|
Ornithology Lecture Notes 2 - Bird Flight I |
 |
|
Ornithology Lecture Notes 2 - Bird Flight I |
 |
| Origin of Flight
Exactly how birds acquired the ability to fly has baffled scientists for years. Archaeopteryx provided a starting point for speculation. Built like a dinosaur, but with wings, scientists guessed at how a hypothetical ancestor might have taken flight. Some scientists support the arboreal hypothesis (e.g., Feduccia 1996) and suggest that the ancestors of Archaeopteryx lived in trees and glided into flapping flight (Figure to the right). But others argue that the claws of Archaeopteryx weren't suited to climbing. So, others support the cursorial hypothesis (e.g., Burgers and Chiappe 1999) and suggest that these ancestors used their long, powerful legs to run fast with their arms outstretched, and were at some point lifted up by air currents and carried into flapping flight (Figure to the bottom right). Studying living animals can throw light on their evolutionary past. Ken Dial (2003) of the Flight Lab at the University of Montana noticed the ability of gamebird chicks to escape danger by scrambling up vertical surfaces. The chicks first run very fast, flapping their immature, partially feathered wings, frantically creating enough momentum to run up a vertical surface to safety. Could this survival instinct be the origin of flight? Whatever the origins, dinosaurs, and birds, eventually took to the air. Images & text used with permission.
|
|
| Dinosaurs' flapping led to flight? The wing-assisted incline running hypothesis -- The feathered forelimbs of small, two-legged dinosaurs may have helped them run up hills or other inclines to escape predators. This half running, half flapping may have evolved into an ability to fly. Dial (2003) reported findings suggesting that the ability to fly evolved gradually. Feathers may have first protected animals from cold & wet weather, then been used out of necessity when something with big teeth was chasing them. Even before their wings develop enough to fly, some living birds use them to improve traction and gain speed. Dial studied birds, like partridges, capable of only limited flight. Energetically, "It's a lot cheaper to run than fly," Dial said. So these baby birds, with big feet & powerful legs, use them in combination with their wings, first to stay balanced and grounded, then to take on steeper and steeper inclines. Using this "wing assisted incline running," Chukar Partridges can negotiate 50 degree inclines right after hatching, 60 degree slopes at 4 days old, and at 20 days, can perform a vertical ascent. "The wings help them stick to the ground," said Dial. The wings only come into play on steep angles because at about a 50 - 60 degree incline the birds start slipping. Then they begin a head to tail movement, like a reptile, that pushes them to the ground to enhance traction. "They use their wings like spoilers on a race car, to give their feet better traction," he said. Use of this wing-assisted running doesn't stop when the birds are old enough to fly. Adult birds often choose the running and flapping option instead of flying because it is more energy efficient. - Written by Marsha Walton, CNN |
Chukar Partridge flapping & climbing
 |
 |
Biplane wing planform and flight performance of a feathered dinosaur (Chatterjee and Templin 2007) -- Microraptor gui, a four-winged dromaeosaur from the Early Cretaceous of China, provides strong evidence for an arboreal-gliding origin of avian flight. It possessed asymmetric flight feathers not only on the manus but also on the pes. A previously published reconstruction shows that the hindwing of Microraptor supported by a laterally extended leg would have formed a second pair of wings in tetrapteryx fashion. However, this wing design conflicts with known theropod limb joints that entail a parasagittal posture of the hindlimb. Here, we offer an alternative planform of the hindwing of Microraptor that is concordant with its feather orientation for producing lift and normal theropod hindlimb posture. In this reconstruction, the wings of Microraptor could have resembled a staggered biplane configuration during flight, where the forewing formed the dorsal wing and the metatarsal wing formed the ventral one. The contour feathers on the tibia were positioned posteriorly, oriented in a vertical plane for streamlining that would reduce the drag considerably. Leg feathers are present in many fossil dromaeosaurs, early birds, and living raptors, and they play an important role in flight during catching and carrying prey. A computer simulation of the flight performance of Microraptor suggests that its biplane wings were adapted for undulatory "phugoid" gliding (see below) between trees, where the horizontal feathered tail offered additional lift and stability and controlled pitch. Like the Wright 1903 Flyer, Microraptor, a gliding relative of early birds, took to the air with two sets of wings.

Phugoid gliding is a type of flight where a plane (or Microraptor gui) pitches up and climbs, and then pitches down and descends,
accompanied by speeding up and slowing down as it goes "uphill" and "downhill.
The Four-winged Dinosaur (NOVA)

A dinosaur fossil unearthed in the Gobi Desert of Mongolia shows that miniaturization, a hallmark of bird origins and a necessary precursor of flight, occurred progressively in primitive dinosaurs (Credit: F. Ippolito, American Museum of Natural History).
Theropod size and avian flight -- An 80-million-year-old dinosaur fossil unearthed in the Gobi Desert of Mongolia demonstrates that miniaturization, long thought to be a hallmark of bird origins and a necessary precursor of flight, occurred progressively in primitive dinosaurs. "This study alters our understanding of the evolution of birds by suggesting that flight is a 'spin-off' adaptation of a much earlier trend toward miniaturization in certain dinosaur lineages," said H. R. Lane (NSF). "Paleontologists thought that miniaturization occurred in the earliest birds, which then facilitated the origin of flight," said Alan Turner (American Museum of Natural History). "Now the evidence shows that this decrease in body size occurred well before the origin of birds and that the dinosaur ancestors of birds were, in a sense, pre-adapted for flight." Because most dinosaurs were too massive to fly, miniaturization is considered crucial to the origin of flight. To date, fossil evidence of miniaturization and other characteristics leading to flight has been sparse. While other dinosaurs of the Cretaceous Period were increased in size, this newly discovered dinosaur (Mahakala omnogovae) represented a step towards miniaturization necessary for flight. Other groups that evolved flight, such as pterosaurs and bats, all evolved from small ancestors. With the discovery of Mahakala, Turner et al. (2007) showed that this miniaturization occurred much earlier." Mahakalal was nearly full-grown when it died, measuring less than two feet in length and weighing about 24 ounces. In the broader context of the dinosaur family tree, Mahakala shows that dinosaurs' size decreased progressively as they evolved toward birds. "Many of the animals that were thought to look like giant lizards only a few years ago are now known to have been feathered, to have brooded their nests, to have been active, and to have had many other defining bird characteristics, like wishbones and three forward-facing toes," said Mark Norell (American Museum of Natural History). "We can now add that the precursors of birds were also small, primitive members of a lineage that later grew much larger--long after their divergence from the evolutionary stem leading to birds."
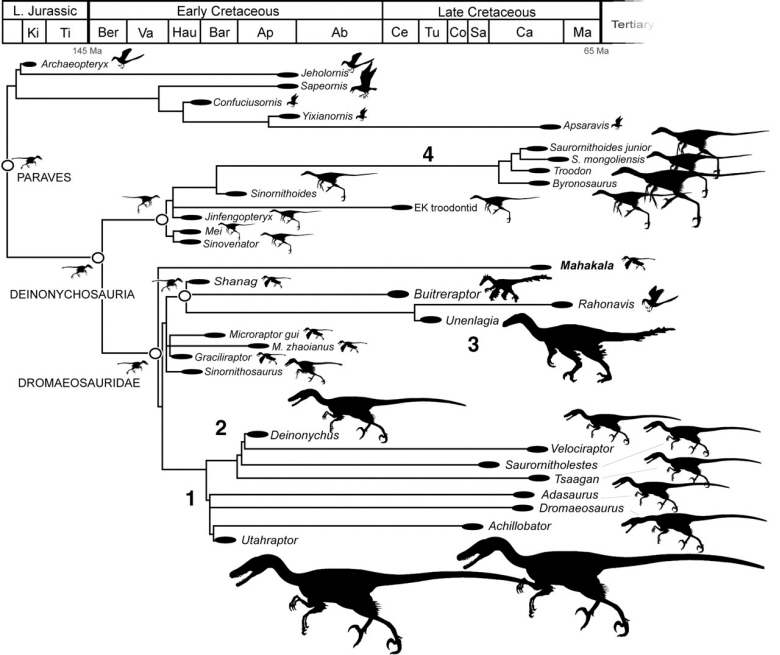
Phylogeny and body size change within paravian theropods. A temporally calibrated cladogram depicting the phylogenetic position of Mahakala and paravian body size through time and across phylogeny is shown. Silhouettes are to scale, illustrating the relative magnitude of body size differences. Left-facing silhouettes near open circles show reconstructed ancestral body sizes. Ancestral paravian body size is estimated to be 600 to 700 g and 64 to 70 cm long. The ancestral deinonychosaur, troodontid, and dromaeosaurid body size is estimated at 700 g. Large numbers (1, 2, 3, and 4) indicate the four major body increase trends in Deinonychosauria. Ma, Maastrichtian; Ca, Campanian; Sa, Santonian; Co, Coniacian; Tu, Turonian; Ce, Cenomanian; Ab, Albian; Ap, Aptian; Bar, Barremian; Hau, Hauterivian; Va, Valanginian; Ber, Berriasian; Ti, Tithonian; Ki, Kimmeridgian. Ma, million years ago (From: Turner et al. 2007).
The flight apparatus in Early Cretaceous birds compared to that of present-day birds.
The flight of birds is powered by two major muscles originating primarily on the sternum: the pectoralis that pulls the wing down, generating thrust and lift, and the supracoracoideus that elevates the wing via a pulley system. This pulley mechanism involves the tendon of supracoracoideus passing through the triosseal canala bony passage formed by the scapula, coracoid, and furculabefore attaching to the humerus.
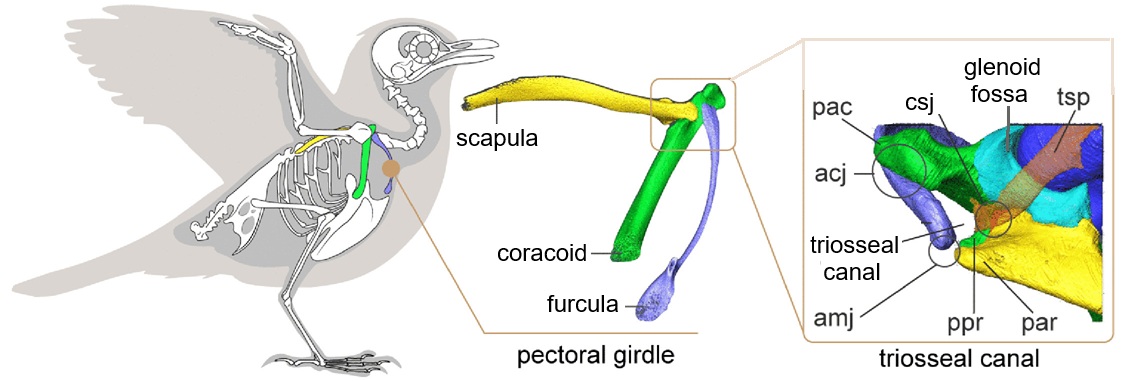
Pectoral girdle and triosseal canal of birds. Abbreviations: acj, acrocoracoclavicular joint; amj, acromioclavicular joint; csj, coracoscapular joint;
pac, acrocoracoid process; par, acromion; ppr, procoracoid process; tsp, tendon of m. supracoracoideus.
During the avian flight stroke, contraction of the supracoracoideus pulls on the tendon of the supracoracoideus (that passes through the triosseal canal) that pulls the head of the humerus upward, rapidly elevating the wing. This rapid elevation of the wing is crucial for minimizing the duration of the upstroke to limit lift loss and maximize lift production during the downstroke. However, morphological studies evaluating how the pectoral girdle relate to the evolution of flapping flight in dinosaurs have yet to focus on the evolution of the triosseal canal. In Archaeopteryx, the most basal avian, the pectoral girdle had no triosseal canal, suggesting very limited flight abilities.
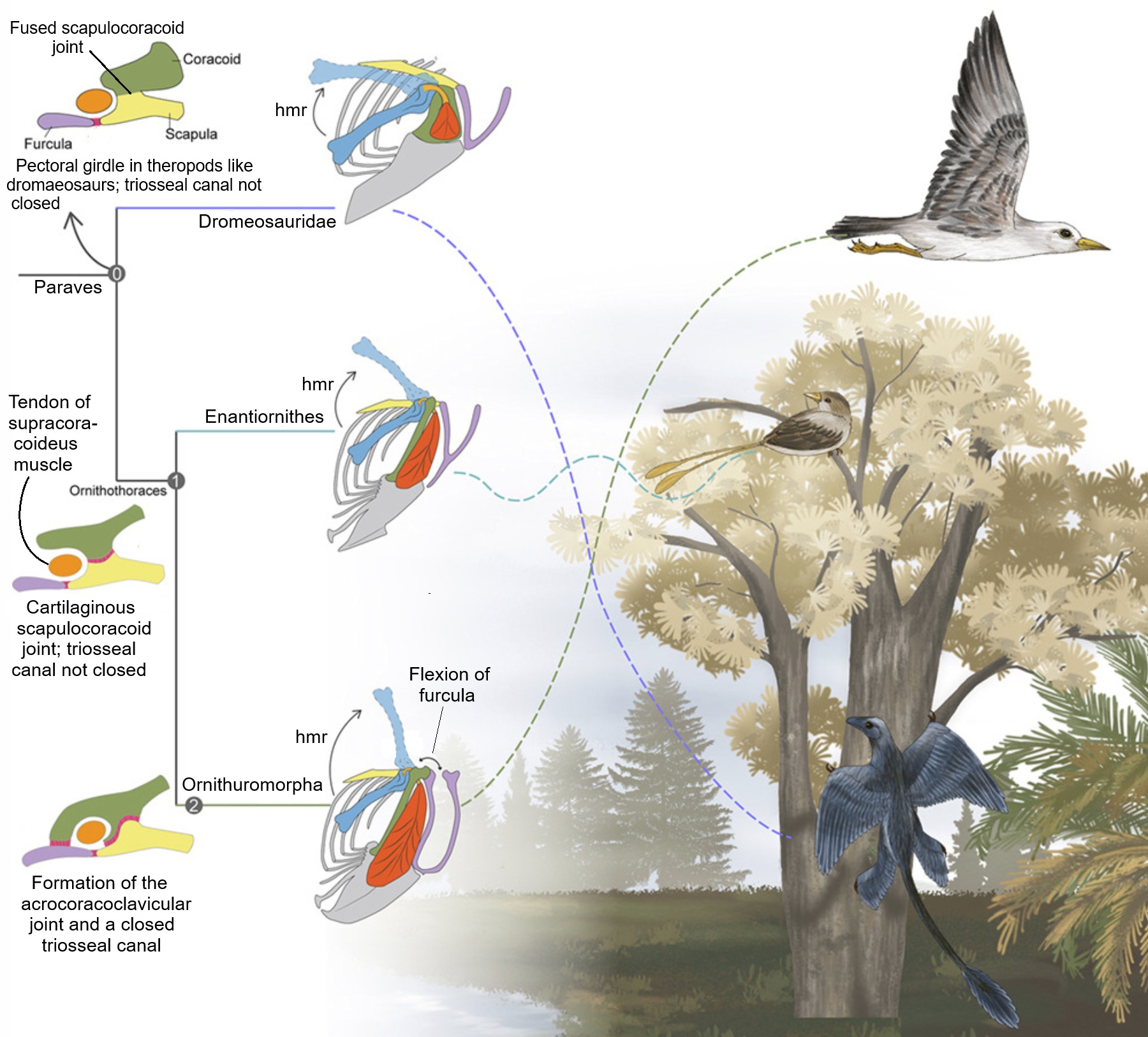
Hypothesized flight styles of dromaeosaurs and enantiornithes compared to that of present-day birds (Ornithuromorpha). The sterna
(and flight muscles) of dromaeosaurs were likely too small to sustain neornithine-like flight so may have been capable of gliding.
Enantiornithes, with larger sterna and flight muscles, may have been intermittent fliers (e.g., employing bounding or flapping-gliding
flight). Early ornithuromorphs, with enlarged sterna, well-developed keel and larger flight muscles, developed superior flapping
capacity early in their evolution enabling ground (or water surface) takeoff comparable to that of present-day birds. Their enhanced
flight performance may have served as a key evolutionary breakthrough, allowing ornithuromorphs to radiate into diverse terrestrial
and aquatic habitats. Yet another factor contributing to differences in the flight styles of early Cretaceous birds and present-day birds
was the evolution of the acrocoracoclavicular joint - a key evolutionary innovation that likely enhanced the powered flight capabilities
of early ornithuromorphs. In crown birds, the furcula flexes during the flight stroke, which may improve flapping efficiency. The
acrocoracoclavicular joint not only facilitates furcular flexion, but also completes the triosseal canal, a hallmark of the neornithine
flight apparatus. Although a pulley-like tendon system exists in some non-ornithuromorph birds (Jeholornis, dromaeosaurs, and
enantiornithines), their canal was partially open. The enclosed triosseal canal in ornithuromorphs may stabilize the tendon during the
contraction of the supracoracoideus, ensuring precise control of the humeral elevation and greater wing motion (elevation-depression).
hmr, hypothesized movement range of the humerus (From: Wu et al. 2026).
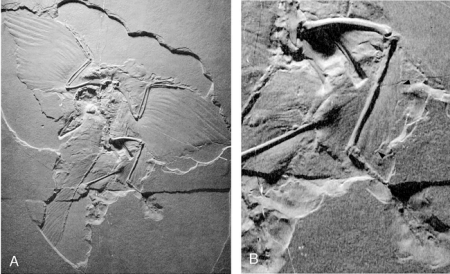
The Berlin Archaeopteryx. In the earliest cast of the main slab (A), long hindlimb feathers are visible (B) (Longrich 2006).
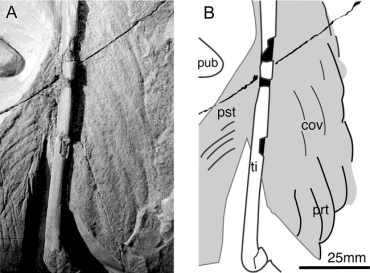 Berlin Archaeopteryx. A, Plumage of the right hindlimb. B, Schematic drawing. Abbreviations: cov, covert feathers; prt, pretibial feathers; pst, shafts of post-tibial feathers; pub, pubis; ti, tibia (Longrich 2006). |
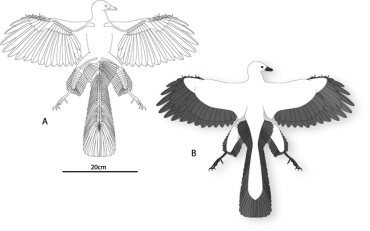 Berlin Archaeopteryx. A, Reconstruction. B, Life restoration. The hindlimbs have been abducted to 90° so as show the area of the leg plumage. The area of the hindlimbs was measured distal to the body contour and proximal to the ankle (Longrich 2006). |
Support for the arboreal hypothesis? -- Feathers cover the legs of the Berlin specimen of Archaeopteryx lithographica, extending from the cranial surface of the tibia and the caudal margins of both tibia and femur. These feathers exhibit features of flight feathers rather than contour feathers, including vane asymmetry, curved shafts, and a self-stabilizing overlap pattern. Many of these features facilitate lift generation in the wings and tail of birds, suggesting that the hindlimbs acted as airfoils. Longrich (2006) presented a new reconstruction of Archaeopteryx where the hindlimbs formed approximately 12% of total airfoil area. Depending upon their orientation, the hindlimbs could have reduced stall speed by up to 6% and turning radius by up to 12%. The presence of the “four-winged” planform in both Archaeopteryx and basal Dromaeosauridae indicates that their common ancestor used both forelimbs and hindlimbs to generate lift. The presence of flight feathers on the hindlimbs would seem to be inconsistent with the cursorial hypothesis and the wing-assisted incline running hypothesis; in these scenarios, such a specialization would serve no purpose, and would impede locomotion. The evidence presented by Longrich (2006), therefore, supports an arboreal origin of avian flight, and suggests that arboreal parachuting and gliding preceded the evolution of avian flight.
Evolution of flight: a summary -- Although the timing remains unclear, the first step toward the evolution of flight involved a reduction in size, with their ancestors decreasing in size during the Triassic and well before the evolution of birds and flight. Endothermy must have evolved sometime between the early Late Triassic, when dinosaurs first appeared in the fossil record and the evolution of modern birds whose ancestors first appeared in the early Late Jurassic. More specifically, coelurosaurs, a diverse group of dinosaurs that likely included the ancestors of birds, exhibited substantial and sustained morphological transformation and this rapid evolution of skeletal diversity may indicate rapidly changing selection pressures as a result of radiation into new ecological niches. The evolution of endothermy may have been more likely in lineages, such as the smaller coelurosaurs, exposed to new selection pressures rather than in more conservative, larger-bodied, lineages (Schluter 2001). For example, the body temperatures of small dinosaurs (< 100 kg) that lived at mid-latitudes (45-55°) or higher would have been well below 30°C during winter if they were crocodile-like ectotherms (Seebacher 2003). Selection pressures for morphological and physiological thermoregulatory adaptations would likely have been strongest in such dinosaurs. Of course, without insulation, the thermoregulatory advantages gained from elevated resting metabolic rates would be limited. Most skin impressions from dinosaurs indicate the presence of naked skin (Sumida and Brochu 2000), except for integumentary structures in coelurosaurs that may have afforded thermal insulation (Chen et al. 1998). Although other dinosaurs may have possessed integumentary structures with insulatory qualities, current evidence suggests that these evolved only in coelurosaurs. The earliest known feathers stem from the Late Jurassic, so if those feathers possessed insulating qualities, true endothermy may have evolved sometime after that (Seebacher 2003).
By the time Archaeopteryx arrived on the scene, therefore, birds obviously had the basic features needed for flight – relatively small with feathers and, if not truly endothermic, then, at minimum, an elevated metabolism. The question then is how the ancestors of Archaeopteryx, with the necessary characteristics, first took to the air. Several hypotheses have been proposed. Primary among them are the arboreal hypothesis (e.g., Feduccia 1996), with the ancestors of Archaeopteryx living in trees (or at least climbing into trees on a regular basis) and initially gliding before developing flapping flight, and the cursorial hypothesis (e.g., Burgers and Chiappe 1999), with these ancestors using long, powerful legs to run fast with their arms (wings) outstretched and, eventually, developing sufficient lift to take flight.
An additional hypothesis is the WAIR (wing-assisted incline running) hypothesis. Dial (2003) noticed the ability of young Chukars to escape danger by scrambling up inclined surfaces. The chicks first run very fast, flapping their rather small, partially feathered wings to creating enough momentum to run up a inclined surface to safety. The ancestors of birds may have using proto-wings in a similar fashion, with wings eventually evolving to the point of permitting not only running up inclined surfaces but, for an animal running across the ground, flight.
There is currently no clear concensus in support of any of these hypotheses for the origin of bird flight. However, a four-winged dromaeosaur (Microraptor gui) from the early Cretaceous of China provides evidence for an arboreal-gliding origin of avian flight. It had asymmetric flight feathers not only on the forelimb, but on the hindlimb as well. Chatterjee and Templin (2007) proposed that the wings of Microraptor could have resembled a staggered biplane configuration during flight, where the forewing formed the dorsal wing and the hindwing formed the ventral one. The contour feathers on the tibia fo the hindlimb were positioned posteriorly, oriented in a vertical plane for streamlining that would reduce the drag considerably. Leg feathers are present in many fossil dromaeosaurs, early birds, and living raptors, and they play an important role in flight during catching and carrying prey. A computer simulation of the flight performance of Microraptor suggested that its biplane wings were adapted for undulatory "phugoid" gliding between trees, where the horizontal feathered tail offered additional lift and stability and controlled pitch. Thus, Microraptor, a gliding relative of early birds, apparently took to the air with two sets of wings.
In further support of the arboreal hypothesis, feathers also cover the legs of the Berlin specimen of Archaeopteryx lithographica, extending from the cranial surface of the tibia and the caudal margins of both tibia and femur. These feathers exhibit features of flight feathers rather than contour feathers, including vane asymmetry, curved shafts, and a self-stabilizing overlap pattern. Many of these features facilitate lift generation in the wings and tail of birds, suggesting that the hindlimbs acted as airfoils. Longrich (2006) presented a new reconstruction of Archaeopteryx where the hindlimbs formed approximately 12% of total airfoil area. Depending upon their orientation, the hindlimbs could have reduced stall speed by up to 6% and turning radius by up to 12%. The presence of “four-wings” in both Archaeopteryx and basal Dromaeosauridae suggests that their common ancestor used both forelimbs and hindlimbs to generate lift. The evidence presented by Longrich (2006), therefore, supports an arboreal origin of avian flight, and suggests that arboreal parachuting and gliding likely preceded the evolution of avian flight just as it apparently did in the evolution of flight in bats (Speakman 2001) and pterosaurs (Naish and Martill 2003).

Archaeopteryx (Source: Nick Longrich)
Although the presence of flight feathers on the hindlimbs would seem to support the arboreal hypothesis for the origin of flight, such feathers do not necessarily indicate that Archaeopteryx and its immediate ancestors were strictly tree-dwellers. Many present-day birds spend time both in trees and on the ground and Archaeopteryx likely did the same. With hindlimb feathers, as well as flight muscles less developed than those of present day birds, Archaeopteryx may have found it difficult, if not impossible, to take off directly from the ground. So, to take flight, Archaeopteryx and its ancestors likely sought elevated perches like trees for ‘launching.’ In doing so, they may very well have used wing-assisted incline running just like some present-day birds. For example, several petrels are known to climb trees to launch themselves into the air (del Hoyo et al. 1992), and, for some seabirds, the presence of ‘take-off trees’ is important in selection of breeding habitat (Sullivan and Wilson 2001).
Neurological evidence supports the idea that Archaeopteryx was a rather accomplished flyer. Reconstruction of the braincase and inner ear of Archaeopteryx revealed strong similarities to present-day birds, with areas of the brain involved in hearing and vision enlarged and an enlarged forebrain that would enhance the rapid integration of sensory information required in a flying animal (Alonso et al. 2004).
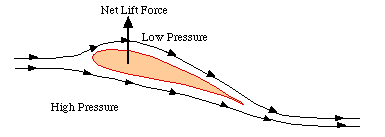
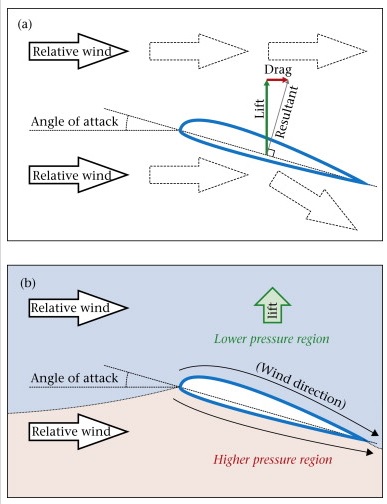
Flight requires lift, which occurs because wings move air downwards. Lift is created only when air strikes a wing at an angle (i.e., the angle of attack). When the leading edge of a wing is higher than the trailing edge, the bottom of the wing 'pushes' the air forward and creates an area of high pressure below and ahead of the wing. At the same time, air is deflected downward so, because of Newton's Third Law of Motion (for every action there is an equal and opposite reaction), the wing is deflected upward. Both the upper and lower surfaces of the wing deflect the air. The upper surface deflects air down because the airflow “sticks” to the wing surface and follows the tilted wing (the “Coanda effect”).
Because of inertia, air moving over the top of the wing tends to keep moving in a straight line while, simultaneously, atmospheric pressure tends to force air against the top of the wing. The inertia, however, keeps the air moving over the wing from 'pushing' against the top of the wing with as much force as it would if the wing wasn't moving. This creates an area of lower pressure above the wing. Because air tends to move from areas of high pressure to areas of low pressure, air tends to move from the high pressure area below and ahead of the wing to the lower pressure area above and behind the wing. This air moves, therefore, toward the trailing edge of the wing, or the same direction as the airflow created by the wing's motion. As a result, air flows faster over the top of the wing. Because air under the wing is dragged slightly in the direction of travel, it moves slower than does the air moving over the top of the wing. Thus, air is flowing slower beneath the bottom of the wing. The faster-moving air going over the top of the wing exerts less pressure than the slower-moving air under the wing and, as a result, the wing is pushed upwards by the difference in pressure between the top and the bottom (the Bernoulli effect). So, both the development of low pressure above the wing (Bernoulli's Principle) and the wing's reaction to the deflected air underneath it (Newton's third Law) contribute to the total lift force generated.

Note that air, both above AND below the wing, is deflected downward.

Why does the slower moving air generate more pressure against the wing than the faster moving air? In calm air, the molecules are moving randomly in all directions. However, when air begins to move, most (but not all) molecules are moving in the same direction. The faster the air moves, the greater the number of air molecules moving in the same direction. So, air moving a bit slower will have more molecules moving in other directions. In the case of a wing, because air under the wing is moving a bit slower than air over the wing, more air molecules will be striking the bottom of the wing than will be striking the top of the wing.
;
How does a wing work?

Clear example of how wings deflect air downward. Notice the trough formed in the clouds.
(Source: http://www.grc.nasa.gov)
Airfoil
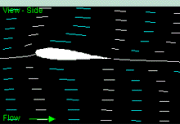
How do airfoil shape, camber, and angle of attack influence lift?
Click on this image!
Lift is generated in two ways. (a) Air is deflected downward when
there is a positive angle of attack (Newtonian lift). (b) When air
moves faster over the top of the wing than the bottom, the pressure
diference (lower on the top of wing) generates Bernoulli lift
(Figure from Bajec and Heppner 2009).
When the curvature over the top becomes greater by increasing
the angle of attack (below), the amount of lift generated increases because the force with which the wing is pushed upward increases. Eventually, however, if the angle
of attack becomes too great, the flow separates off the wing and less
lift
is generated. The result is stalling. Birds also tend
to
stall at low speeds because slower moving air may not move smoothly
over
the wing.

If the angle of attack is too great, air flow over the
top of the wing may become more turbulent & the result is less lift.
Flying from an eagle's point of view
A 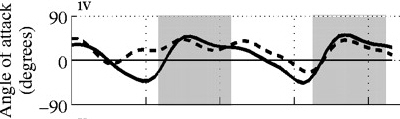 |
C 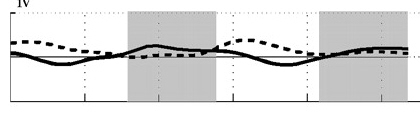 |
B 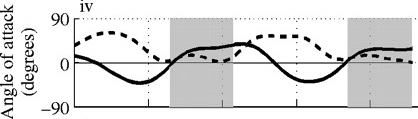 |
D 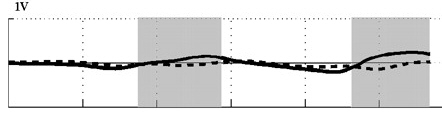 |
Angle of attack decreases with increasing speed. Angle of attack during two wingbeats of a Ringed Turtle-Dove (Streptopelia risoria) flying at 1 meter/sec (A), 5 meters/sec (B),
9 meters/sec (C), and 17 meters/sec (D). Angle of attack at low speeds peaked at 52 degrees (proximal wing) and 43 degrees (distal wing), much
greater than those commonly used by aircraft (0-15 degrees). At faster speeds, mean angle of attack decreased to 9-14 degrees (proximal wing) and -5-14 degrees
(distal wing), within the range employed by aircraft. Shaded areas indicate downstroke; solid line = distal wing & dashed line = proximal wing (Hedrick et al. 2002).
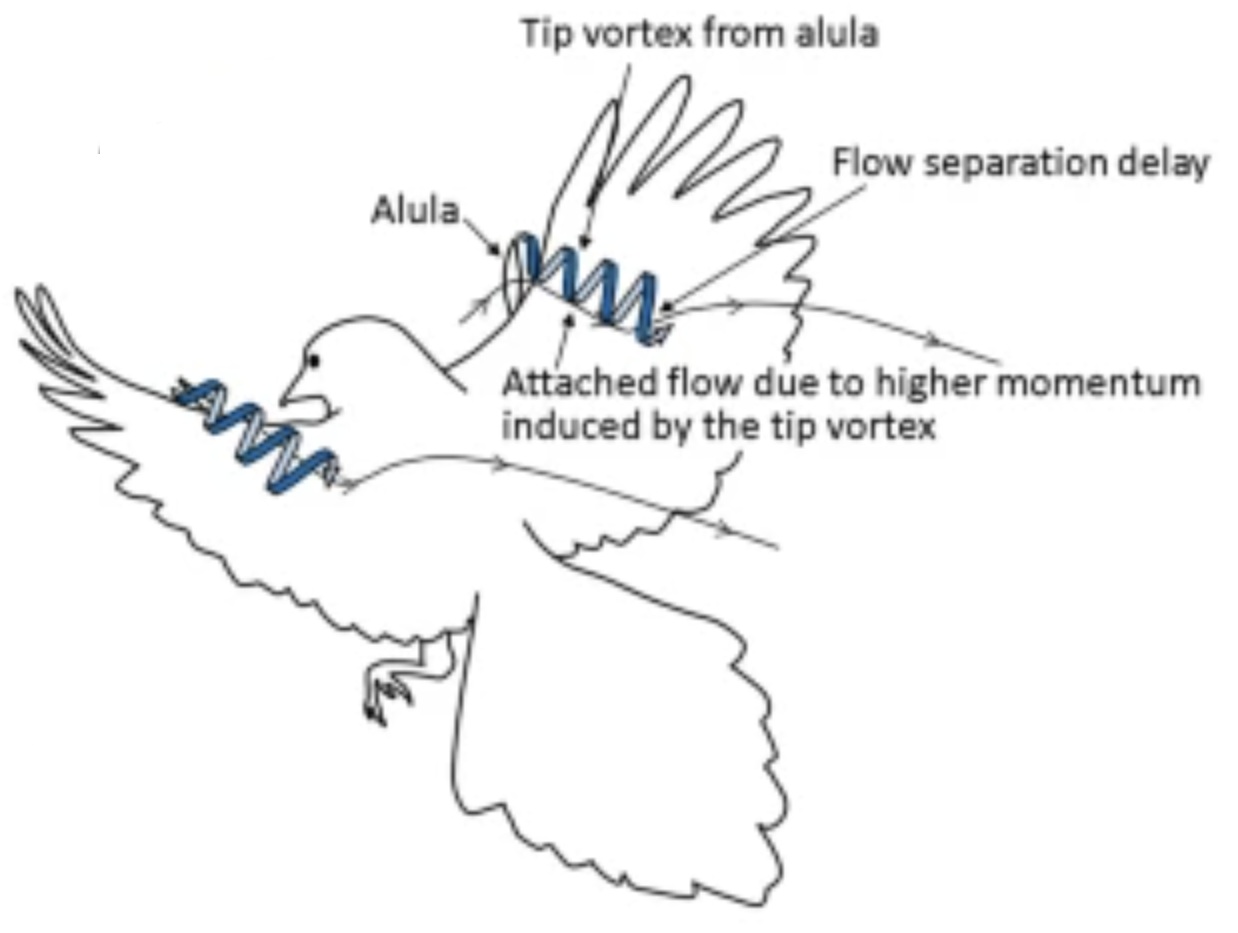
At low speeds (such as during take-off & landing), birds can maintain smooth air flow over the wing (and, therefore, maintain lift) by using the alula (also called the bastard wing). The alula is formed by feathers (usually 3 to 5) attached to the first digit.
 Source: http://courses.egr.duke.edu/PROJECTS/Modules/mammals/lesson/wingskeleton.html |
 Source: http://www.windsofkansas.com/fltphoto.html |
When these feathers are elevated (above right & below right), they keep air moving smoothly over the wing & help a bird maintain lift.
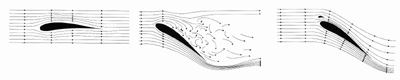
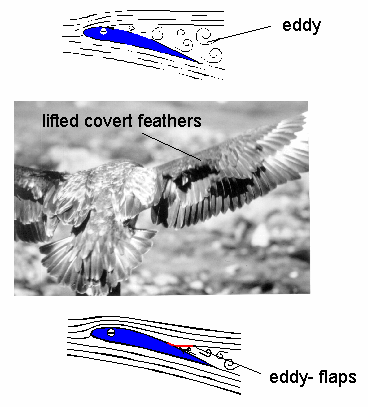 At increasing angles of attack, an eddy starts to propagate from the trailing edge towards the leading edge of the wing. As a result, air flowing over the top of the wing separates from the upper surface and lift is lost. However, when coverts are lifted upward by the eddy, they prevent the spread of the eddy and work as 'eddy-flaps.' 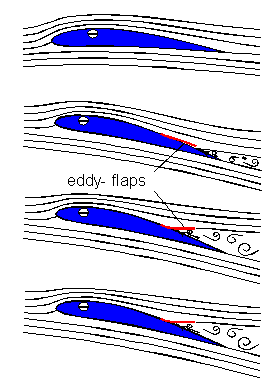 The 'covert eddy-flaps', by preventing the spread of the eddy toward the leading edge of the wing, help maintain lift (i.e., prevent stalling) at high angles of attack, e.g., when taking off or landing. |
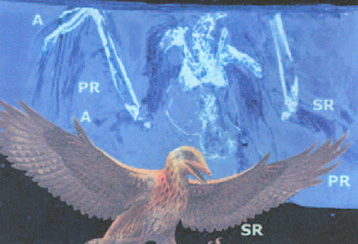 Eoalulavis hoyasi. Top, fluorescence induced ultraviolet photo of the specimen before preparation. Bottom, reconstruction. A - alula, PR - primary remiges, and SR - secondary remiges (Sanz and Ortega 2002). |
The fossilized remains of a tiny bird
provides
evidence that birds flew as nimbly 115 million years ago as their
descendants
do today. The fossilized bird, Eoalulavis hoyasi, was found in
a
limestone quarry in Spain (Sanz et al. 1996). About the size of a
goldfinch, the bird had an alula, or bastard wing, that would have
helped
it stay aloft at slow speeds. Eoalulavis is the most primitive
bird
known with an alula.
Archaeopteryx probably flapped and glided, but did not have an alula. Eoalulavis provides evidence that by 30 million years after Archaeopteryx, at least one group of early birds had developed the alula. Eoalulavis hoyasi, which means "dawn bird with a bastard wing from Las Hoyas," was discovered at a site where a freshwater lake existed millions of years ago. The bird may have hunted by wading in shallow water the way plovers and other shorebirds do today. |
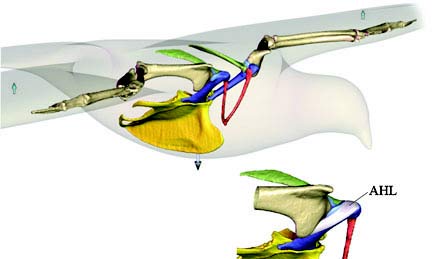
Linchpin of flight
Using computer modeling, treadmills and the fossil record, researchers have shown that the acrocoracohumeral ligament (AHL), a short band of tissue
that connects the humerus to the shoulder joint in birds, was a critical element in the evolution of flight (Image: David Baier/Brown University).
To find out if this ligament played the same shoulder-stabilizing role in primitive animals, they looked to the alligator. Alligators are close relatives of birds and both are archosaurs, the “ruling reptiles” that appeared on the planet some 250 million years ago and evolved into the dinosaurs that dominated during the Mesozoic Era. So to understand the sweep of evolution, the alligator was a great starting place. In the lab, three alligators were put on motorized treadmills and X-ray videos were made. The video was used to make a 3D computer animation that showed the precise positioning of the shoulder as the animal walked. They found that alligators use muscles – not ligaments – to do the hard work of supporting the shoulder. Then Baier studied the skeleton of Archaeopteryx lithographica, and even traveled to Beijing to examine the fossilized remains of Confuciusornis, Sinornithoides youngi and Sinornithosaurus millenii, close relatives of modern birds recently discovered in China.
If the acrocoracohumeral ligament was critical to the origin of flight, Baier expected to find evidence of it in Archaeopteryx. Surprisingly, however, the new ligament-based force balance system appears to have evolved more gradually in Mesozoic fliers. “What this means is that there were refinements over time in the flight apparatus of birds,” Baier said. “Our work also suggests that when early birds flew, they balanced their shoulders differently than birds do today. And so they could have flown differently. Some scientists think they glided down from trees or flapped off the ground. Our approach of looking at this force balance system can help us test these theories.”
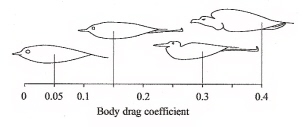
| Of course, a bird moving through the air is opposed by friction & this is called drag. The types of drag acting on birds are parasitic drag, pressure (or induced) drag, & friction (or profile) drag. Parasitic drag is caused by friction between a bird’s body and the air (and is termed parasitic because the body does not generate any lift). Induced drag occurs when the air flow separates from the surface of a wing, while friction drag is due to the friction between the air and bird moving through the air. Friction drag is minimized by a wing's thin leading edge (wings 'slice' through the air). Induced drag occurs at low speeds and at higher speeds as, at wing tips, air moves from the area of high pressure (under the wing) to the area of low pressure (top of the wing). As wings move through the air, this curling action causes spirals (vortices) of air (see photo of continuous vortices to the right) which can disrupt the smooth flow of air over a wing (and reduce lift). |  |

A 'smoke angel' created after flares were released and caused by wingtip vortices (Photo source: US Air Force).
Induced drag
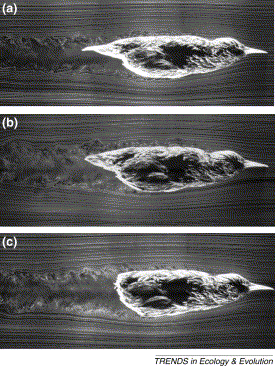 |
Bird tails & flight -- Most birds have rather short triangular tails when spread. In flight, the tail is influenced by the time-varying wake of flapping wings and the flow over the body. It is reasonable to assume that body, wings and tail morphology have evolved in concert. Modelling the interaction between the wings and tail suggests that the induced drag of the wing–tail combination is lower than that for the wings alone. A tail thus enables the bird to have wings that are optimized for cruising speed (with the tail furled to minimize drag) and, at low speeds, the spread tail reduces induced drag during manoeuvring and turning flight. Observations show that tails are maximally spread at low speeds and then become furled increasingly with increasing speed (Hedenström 2002). Figure to the left. Flow visualization around mounted wingless starling bodies using the smoke-wire technique in a wind tunnel at 9 ms−1. (a) The bird with intact tail and covert feathers; (b) tail feathers protruding beyond ventral coverts are trimmed to the same length as coverts; (c) tail feathers, ventral and dorsal covert feathers removed. The height of the wake increases from (a) to (c). The dorsal boundary layer also becomes increasingly turbulent in (b) and (c) compared with the intact tail-body configuration in (a). From: Hedenström (2002). |
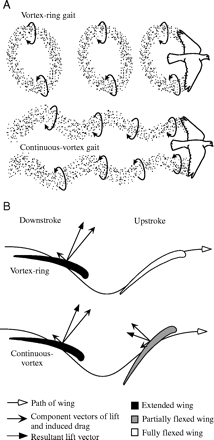 |
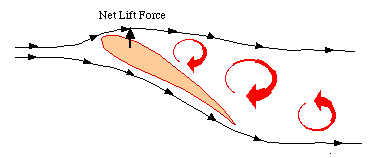
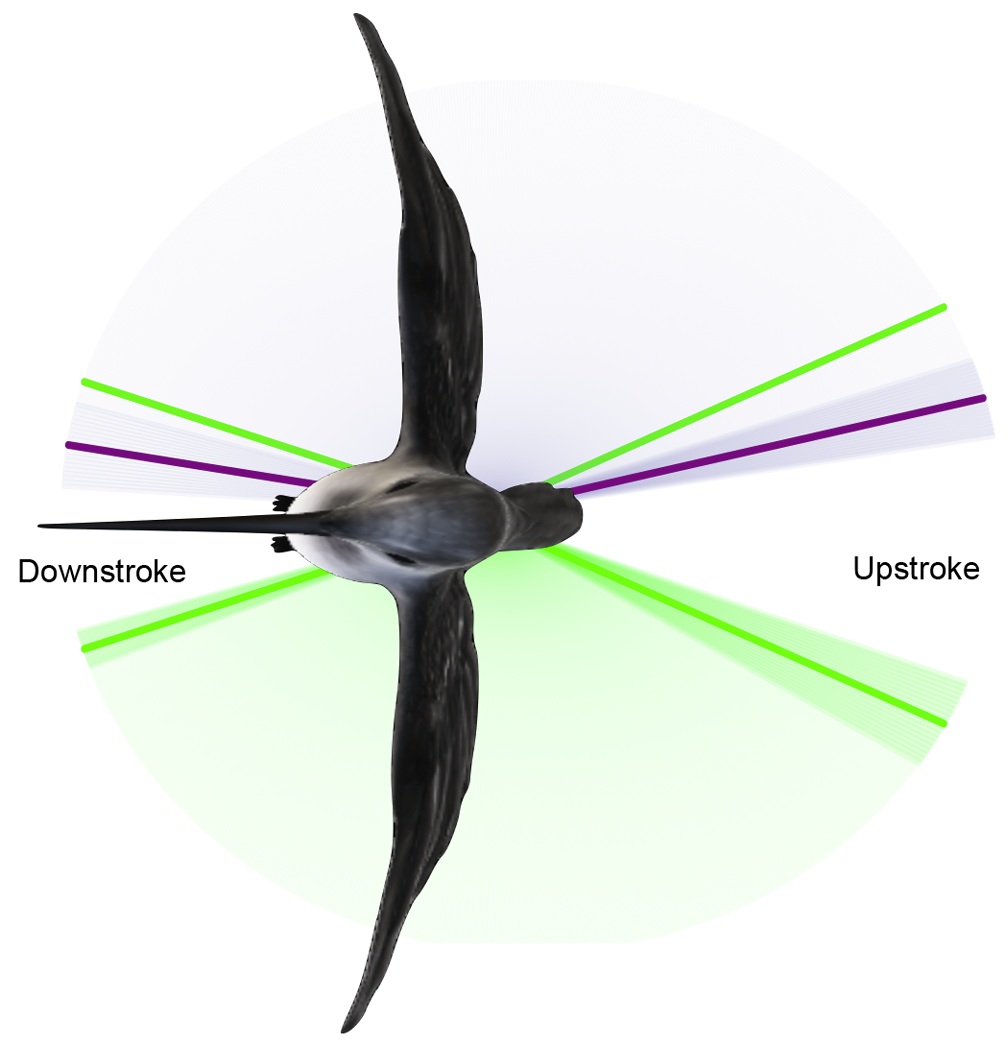
(A) Depictions of the vortex-ring and continuous-vortex gaits. (B) Cross-sectional view of the wing profile. Lift produced during flapping provides weight support (upward force) and thrust (horizontal force). In the vortex-ring gait, lift is produced only during the downstroke, providing positive upward force and forward thrust. In the continuous-vortex gait, lift is produced during both the upstroke and the downstroke. The downstroke produces a positive upward force and forward thrust; the upstroke produces a positive upward force and rearward thrust. Partial flexion of the wing during the upstroke reduces the magnitude of the rearward thrust to less than that of the forward thrust produced during the downstroke, providing net positive thrust per wingbeat (From Hedrick et al. 2002). |
| Birds are known to employ two different gaits in flapping flight, a vortex-ring gait in slow flight and a continuous-vortex gait in fast flight. In the vortex ring gait, the upstroke is aerodynamically passive (there is no bound circulation during this phase, and hence no trailing vortex), and the wings flex and move close to the body to minimize drag. In the continuous vortex gait (where each wingtip sheds a separate vortex trail during both the upstroke and downstroke), the wings are aerodynamically active throughout (i.e., lift is generated both during the downstroke and the upstroke), while the wings remain near-planar throughout and deform only by flexure at the wrist. Hedrick et al. (2002) studied the use of these gaits over a wide range of speeds in Cockatiels and Ringed Turtle-doves trained to fly in a wind tunnel. Despite differences in wing shape and wing loading, both species shifted from a vortex-ring to a continuous-vortex gait at a speed of 7 meters/sec. They found that the shift from a vortex-ring to a continuous-vortex gait depended on sufficient forward velocity to provide airflow over the wing during the upstroke similar to that during the downstroke. This shift in flight gait appeared to reflect the need to minimize drag and produce forward thrust in order to fly at high speed. |
 |
 |
Flow visualization images by helium-bubble multi-flash photography (top) and sketch of vortex wake (below)
as reconstructed by stereophotogrammetry, for the vortex ring gait of a slow-flying Rock Pigeon (Columba livia; left),
and for the continuous vortex gait of a European Kestrel (Falco tinnunculus; right) in cruising flight (Rayner and Gordon 1997).
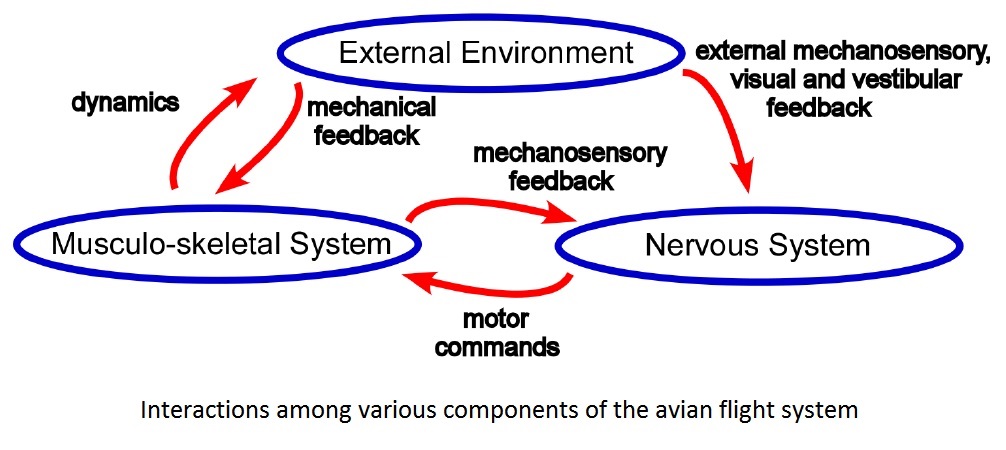
The amount of drag varies with a bird's mass (increased mass = increased friction drag), shape, & speed, and with a wing's surface area & shape.
Increased streamlining (e.g., no trailing legs and extended head)
reduces drag (Pennycuick et al. 1996).
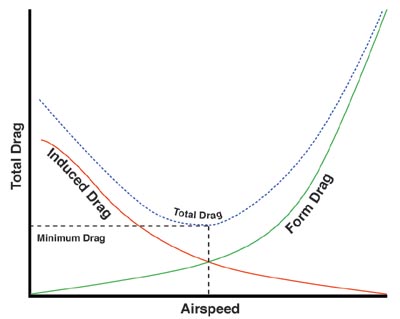
Form drag = parasitic drag + friction drag
(figure from http://en.wikipedia.org/wiki/Parasitic_drag#mediaviewer/File:Drag_Curve_2.jpg)
As described below, some wing shapes help to reduce induced drag. Wing shapes vary substantially among birds:
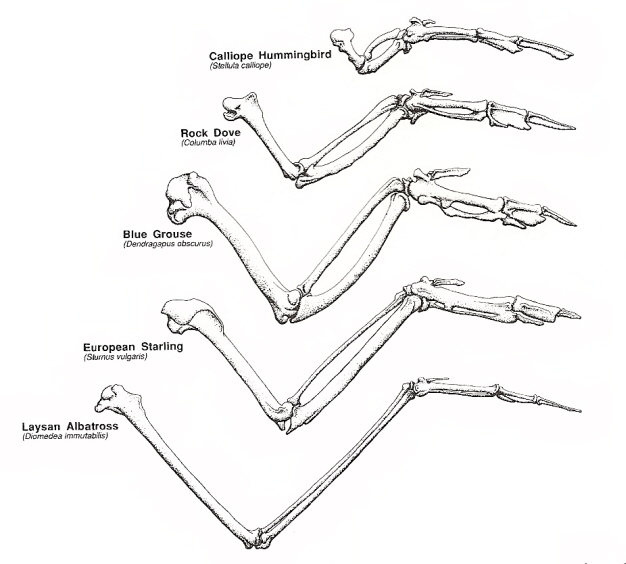
Skeletal elements of the wing of five species of birds scaled so the carpometacarpi are of equal length (Dial 1992).
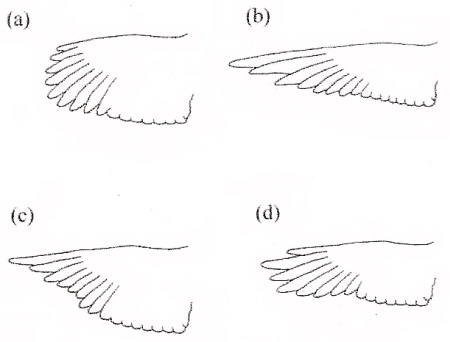 Theoretical wings that illustrate extremes of pointedness (shift in wingtip toward the leading edge) and convexity (decrease in acuteness of the wingtip). (a) rounded (low aspect ratio) and (b) pointed (high aspect ratio) wings; (c) concave and (d) convex wings (From: Lockwood et al. 1998). |
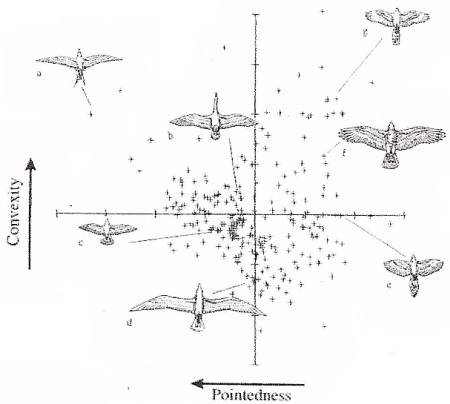 Distribution of species in terms of wing pointedness and convexity. Each point represents one species. a. tern, b. duck, c. pigeon, d. gull, e. magpie, f. buzzard (soaring hawk), and g. sparrowhawk (accipiter) (From: Lockwood et al. 1998). |
Aspect ratio affects the relative magnitude of induced and profile drag; if mass, wing area, and other wing shape parameters remain constant, a long, thin high-aspect ratio wing reduces the cost of flight and extends range. However, high aspect ratio is not necessarily associated with high speed (favored by smaller wings). Elliptical wings (low aspect ratio) can maximize thrust from flapping, whereas as more pointed wing (high-speed) with a sharp wingtip minimizes wing weight and wing inertia. Short wings must be flapped at high frequency to provide sufficient thrust. So, relatively short, pointed wings allow rapid wing-beats with reduced inertia and that translates into greater speed (e.g., shorebirds, auks, and ducks). More rounded (convex) wings produce more lift toward the wingtip (where the wing moves faster) and are particularly effective for birds that fly at slow speeds (e.g., taking off from the ground) or need high levels of acceleration. Many small passerines often fly slowly or in 'cluttered' habitats, or need rapid acceleration to escape predators. The same is true for birds like accipiters and corvids (crows and jays; Lockwood et al. 1998).
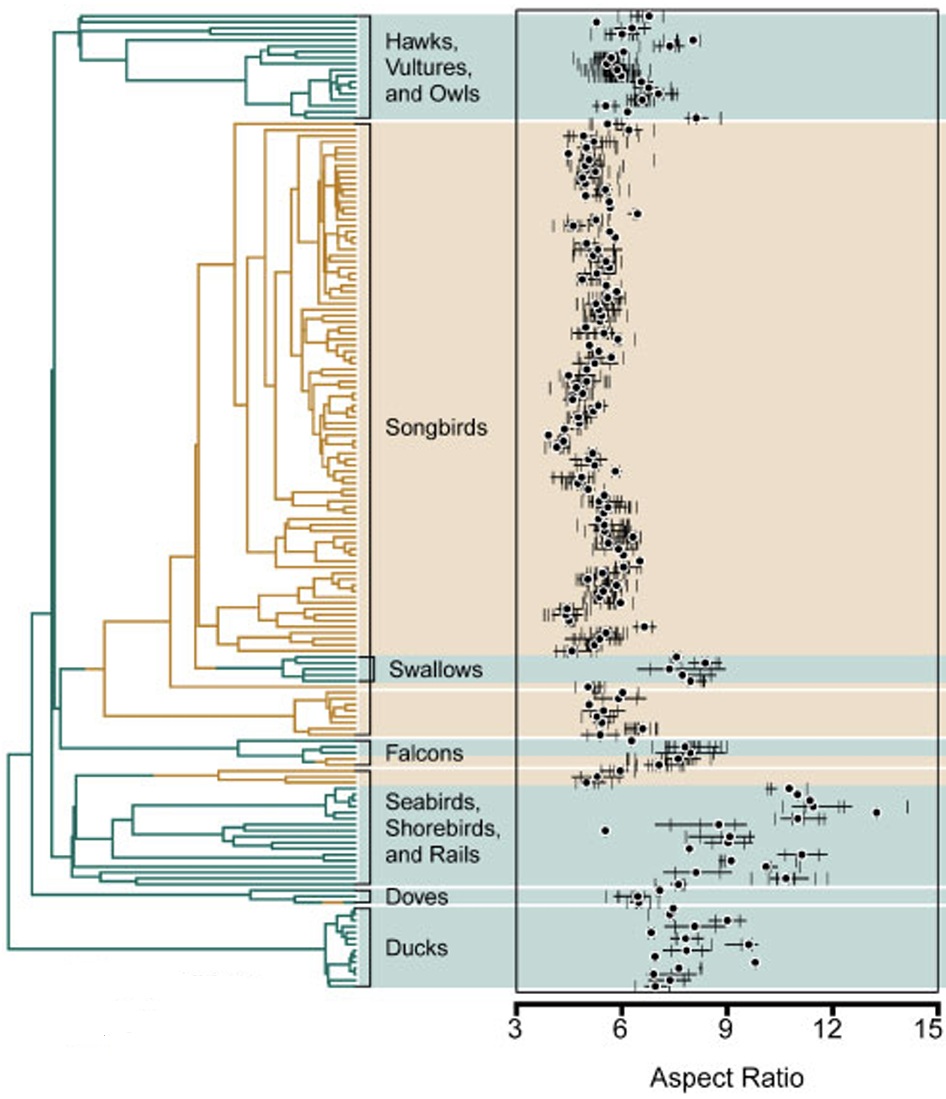
Aspect ratios of several kinds of birds. Rader et al. (2020) scanned 1094 wings of 163
species of birds in 30 major lineages of birds. Aspect ratios varied from 3.90 to 13.26. Note that
swallows, falcons, and some seabirds and shorebirds, species with either high-speed wings
or soaring wings (seabirds) had relatively high aspect ratios compared to most other speices of birds.

Laysan Albatross wing (Source: http://www.ups.edu/biology/museum/wingphotos.html)
Brown Booby in flight
| Whistle through the wing - Birds molt for a variety of reasons. Molting regulates body temperature, keeps feathers neat and waterproof and allows seasonal changes in appearance for mating or migration. However, generating new feathers uses extra energy; staying warm with less plumage uses extra energy, and flying with smaller, work-in-progress wings requires extra energy. So, not all birds molt in the same way. Ducks, swans and geese, for example, shed all their flight feathers at once and are flightless until replacements have grown. Most other birds, however, lose and renew their feathers according to a continuous, pre-programmed sequence. This sequential molting gives rise to a range of temporary feather gaps that seem to reduce take-off speed, take-off angle and level flight speed and to impede predator evasion by raising a bird's minimum turning radius. Anders Hedenstrom and Shigeru Sunada of Cambridge University estimated how the aerodynamics of flight are affected by molting (Hedenstrom & Sunada 1999). They estimated drag and lift by analyzing the fluid dynamics of symmetrical gaps in flat, rectangular model wings of various width-to-length (aspect) ratios, at a fixed angle with respect to air flow – a system that reasonably approximates a bird in gliding, but not flapping, flight. Although the effects were small, Hedenstrom and Sunada concluded that both feather gap size and position affect flight performance. Large gaps, and gaps in the middle of the wing impede aerodynamic efficiency more than small, wing-tip gaps. They also found that the detrimental effect of molt gaps increases with increasing aspect ratio. In other words a bird with short, broad wings, like a vulture, won't miss a few feathers as much as one with long, narrow wings, like an albatross. "This is of great ecological significance," they muse, "as it could help explain why large birds show relatively slow rates of molting that are associated with rather small gaps." -- Sara Abdulla, Nature Science Update |
Wilson's Storm Petrel Photo by Brian Patteson http://www.patteson.com/image2.htm |
Molt is a costly event in the annual cycle, when birds replace their feathers, including all
of their primary feathers, which, for hummingbirds, comprise most of the area of the wing. So, how does
the molting of wing feathers influence hummingbird flight? Top-view showing that molting hummingbirds
show higher stroke amplitudes. Molting hummingbirds (right wing in purple) show larger wingtip trajectories than
non-molting hummingbirds (left wing in green). Solid lines showing stroke amplitude angle average
and shades represent their standard deviation. The non-molting stroke average is also shown
in green on the right wing to facilitate comparison. Purple represents molting and green non-molting.
Diaz-Salazar et al. (2024) examined high-speed videos of three species of hummingbirds to compare the wing kinematics of
molting and non-molting hummingbirds. Analysis revealed that molting hummingbirds rotated their wings at more
acute angles during both downstroke and upstroke than non-molting hummingbirds (10° versus 20°, and 15°
versus 29°, respectively), while other flight parameters remained unchanged. These results show that hummingbirds
can sustain hovering flight and even under impressive wing area reductions by adjusting their stroke amplitudes.

Chimney Swift wing (Source: http://www.ups.edu/biology/museum/wingphotos.html)
Gyrfalcon
| How pigeons give falcons the slip -- A Peregrine Falcon dive-bombing at several hundred miles an hour to
knock a pigeon out of the sky would seem to be a study in
single-mindedness.
At those speeds, attention must be paid. But even a falcon in hot
pursuit
can become distracted. And what distracts it, is a patch of white
feathers
on the rump of an otherwise blue-gray pigeon. "The brain can be primed
by a conspicuous thing," said Alberto Palleroni. The falcon, he said,
sees
the conspicuous thing -- the white patch -- and doesn't notice the
pigeon
starting to turn away and escape. "In effect, it's a kind of a card
trick
or a ruse" on the part of the pigeon, Palleroni said.
Palleroni et al. (2005) observed more than 1,800 falcon attacks on wild pigeons over seven years. They recorded the plumage types among the pigeons and noticed that while birds with white rump patches made up 20% of the pigeon population, very few were captured by the falcons. When a Peregrine Falcon attacks a pigeon, it plunges at speeds greater than 200 miles an hour, levels off and comes upon the pigeon from behind, punching it with what amounts to a closed fist. At those speeds even a grazing blow kills the pigeon; the falcon then circles back and picks it up. The only way the slower-flying pigeon can escape is by dipping a wing, rolling and veering off. If the falcon is distracted by the white patch, it won't notice the dipping of the wing (which, being blue-gray, blends with the landscape) until it's too late. Plumage color in pigeons is an independently heritable trait, Palleroni said, meaning it is not tied to selection involving sexual or other traits. So it is highly likely that the white rump feathers are an anti-predator adaptation to high-speed attacks. Not bad for a bird that many people disdain. "The feral pigeon is an amazing balance of adaptations and success," Palleroni said. - Henry Fountain, New York Times |
 |

Rock Pigeon in flight showing its white rump.
![]()
Golden Eagle in flight
A Golden Eagle (Tilly) wears video cameras to record her flight.

Another important factor that influences a bird's flying ability is
wing loading - the weight (or mass) of a bird divided by wing area
(grams/total
wing area in square centimeters). Birds with low wing loading need less
power to sustain flight. Birds considered to be the 'best' flyers, such
as swallows & swifts, have lower wing loading values than other
birds.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
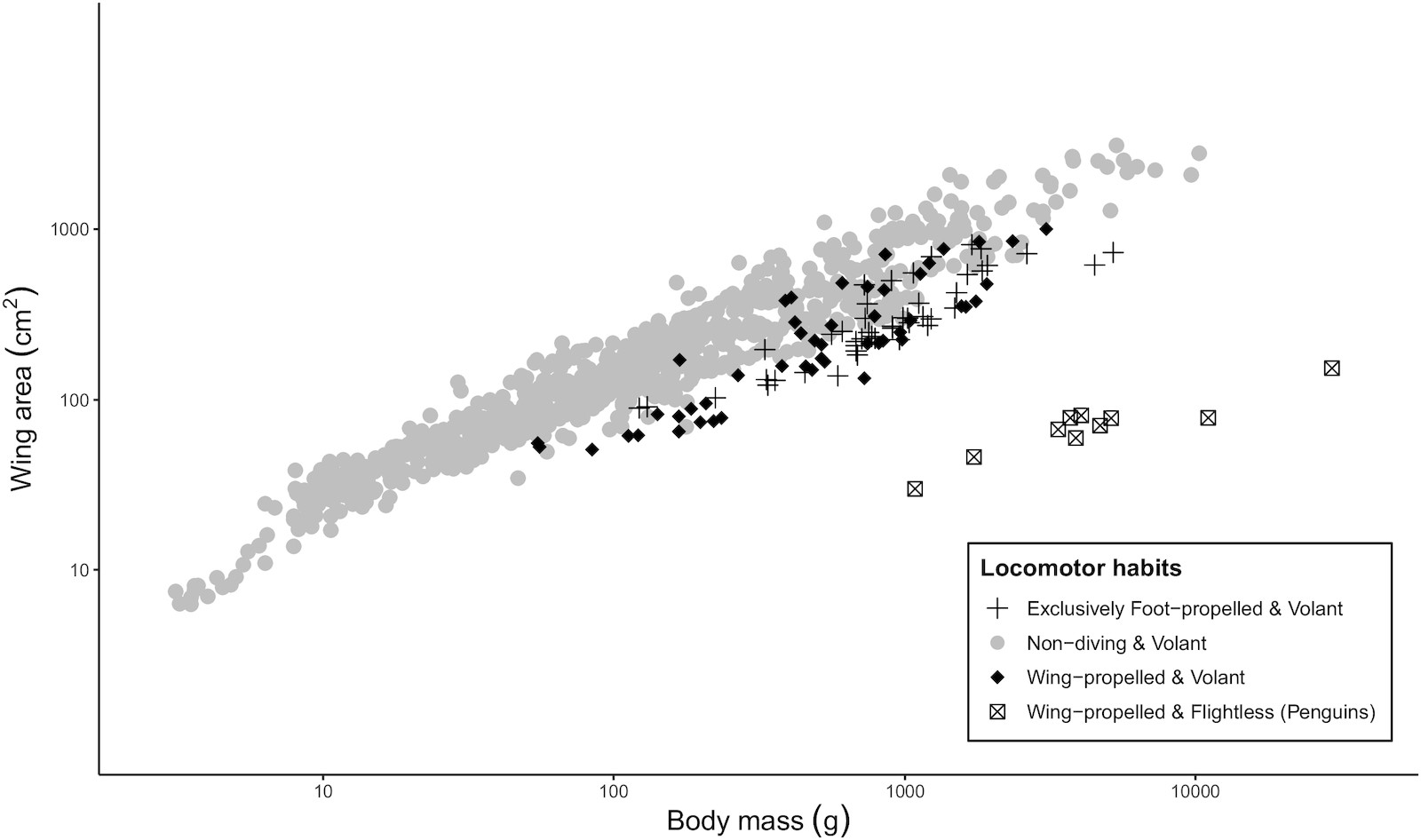
Wing area (cm2) versus body mass (g) for 951 species of birds.
The high wing-loading of diving birds is frequently associated with the challenge of using forelimbs adapted for flight for locomotion in a draggier fluid, but this does not explain why species that rely exclusively on their feet to dive (like grebes and loons) should have relatively small wings, as well. Therefore, others have hypothesized that ecological factors shared by wing-propelled and foot-propelled diving birds drive the evolution of high wing-loading. Following a reexamination of the aquatic habits of birds, we tested between hypotheses seeking to explain high wing-loading in divers using comparative data and phylogenetically informed analyses. Analysis revealed little evidence that wing-propelled diving selects for small wings, because wing-propelled and foot-propelled species share similar wing-loadings. Instead, it appears that selection to reduce buoyancy has driven high wing-loading in divers (From: Lapsansky et al. 2022).
The
Flight Strategy of Magnificent Frigatebirds -- Frigatebirds
cannot
land on the sea because their feathers are not waterproof. If they did
land, they would find it even harder to take off again because their
legs
are too short. Despite this, frigatebirds are perfectly  suited
for an aerial life over the sea because they have the lowest
wing-loading
(large wing area & low body mass) of any bird. Weimerskirch et al.
(2003) investigated the movements of Magnificent Frigatebirds (Fregata
magnificens) while foraging at sea off the coast of French Guiana.
Because they are very light in comparison to their wing surface,
frigatebirds
can glide at altitudes of up to 2,500 meters. Then they glide downward,
taking advantage of the next current. This flight strategy, which
limits
the bird's physical efforts, is the same as that used by migratory
birds
during long flights over land. Migratory birds, however, avoid flying
over
the sea due to a lack of thermals, while frigatebirds fly over the sea.
As it turns out, ascending air currents are found over the sea only in
tropical regions where the waters are warm enough to create such
currents
on a continuous basis. Frigatebirds can therefore fly night and day
using
this technique. To investigate the movements of Magnificent
Frigatebirds,
Weimerskirch et al. (2003) fitted the birds with satellite transmitters
and altimeters, which allowed them to observe that the birds only
occasionally
come close to the sea surface to catch prey. They catch flying fish or
squid driven above the surface by underwater predators like schools of
tuna or dolphins. To identify such feeding opportunities, which are
very
rare, requires long hours of flight at high altitudes. Frigatebirds
rarely
feed their young, which consequently grow very slowly. The species is,
however, well-adapted: it has a low reproductive rate and parent birds
care for their young for over one year, the longest period of parental
care of any bird. suited
for an aerial life over the sea because they have the lowest
wing-loading
(large wing area & low body mass) of any bird. Weimerskirch et al.
(2003) investigated the movements of Magnificent Frigatebirds (Fregata
magnificens) while foraging at sea off the coast of French Guiana.
Because they are very light in comparison to their wing surface,
frigatebirds
can glide at altitudes of up to 2,500 meters. Then they glide downward,
taking advantage of the next current. This flight strategy, which
limits
the bird's physical efforts, is the same as that used by migratory
birds
during long flights over land. Migratory birds, however, avoid flying
over
the sea due to a lack of thermals, while frigatebirds fly over the sea.
As it turns out, ascending air currents are found over the sea only in
tropical regions where the waters are warm enough to create such
currents
on a continuous basis. Frigatebirds can therefore fly night and day
using
this technique. To investigate the movements of Magnificent
Frigatebirds,
Weimerskirch et al. (2003) fitted the birds with satellite transmitters
and altimeters, which allowed them to observe that the birds only
occasionally
come close to the sea surface to catch prey. They catch flying fish or
squid driven above the surface by underwater predators like schools of
tuna or dolphins. To identify such feeding opportunities, which are
very
rare, requires long hours of flight at high altitudes. Frigatebirds
rarely
feed their young, which consequently grow very slowly. The species is,
however, well-adapted: it has a low reproductive rate and parent birds
care for their young for over one year, the longest period of parental
care of any bird. |
Frigatebirds
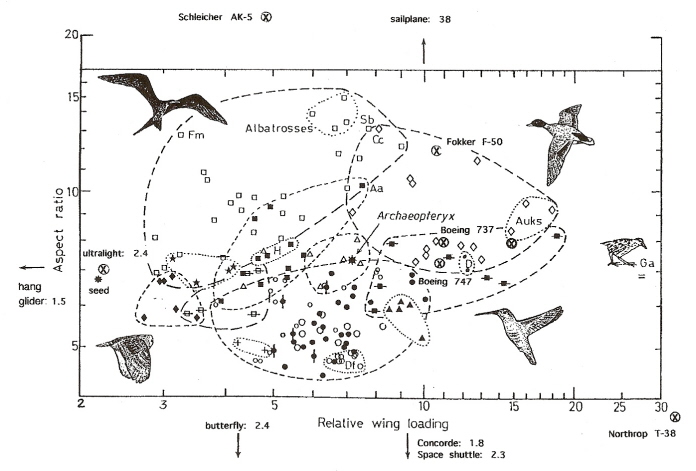
Aspect ratio vs. wing loading index in some birds, airplanes, a hang-glider, a butterfly, and a maple seed.
The numbers after various flying objects refer to aspect ratio. Fm, Fregata magnificens (Magnificent Frigatebird); Ga, Gallirallus australis (Common name: Weka;
an endemic New Zealand bird in the rail family) (From: Norberg 2002).
Different combinations of wing loading and aspect ratio permit particular flight modes and foraging strategies. Species with long wings and high aspect ratios also have low wing loadings, particularly those with low body mass, and their flight is inexpensive, e.g., many seabirds, swifts, and swallows. Birds with high wing loading and short wings, but still with high aspect ratios, are adapted to fast and rather inexpensive flight (short wings reduce profile power that is large in fast flight), e.g., loons, mergansers, geese, swans, ducks, and auks. Birds flying close to or among vegetation, e.g., flycatchers, tend to have low aspect ratios that contribute to high induced drag, but their low mass and wing loading reduce flight costs. The very low aspect ratios of many smaller birds that occupy densely-vegetated habitats, e.g., gallinaceous birds, mean that the energetic cost of flight is expensive, so these species spend much of their time walking. Birds with higher wing loading, e.g., penguins, are flightless (Norberg 2002).
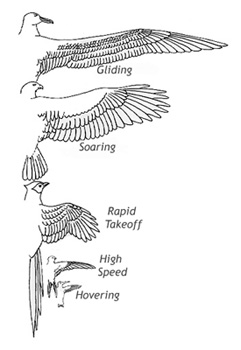
Flight styles -- Based on differences in aspect ratios and wing loading (Rayner 1988; see figure below), flight styles can also be categorized as either specialized or non-specialized. The non-specialists have average aspect ratios and average wing loading and are excellent flyers (capable of long flights and with good maneuverability) that typically use flapping flight. The non-specialists can be further subdivided, based on aspect ratio and speed, as slow non-specialists and fast non-specialists. In the slow category would be most passerines (Passeriformes), pelicans (Pelicaniformes), herons, egrets, ibises, and storks (Ciconiiformes), pigeons and doves (Columbiformes), cuckoos (Cuculiformes), most owls (Strigiformes), trogons (Trogoniformes), most birds in the order Gruiformes (e.g., gallinules, rails, and bustards), mousebirds (Coliiformes), woodpeckers (Piciformes), and parrots (Psittaciformes). Fast non-specialists include many falcons (Falconidae), gulls (Larinae), and storm-petrels (Hydrobatidae).
Birds with morphological attributes (aspect ratio and wing loading) that differ (beyond one or two standard deviations) from those of ‘typical’ birds exhibit specialized flight styles (Rayner 1988). Among these specialized styles are:
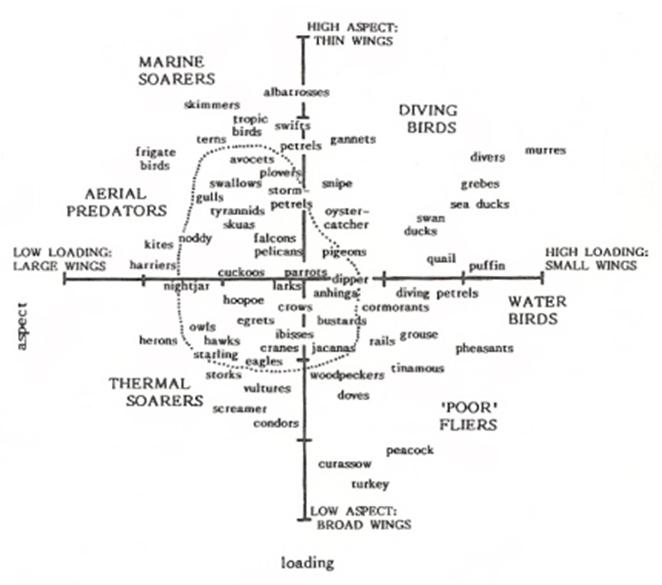
Approximate centroids of major groups of birds relative to aspect ratios and wing loading (From: Rayner 1988).

Modified version of Rayner's (1998) figure (From: http://speculativeevolution.wikia.com/wiki/File:BirdWingGraph.png)
Bird Flight Speeds (m/s) Plotted in Relation to Body Mass (kg) and Wing Loading (N/m2) for 138 Species of Six Main Monophyletic Group |
Bird flight speeds -- Alerstam et al. (2007) examined the cruising speeds of 138 different species of migrating birds in flapping flight using tracking radar. Mean airspeeds among the 138 species ranged between 8 and 23 m/s (or about 18 to 51 mph). Birds of prey, songbirds, swifts, gulls, terns, and herons had flight speeds in the lower part of this range, while pigeons, some of the waders, divers, swans, geese, and ducks were fast flyers in the range 15–20 m/s (33 - 45 mph). Cormorants, cranes, and skuas were among the species flying at intermediary speeds, about 15 m/s. The diving ducks reached the fastest mean speeds, with several species exceeding 20 m/s (and up to 23 m/s). An important factor in explaining variation in flight speed was phylogenetic group; species of the same group tended to fly at similar characteristic speeds.
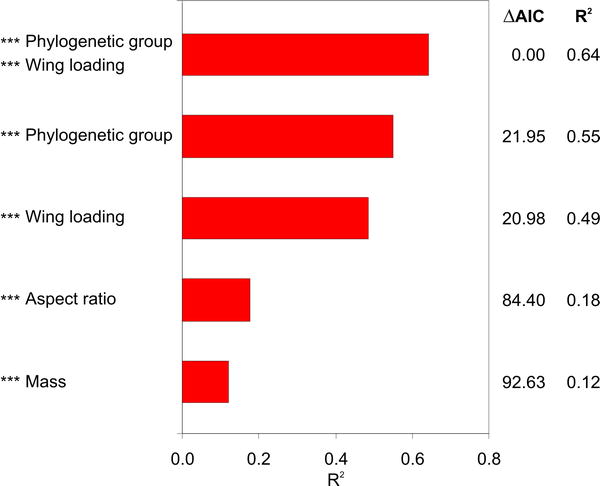
Depending on their ecological life style and foraging, birds are adapted to different aspects of flight performance, e.g., speed, agility, lift generation, escape, take-off, and energetic cost of flight.. These adaptations are likely to have implications for the flight apparatus (anatomy, physiology, and muscle operation) and the flight behavior that may constrain the cruising flight speed. Species flying at comparatively slow cruising speeds frequently use thermal soaring (raptors and storks), are adapted for hunting and load carrying (raptors), or for take-off and landing in dense vegetation (herons). Associated with these flight habits they have a lower ratio of elevator (supracoracoideus) to depressor (pectoralis) flight muscle (particularly low among birds of prey) compared with shorebirds and waterfowl. Alerstam et al. (2007) suggested that functional differences in flight apparatus and musculature among birds of different life and flight styles (differences often associated with evolutionary origin) have a significant influence on a birds performance and speed in sustained cruising flight.
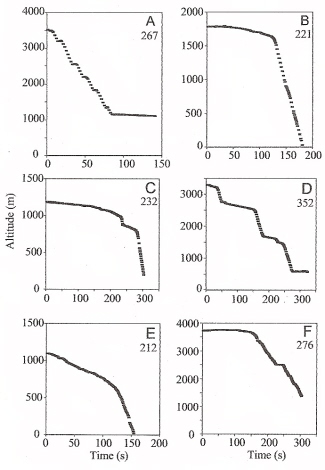 Altitude vs. time showing rapid descents during migratory flights as recorded by radar. (A) Barn Swallow, (B) Yellow Wagtail, (C) Reed Warbler, (D) Yellow Wagtail, (E) Meadow Pipit, and (F) Yellow Wagtail. |
Diving speeds -- Hedenstrom and Liechti (2001) used radar to track the flights of migrating birds as they descended from their cruising altitudes after crossing the Mediterranean Sea. Dive angles were as great as 83.5 degrees and the maximum speed recorded was 53.7 meters/sec (or about 120 miles/hour). Larger birds can attain even greater speeds, with estimates of the top speed of Peregrine Falcons as high as 89 - 157 meters/sec (or about 200-350 miles/hour). Although such estimates may be correct, their accuracy is unknown because the speed of a diving falcon is difficult to measure. The required instrumention is complex, and the dive is a brief, rare event that takes place at unpredictable places and times (Tucker 1998). |
The high wing loading of birds like grebes, loons, and swans (see Tundra Swan below) means that it's more difficult for them to generate sufficient lift to take-off. That's why these birds often run along the surface of a lake for some distance before taking flight. They must generate enough speed to generate enough lift to get their relatively heavy bodies into the air!

Source: http://www.hedweb.com/animimag/swantako.htm
Albatross taking off (slow motion)
Mute Swans taking off in slow motion
 |
Take-off! -- Initiating flight is challenging, and considerable effort has focused on understanding the energetics and aerodynamics of take-off for both machines and animals. Available evidence suggests that birds maximize their initial flight velocity using leg thrust rather than wing flapping (e.g., see the drawings of a European Starling taking off from the ground below). The smallest birds, hummingbirds, are unique in their ability to perform sustained hovering but have small hindlimbs that could hinder generation of high leg thrust. During take-off by hummingbirds, Tobalske et al. (2004) measured hindlimb forces on a perch mounted with strain gauges and filmed wingbeat kinematics with high-speed video. Whereas other birds obtain 80–90% of their initial flight velocity using leg thrust, the leg contribution in hummingbirds was 59% during normal take-off. Unlike other species, hummingbirds beat their wings several times as they thrust using their hindlimbs. In a phylogenetic context, these results show that reduced body and hindlimb size in hummingbirds limits their peak acceleration during leg thrust and, ultimately, their take-off velocity. Previously, the influence of motivational state on take-off flight performance has not been investigated for any bird. Tobalske et al. (2004) studied the full range of motivational states by testing performance as the birds took off: (1) to initiate flight autonomously, (2) to escape a startling stimulus or (3) to aggressively chase a conspecific away from a feeder. Motivation affected performance. Escape and aggressive take-off featured decreased hindlimb contribution (46% and 47%, respectively) and increased flight velocity. When escaping, hummingbirds shortened their body movement prior to onset of leg thrust and began beating their wings earlier and at higher frequency. Thus, hummingbirds are capable of modulating their leg and wingbeat kinetics to increase take-off velocity. |
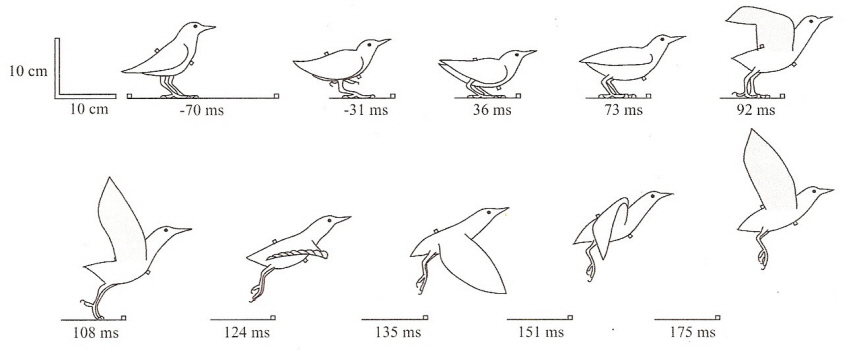
European Starling taking off from the ground. Time notations (milliseconds) are relative to the defined start of take-off (vertical
force > 105% of body weight). Key events: wings begin unfolding (73 ms) and start of downstroke (108 ms). From: Earls (2000).
European Starling taking off in slow motion
Rock Pigeon taking off in slow motion
 |
|
Mean distances between two bones in Diamond Doves and Zebra Finches during take-off. (a) Between the head and the trunk (dist_neck), and (b) between the most proximal
part of the trunk and the most distal point of the tarsometatarsus (dist_limb). Shading in the graphs represents the standard deviation across all trials (Figure from Provini and Abourachid 2018).

Tree Swallow landing
Photo by Anupam
Pal & used with his permission
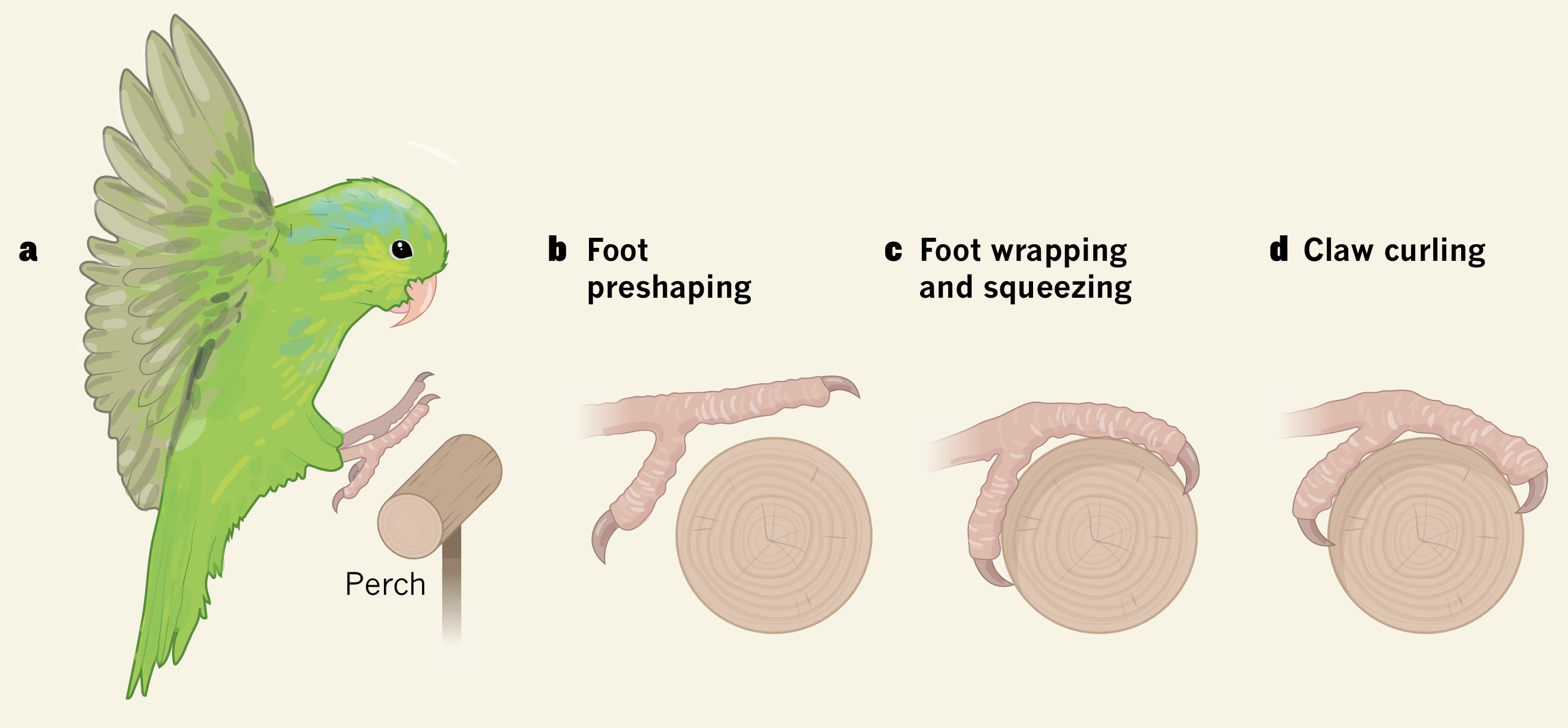
How a Pacific Parrotlet (Forpus coelestis) lands stably on a perch. (a) When a bird is about to land, its wings, body and legs
are positioned as describe above, i.e., increased angle of attack to reduce speed and lift, tails spread and lowerd to aid in breaking,
legs and feet are extended toes and claws are outstretched. (b) When the bird is close to making contact with the perch, its toes
begin to close (i.e., preshaping). (c) When the birds toes contact the perch, they wrap around it and squeeze it. (d) The claws then
begin to curl, which can be a superfast event (12 milliseconds) (From: Biewener 2019).
Rock Pigeon landing in slow motion - note the position of the alulas and the spread tail feathers
Bald Eagle landing
Eagle Owl landing
Birds landing on a bird feeder in slow motion
Swan landing
| Leading-edge vortex lifts swifts --
How do birds
fly up to a branch and land smoothly and precisely? It turns out that
they
may use a completely different kind of lift -- which not only works at
slow speeds, but even helps birds brake to a stop. Using a model of a
bird
wing in water containing particles lit with a laser, Videler et al.
(2004)
discovered how Common
Swifts (Apus apus) create lift with a "leading-edge vortex" (LEV). Think of an LEV as a horizontal tornado that forms above a
sharp,
swept-back wing as it cuts through the air. The vortex is a
low-pressure
zone. Like the low-pressure zone formed above conventional wings, it
creates
lift. Until this study, it had been seen in insects, but not birds.
Birds have two-part wings. The proximal "arm wing" is rounded on front, humped on top, and sharp on the back -- just like most airplane wings. Further away, the "hand wing" is flatter on top and extremely sharp on the front. The hand wing resembles the wing of a fighter plane, and it is also often swept back -- angled -- toward the rear. Wings on some high-performance jets can change angle to alter the leading-edge vortex. Wings that are nearly straight out create more lift. Swept-back wings create more drag (air friction). Acrobatic birds may also take advantage of the LEV; changing wing angle gives them the ratio of lift and drag they need for flying and snatching insects in mid-air. The LEV not only creates lift, especially at slow speeds, but also confers another benefit that helps the swift perform insectivorous aerobatics. While conventional lift is chiefly an upward force, the LEV can also produce drag, which allows sudden steering. "The LEV can be used for controlling flight," says Videler. "It's very suited for that because there is no time delay, the forces are produced instantaneously. That's very useful if you want to maneuver very quickly." -- Courtesy of the University of Wisconsin Board of Regents |
 Swifts hunt in the air, catching flying insects on the wing. To snag its prey, a swift has to be able to fly fast and make very tight turns, just like a jet fighter (From: Müller and Lentink 2004).  When gliding, a Common Swift shows a torpedo-shaped body. Its arm-wing (close to its body) has a rounded leading edge. The bird's long, slender hand-wing has a much sharper profile. The inset shows the feathers at the hand-wing's leading edge. |
As a hummingbird moves its wings forward and down, tiny vortices form over the leading and trailing edges
and then merge into a single large vortex, forming a low-pressure area that provides lift.
Literature cited:
Alerstam, T., M. Rosén, J. Bäckman, P. G. P. Ericson, and O. Hellgren. 2007. Flight speeds among bird species: allometric and phylogenetic effects. PLoS Biology 5: e197.
Alonso, P. D., A. C. Milner, R. A. Ketcham, M. J. Cookson and T. B. Rowe. 2004. The avian nature of the brain and inner ear of Archaeopteryx. Nature 430: 666 - 669.
Baier, D. B., S. M. Gatesy, and F. A. Jenkins. 2007. A critical ligamentous mechanism in the evolution of avian flight. Nature 445: 307-310.
Bajec, I. L., and F. H. Heppner. 2009. Organized flight in birds. Animal Behaviour 78: 777-789.
Biewener, A. A. 2019. Getting to grips with bird landing. Nature 574: 180-181.
Burgers, P. and L. M. Chiappe. 1999. The wings of Archaeopteryx as a primary thrust generator. Nature 399: 60-62.
Carey, J.R. and J. Adams. 2001. The preadaptive role of parental care in the evolution of avian flight. Archaeopteryx 19: 97 - 108.
Chatterjee, S. and R. J. Templin. 2007. Biplane wing planform and flight performance of the feathered dinosaur Microraptor gui. Proceedings of the National Academy of Science 104: 1576-1580.
Chen P., Z. Dong, and S. Zhen. 1998. An exceptionally well-preserved theropod dinosaur from the Yixian Formation of China. Nature. 391:147–152.
del Hoyo, J., A. Elliott, and J. Sargatal (eds.). 1992. Handbook of birds of the world, volume 1. Lynx Edicions, Barcelona, Spain.
Diaz-Salazar et al. 2024. Winging it: hummingbirds alter flying kinematics during molt. Biology Open 13: bio060370.
Dial, K. P. 1992. Avian forelimb muscles and nonsteady flight: can birds fly without using the muscles in their wings? Auk 109: 874-885.
Dial, K. P. 2003. Wing-assisted incline running and the evolution of flight. Science 299:402-404.
Earls, K. D. 2000. Kinematics and mechanics of ground take-off in the starling (Sturnus vulgaris) and the quail (Coturnix coturnix). Journal of Experimental Biology 203:725-739.
Feduccia, A. 1996. The origin and evolution of birds. Yale Univ. Press, New Haven.
Hedenström, A. 2002. Aerodynamics, evolution and ecology of avian flight. Trends in Ecology and Evolution 17: 415-422.
Hedenström, A. and F. Liechti. 2001. Field estimates of body drag coefficient on the basis of dives in passerine birds. Journal of Experimental Biology 204: 1167-1175.
Hedenstrom, A. and S. Sunada. 1999. On the aerodynamics of moult gaps in birds. Journal of Experimental Biology 202:67-76.
Hedrick, T. L., B. W. Tobalske, and A. A. Biewener. 2002.Estimates of circulation and gait change based on a three-dimensional kinematic analysis of flight in cockatiels (Nymphicus hollandicus) and Ringed Turtle-doves (Streptopelia risoria). Journal of Experimental Biology 205:1389-1409.
Lapsansky et al. 2022. High Wing-Loading Correlates with Dive Performance in Birds, Suggesting a Strategy to Reduce Buoyancy. Integrative and Comparative Biology 62: 878889.
Lee et al. 2015. The Function of the Alula in Avian Flight. Scientific Reports 5: 9914.
Lockwood, R., J. P. Swaddle, and J. M. V. Rayner. 1998. Avian wingtip shape reconsidered: wingtip shape indices and morphological adaptations to migration. Journal of Avian Biology 29: 273-292.
Longrich, N. 2006. Structure and function of hindlimb feathers in Archaeopteryx lithographica. Paleobiology 32: 417-431.
Müller, U. K. and David Lentink. 2004. Turning on a dime. Science 306: 1899 - 1900.
Naish, D. and D. M. Martill. 2003. Pterosaurs – a successful invasion of prehistoric skies. Biologist 50: 213-216.
Noberg, U. M. L. 2002. Structure, form, and function of flight in engineering and the living world. Journal of Morphology 252:52-81.
Palleroni, A., C. T. Miller, M. Hauser, and P. Marler. 2005. Predation: Prey plumage adaptation against falcon attack. Nature 434:973-974.
Pennycuick C. J., M. Klaassen, A. Kvist, and A. Lindström. 1996. Wingbeat frequency and the body drag anomaly: wind-tunnel observations on a thrush nightingale (Luscinia luscinia) and a teal (Anas crecca). Journal of Experimental Biology 199: 2757–2765.
Provini, P., and A. Abourachid. 2018. Whole-body 3D kinematics of bird take-off: key role of the legs to propel the trunk. Science of Nature 105:12.
Rader et al. 2020. Functional Morphology of Gliding Flight II. Morphology Follows Predictions of Gliding Performance. Integrative and Comparative Biology 60: 1297-1308.
Rader, J. A. and T. L. Hedrick. 2023. Morphological evolution of bird wings follows a mechanical sensitivity gradient determined by the aerodynamics of flapping flight. Nature Communications 14: Article number 7494.
Rayner, J. M. V. 1988. Form and function in avian flight. Current Ornithology 5: 1-66.
Ros, I. 2013. Low speed avian maneuvering flight. Ph.D. dissertation, Harvard University, Cambridge, MA.
Sanz, J. L., L. M. Chiappe, P. Perez-Moreno, A. D. Buscalioni, J. J. Moratalla, F. Ortega, & F. J. Payata-Ariza. 1996. An Early Cretaceous bird from Spain and its implications for the evolution of avian flight. Nature 382: 442-445.
Sanz, J. L. and F. Ortega. 2002. The birds from Las Hoyas. Science Progress 85:113-130.
Schluter D. 2001. Ecology and the origin of species. Trends in Ecology and Evolution. 16:372–380.
Seebacher, F. 2003. Dinosaur body temperatures: the occurrence of endothermy and ectothermy. Paleobiology 29: 105-122.
Speakman, J. R. 2001. The evolution of flight and echolocation in bats: another leap in the dark. Mammal Review 31: 111-130.
Sullivan, W. and K.-J. Wilson. 2001. Differences in habitat selection between Chatham Petrels (Pterodroma axillaris) and Broad-billed Prions (Pachyptila vittata): implications for management of burrow competition. New Zealand Journal of Ecology 25: 65-69.
Sumida S. S., and C. A. Brochu. 2000. Phylogenetic context for the origin of feathers. American Zoologist. 40:486–503.
Videler, J.J., E.J. Stamhuis, and G.D.E. Povel. 2004. Leading-edge vortex lifts swifts. Science 306:1960-1962.
Weimerskirch, H., O. Chastel, C. Barbraud, and O. Tostain. 2003. Frigatebirds ride high on thermals. Nature 421:333-334.
Wu et al. 2026. Critical innovations in the assembly of the modern f light apparatus in Early Cretaceous birds. iScience 29: 115506.