hierarchy may be associated with physiological costs such as increased stress and energy expenditure or suppressed immune functions. Lindstrom et al. (2005) experimentally manipulated the social status of House Sparrows so that each bird experienced two social environments in random sequence: being dominant and subordinate. For 14 males, Lindstrom et al. examined how corticosterone concentrations, energy expenditure and immune functions were affected by these changes in social status position. The cost of maintaining a social status position differed between individuals and were related to individual body size. Birds with small body size had increased costs in terms of increased stress responses and reduced cell-mediated immune responses while being experimentally kept as dominants, while birds with large body size had increased costs while they were subordinates. Lindstrom et al. also found that birds with increased energetic and immunological costs as dominants obtained a low status position in the large group, while birds with increased costs as subordinates obtained a high status position in the large group. In summary, the costs associated with the maintenance of social status position differed between individuals and was related to the individuals’ body size. Furthermore, in a large group, individuals maintained a social status position that minimized energetic and immunological costs.
Why do birds participate in mixed-species foraging flocks? Mixed-species flocks of foraging birds have been documented from terrestrial habitats all over the world and are thought to form for either improved feeding efficiency or better protection from predators. Two kinds of flock participants are recognized: those that join other species (followers) and are likely to be the recipients of the benefits of flock participation and those that are joined (leaders). Through comparative analyses, using a large sample of flocks from around the world, Sridhar et al. (2009) found that (1) followers tend to be smaller, more insectivorous, and feed in higher strata than species that are less likely to join flocks, and (2) leaders tend to be cooperative breeders more often than species not known to lead flocks. In addition, meta-analyses of published results from across the world revealed that bird species in terrestrial mixed-species flocks increase foraging rates and reduce vigilance compared to when they are solitary or in conspecific groups. Moreover, the increase in foraging rates is seen only with flock followers and not flock leaders. These results suggest a role for predation in the evolution of mixed-species flocking. Species that are vulnerable to predation follow species whose vigilance they can exploit. By doing so, they are able to reduce their own vigilance and forage at higher rates.
3 - Information center hypothesis. Groups or colonies may be advantageous if individuals share information about the location of ephemeral patches of food (e.g., Krebs 1974)

Rate of food intake of adult Great Blue Herons as a function of flock size. The regression
equation describing the relationship was arrived at by step-wise multiple regression. The
data are grouped into blocks according to flock size purely to make the figure look neater;
the analysis was done on the original points. The vertical bars represent standard errors
of the means of grouped data (From: Krebs 1974).

Red-billed Quelea
4 - Groups may flush prey from hiding places & reduce time between successive prey captures
5 - Group hunting can influence the availability of different food resources & the efficiency of capture:
Social dynamics of core members in mixed-species bird flocks change across a gradient of foraging habitat quality - Social associations within mixed-species bird flocks can promote information flow about food availability and provide predator avoidance benefits. The relationship between flocking propensity, foraging habitat quality, and interspecific competition can be altered by human-induced habitat degradation. Richardson et al. (2022) examined sociality of two ecologically important flock-leader (core) species, Carolina Chickadees (Poecile carolinensis) and Tufted Titmice (Baeolophus bicolor), to better understand how degradation of foraging habitat quality affects mixed-species flocking dynamics. The authors compared interactions of free ranging wild birds across a gradient of foraging habitat quality in three managed forest remnants. Specifically, they examined aspects of the social network at each site, including network density, modularity, and species assortativity. Differences in the social networks between each end of our habitat gradient suggest that elevated levels of interspecific association were more valuable in the habitat with low quality foraging conditions. This conclusion was supported by two additional findings: First, foraging height for subordinate Carolina Chickadees relative to Tufted Titmice decreased with an increase in the number of satellite species in the most disturbed site, but not at the other two sites. Second, the chickadee gargle call rate, an acoustic signal given during agonistic encounters between conspecifics, was relatively higher at the high-quality site. Collectively, these results suggest an increase in heterospecific associations increased the value of cross-species information flow in degraded habitats - From: Richardson et al. 2022
|
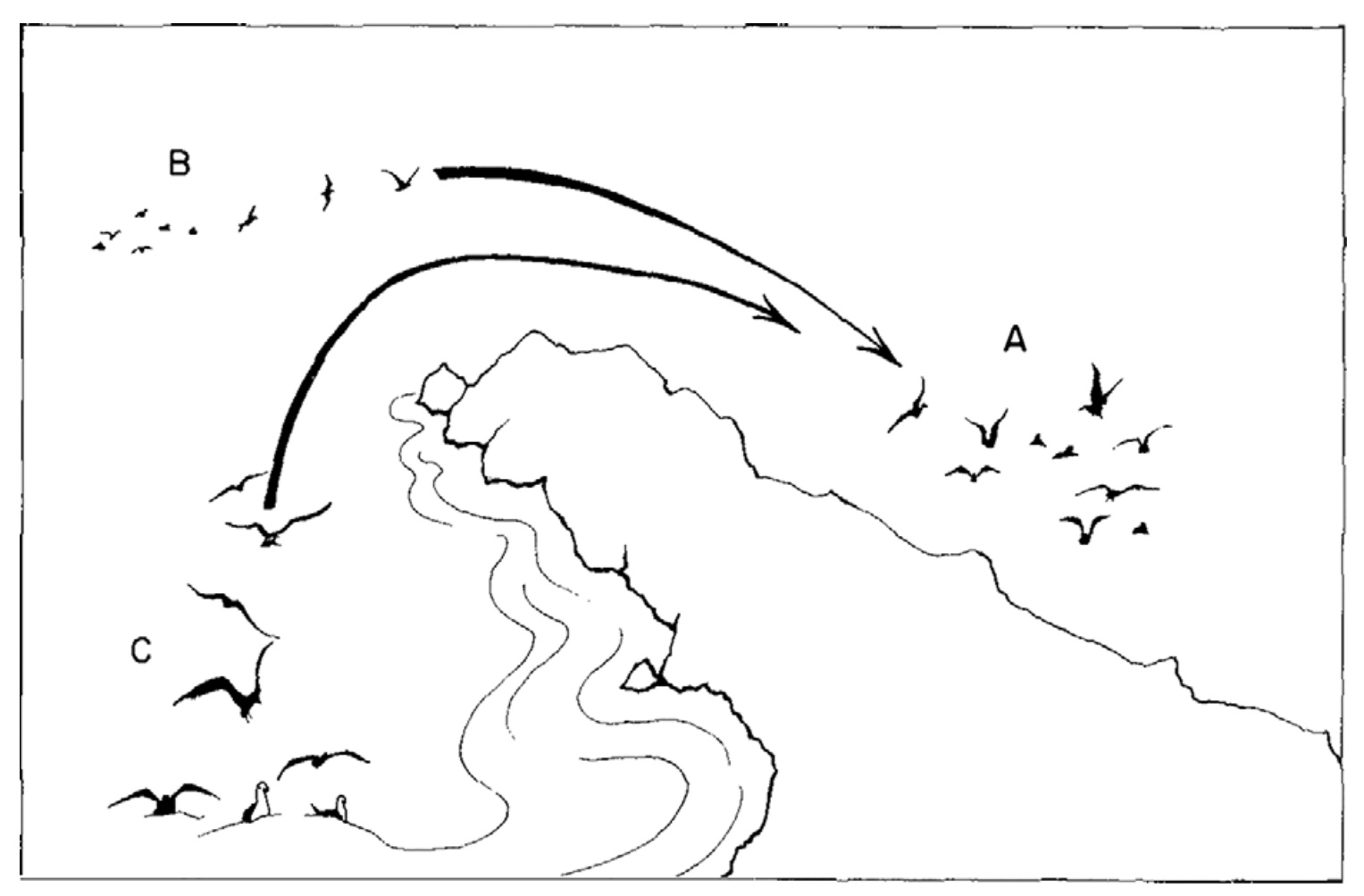
Seabirds use social cues to locate prey swarms. Patchfinding predators catch the attention of
other predators, alerting
them to the patch, a phenomenon known as local enhancement;
the
approaching birds benefit from the conspicuousness of the
others. In this image, Group A
draws in Group B. Group C
cannot see Group A or the patch itself, but takes cues from
Group
B, demonstrating the utility of at-sea associations in
locating even distant patches (From: Monier 2024).
Thiebault et al. (2014) observed that large
aggregations of predators could increase patch-detection distances by 1650% compared to solitary detection of a patch.
An experiment by Bairos-Novak et al. (2015) quantified the
use of local enhancement and found that the presence of
decoys on the water was up to 17 times more likely to elicit
a response from birds (circling, landing, etc.) relative to controls. Similar results have been obtained from observational
studies: McInnes and Pistorius (2019) found a strong relationship between penguins and flying birds at sea (flying birds
arrived quicker in response to larger groups of diving penguins) independent of prey biomass. Some research suggests
that seabirds generally do not search for prey directly, but
instead look for indicators of the presence of prey, likely
forming search images of other predators.

African Penguins feeding in a clockwise pattern that suggests coordination. At the center of these aggregations fish
sometimes move toward the surface. In this photo, other seabird species have joined the penguins to forage on the fish near
the surface (Image from Ryan et al. 2012).
Foraging & Group Size
Bird groups and predation
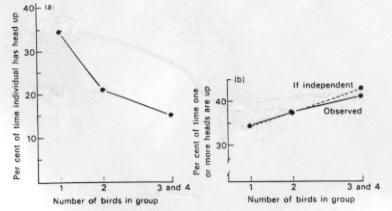
1. Groups may detect predators sooner than solitary individuals:
- Ostriches (Bertram 1980)
 |
 |
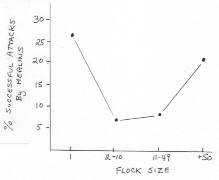
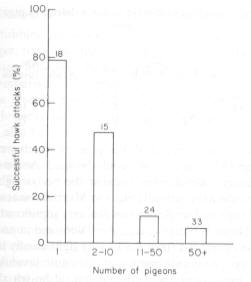 The percentage of hawk attacks which were successful at single pigeons and at flocks. Number of attacks is given above histograms (from Kenward 1978). |
 |

European Sparrowhawk
Photo source: BTO
| Sparrowhawks more often attack the more vulnerable prey group -- Whether predators always attack the most vulnerable prey or simply attack prey that exceeds a minimum vulnerability level is an important question to answer in furthering our understanding of predator and antipredation behaviour. Predators may attack any reasonably vulnerable prey rather than waste time identifying the most vulnerable prey, particularly when prey can respond quickly to alter their vulnerability in response to a predator. Cresswell and Quinn (2004) tested whether European Sparrowhawks always choose to attack the group of prey that maximises their capture probability, or whether they simply attack any group above a minimum vulnerability. They modelled sparrowhawk attack success when hunting Redshanks using data from three winters and found that probability of capture increased when group size or distance to predator-concealing cover decreased. They then used this model to predict the relative vulnerability to capture of redshank groups occurring in pairs in a fourth winter and found that sparrowhawks attacked the most vulnerable prey group twice as often as not (66%, N = 59 pairs). When sparrowhawks attacked the less vulnerable group, there was no tendency for both groups to be particularly vulnerable or for the difference in the vulnerability between the two groups to be relatively small. This suggests that, while sparrowhawks do on average attack the most vulnerable group available, they consider other factors that affect vulnerability or that additional factors lead them to also attack opportunistically. This suggests that there will be selection for the predator to monitor a large number of prey individuals and groups and for prey to have the ability to monitor the behaviour of conspecifics in the same and different groups so that they can assess relative vulnerability. |
The probability of capture dependent on group size and distance to cover modelled from empirical data of whether a sparrowhawk attack led to the capture of a redshank. Both graphs show the same model but the top graph plots the probability in terms of group size with isoclines for distance from cover and the bottom graph plots the probability in terms of distance from cover with isoclines for group size. |
So, individuals in groups may be safer from predators. However,
there may be limits to this benefit. If groups get too large, individuals
may be more vulnerable to predators.
 |
 |
In some groups, certain individuals or species (in mixed-species groups) may act as 'lookouts', e.g., scrub jays, and mixed-species flocks of birds in the tropics. For example, Munn (1986) found that, among mixed species flocks of birds in the Amazon forest, White-winged Shrike-tanagers act as sentinels in canopy flocks & Bluish-slate Antshrikes as sentinels in understory flocks. These species are usually the first to give alarm when bird-eating hawks approach; other species respond by freezing or seeking cover. Both species occasionally utter 'false' alarm calls to distract other birds & increase their chances of capturing prey. The 'penalty' for individuals of other species that ignore calls may be death.
4. Group members may benefit from the 'dilution effect'
2. Prey may form groups in an attempt to place conspecifics between themselves & an attacking predator.
- Individuals on periphery of group may experience increased predation
- 'Selfish-herding' may interact with other anti-predator benefits or foraging benefits (if not, why should 'peripheral' individuals remain in the group?)
3. Tightly aggregated prey may confuse or inhibit predator
- A Peregrine Falcon, for example, stooping at a dense flock of starlings could be injured (see video below).
Peregrine Falcon attacking large flock
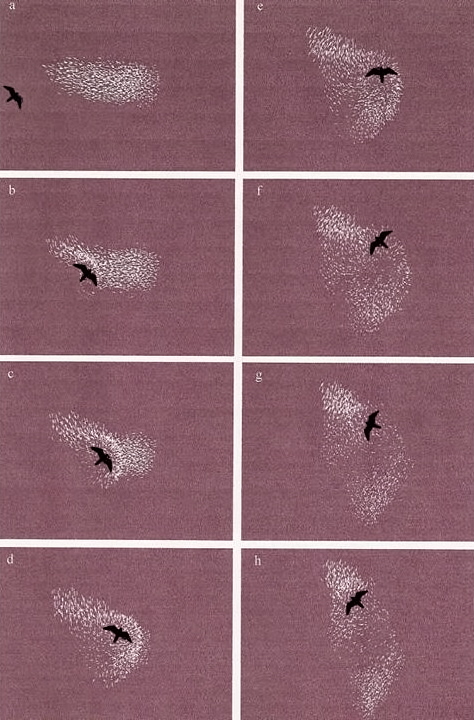
Simulation of a predator attacking a bird flock (a, upper left, through h, lower right). The predator follows a simple rule: move toward
the highest
perceived density of individuals. Flock members detect and move away from the predator and exhibit typical patterns, e.g.,
'flash expansion' as individuals move rapidly from the predator as it strikes, 'vacuolation', where expansion results in a cavity
forming around the predator, and the 'split effect', where the group fragments (Couzin et al. 2002).
A murmuration of European Startlings
New hypothesis: Murmurations as extended strategic negotiations over roost hierarchy and safe roost positions, particularly in starlings (Sturnus vulgaris)
|
Reduced flocking by birds on islands with relaxed |
What is the optimum group size?
Optimal flock size model. (a) As flock size increases, birds spend more time fighting & less time scanning.
(b) At higher temperatures (or when food is more abundant), dominant birds can spend more time attacking
subordinates. The optimal flock size then decreases. (c) When predation risk increases, scanning should increase
and the optimal flock size then increases. Based on Pulliam (1976) and Caraco et al. (1980). From: Krebs and Davies (1987).
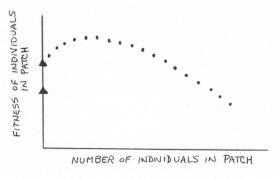
Once individual fitness declines so much (because of increased group size) that a bird could do better in a lower-quality patch
(represented by the bottom triangle on the vertical axis), then some flock members should leave the higher-quality patch and move to the lower-quality patch.
With fewer individuals present, birds in the lower-quality patch may have greater fitness than those in the higher-quality patch.
| For each group size, the fitnesses of all individuals are shown as separate points. Habitat 2 (triangle) will first be used when the most subordinate individual in Habitat 1 would be better off using the poorer habitat. | 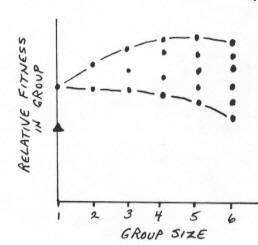 |
Individuals should join flocks based on their ability to dominate others & the current size of groups. |
CONCLUSIONS:
What is the optimal group size?
The group-size paradox: effects of learning and patch departure rules -- Joining groups is thought to enhance foraging success in many species. However, groups are often predicted to be larger than the size that represents the best option for individuals. This is because other solitary foragers in the habitat are expected to continue joining a group until remaining alone or joining the group becomes equivalent alternatives for these foragers. If such a mechanism were pervasive in nature, there would be paradoxically little incentive for group foraging to evolve as a successful strategy. Beauchamp and Fernández-Juricic (2005) proposed a solution to the group-size paradox by allowing foragers to learn about habitat quality. Foragers can learn what to expect in terms of food intake rate through repeated encounters with food patches within their habitat. Foragers are therefore predicted to leave groups when their current food intake rate falls below that expected for the whole habitat. Using a foraging simulation in a virtual habitat, they showed that under a wide range of population sizes, foragers using the above rules abandon under- and overcrowded patches ensuring that group size remains close to the preferred value. Beauchamp and Fernández-Juricic concluded that groups need not become too large when the best outcome for individuals occurs in groups of intermediate sizes. Therefore, there is no need to invoke relatedness between joiners and established group members, group defense against joiners, or other mechanisms that were proposed earlier to prevent groups from becoming too large. |
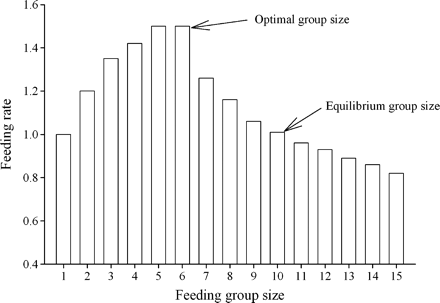 Intake rate as a function of the number of foragers in a food patch. In this example, solitary foragers in a food patch obtain one food item per time step. The addition of foragers to the patch first increases and then decreases food intake rate. The optimal and equilibrium group sizes are shown. |


Communal roosting in birds -- Three main benefits are thought to underlie communal roosting in birds: a reduction in thermoregulation demands, a decrease in predation risk, and an increase in foraging efficiency. Beauchamp (1999) investigated interspecific variation in communal roosting tendencies across categories of several ecological factors to examine the relevance of each functional hypothesis in the evolutionary transition to communal roosting and the secondary reversal to solitary roosting habits. The study phylogenetic tree included 30 families and 437 species. Evolutionary transitions to communal roosting occurred more often on branches with flocking species and with larger species but were not associated with diet, territoriality, geographical area, or time of day. The association with flocking activities suggests that increased foraging efficiency, a factor thought to operate through the formation of flocks, may have been a key factor in the origin of avian communal roosting. However, several transitions to communal roosting occurred on branches with nonflocking species, indicating that foraging efficiency may not be the only factor involved in the evolution of communal roosting. Secondary losses of communal roosting habits occurred on several branches, with a concomitant loss of flocking behavior and a tendency to exhibit territorial behavior and nocturnal foraging. Secondary losses suggest that communal roosting is costly to perform and maintain and may be lost when an asocial selection regime operates. Two lines of evidence suggest that communal roosting was costly to maintain and that under asocial conditions the behavior was selected against in favor of solitary roosting. First, most reversals occurred at the species level, which suggests that an asocial selection regime operated only recently, leaving little time for random drift mutations to act so consistently across so many species. Second, roosting behavior is often polymorphic in birds. In polymorphic species, some members of the population roost solitarily, while other members roost communally.
Territoriality and nocturnal foraging could promote an asocial selection regime and be involved in the transition to solitary roosting. Several species that reverted to solitary roosting are specialized foragers holding year-round territories. Joining a distant communal roost to increase foraging efficiency makes little sense in a territorial species and increases the risk that competitors take over the abandoned territory. Similarly, foraging at night can reduce the efficiency of visual recruitment and render group cohesion more difficult to maintain. Joining distant communal roosts can increase commuting costs, and, in the absence of benefits, communal roosting may make little sense for nocturnal foragers. In general, conditions that favor solitary foraging can create an asocial selection regime that could lead to the disappearance of costly social functions.
Literature cited:
Aarif et al. 2026. Spatio-temporal responses of a migratory shorebird community reflect complex trade-offs among overabundance of predators and disturbance. Ecological Solutions and Evidence, https://doi.org/10.1002/2688-8319.70199.
Bairos-Novak et al. 2015. Relative importance of local enhancement as a search strategy for foraging seabirds: an experimental approach. Animal Behaviour 106: 71-78.
Baker, M.C., C. S. Belcher, L. C. Deutsch, G. L. Sherman, and D. B. Thompson. 1981. Foraging success in junco flocks and the effects of social hierarchy. Anim. Behav. 29:137-142.
Beauchamp, G. 1999. The evolution of communal roosting in birds: origin and secondary loss. Behavioral Ecology 10: 675-687..
Beauchamp, G. 2004. Reduced flocking by birds on islands with relaxed predation. Proceedings of the Royal Society of London B 271: 1039-1042.
Beauchamp, G. and E. Fernández-Juricic. 2005. The group-size paradox: effects of learning and patch departure rules. Behavioral Ecology 16:352–357.
Bertram, B.C.R. 1980. Vigilance and group size in ostriches. Anim. Behav. 28: 278-286.
Buckley, N. J. 1996. Food finding and the influence of information, local enhancement, and communal roosting on foraging success of North American vultures. Auk 113:473-488.
Caccamise, D. F. and D. W. Morrison. 1988. Avian communal roosting: implications of "diurnal activity centers." American Naturalist 128:191-198.
Caraco, T. 1979. Time budgeting and group size: a theory. Ecology 60:611-618.
Caraco, T., S. Martindale, and H. R. Pulliam. 1980. Flocking: advantages and disadvantages. Nature 285: 400-401.
Couzin, I. D., J. Krause, R. James, G. D. Ruxton, and N. R. Franks. 2002. Collective memory and spatial sorting in animal groups. Journal of Theoretical Biology 218: 1-11.
Cresswell, W. and J. L. Quinn. 2004. Faced with a choice, sparrowhawks more often attack the more vulnerable prey group. Oikos 104: 71-76.
DeGroot, P. 1980. Information transfer in a socially roosting weaver bird: an experimental study. Anim. Behav. 28:1249-1254.
Du Plessis, M. A., W. W. Weathers, and W. D. Koenig. 1994. Energetic benefits of communal roosting by Acorn Woodpeckers during the non-breeding season. Condor 96:631-637.
Eiserer, L. A. 1984. Communal roosting in birds. Bird Behaviour 5: 61-80.
Evans, R. M. 1982. Foraging-flock recruitment at a Black-billed Gull colony: implications for the information center hypothesis. Auk 99:24-30.
Fretwell, S.D. 1972. Populations in a seasonal environment. Princeton Univ. Press, Princeton, NJ.
Kenward, R.E. 1978. Hawks and doves: factors affecting success and selection in Goshawk attacks on wood-pigeons. J. Anim. Ecol. 47:449-460.
Krebs, J.R. 1974. Colonial nesting and social feeding as strategies for exploiting food resources in the Great Blue Heron (Ardea herodias). Behaviour 51: 99-134.
Krebs, J. R. and N. B Davies. 1987. An introduction to behavioural ecology. Sinauer Associates, Sunderland, MA.
Krebs, J.R., M.H. MacRoberts, and J.M. Cullen. 1972. Flocking and feeding in the Great Tit: an experimental study. Ibis 114:507-530.
Lima, S.L. 1995. Back to the basics of antipredatory vigilance: the group size effect. Anim. Behav. 49:11-20.
Lima, S.L. and P.A. Bednekoff. 1999. Back to the basics of antipredatory vigilance: can nonvigilant animals detect attack? Anim. Behav. 58:537-543.
Lindstrom, K. M., D. Hasselquist, and M. Wikelski. 2005. House Sparrows adjust their social status position to their physiological costs. Hormones and Behavior 48: 311-320.
Mangini et al. 2023. A classification scheme for mixed-species bird flocks. Philos. Trans. R. Soc. B 378: 20220100.
McInnes, A. M. and P. S. Pistorius 2019. Up for grabs: prey hearding by penguins facilitates shallow foraging by volant seabirds. Royal Society Open Science 6: 190333.
Monier, S. A. 2024. Social information and information use by foraging seabirds. Biological Reviews 99: 1717-1735.
Morand-Ferron, J., and J. L. Quinn. 2011. Larger groups of passerines are more efficient problem solvers in the wild. Proceedings of the National Academy of Sciences USA, online early.
Munn, C.A. 1986. Birds that 'cry wolf'. Nature 319:143-145.
Page, G. and D.F. Whitacre. 1975. Raptor predation on wintering shorebirds. Condor 77:73-83.
Pulliam, H. R. 1976. The principle of optimal behavior and the theory of communities. In: Perspectives in ethology (P. H. Klopfer and P. P. G. Bateson, eds.), pp. 311-332. Plenum Press, New York, NY.
Pulliam, H.R., G.H. Pyke, and T. Caraco. 1982. The scanning behavior of juncos: a game-theoretical approach. J. theor. Biol. 95:89-103.
Richardson, K. E., D. P. Roche, S. G. Mugel, N. D. Lancaster, K. E. Sieving, T. M. Freeberg, and J. R. Lucas. 2022. Social dynamics of core members in mixed-species bird flocks change across a gradient of foraging habitat quality. PLoS ONE 17: e0262385.
Richner, H. and P. Hebb. 1995. Is the information centre hypothesis a flop? Advances in the Study of Behavior 24:1-45.
Ryan et al. 2012. African Penguins, bait balls and the Allee effect. Ardea 100: 89-94.
Sridhar et al. 2009. Why do birds participate in mixed-species foraging flocks? A large-scale synthesis. Animal Behaviour 78: 337-347.
Thiebault et al. 2014. Local enhancement in a seabird: reaction distances and foraging consequence of predator aggregations. Behavioral Ecology 25: 1302-1310.
Ward, P. and A. Zahavi. 1973. The importance of certain assemblages of birds as `information-centres' for food-finding. Ibis 115:517-534.
Weatherhead, P. J. 1983. Two principal strategies in avian communal roosts. American Naturalist 121:237-243.
Yom-Tov, Y., A, Imber, and J. Otterman. 1977. The microclimate of winter roosts of the starling Sturnus vulgaris in Israel. Ibis 119:366-368.